Sourcing Guide Contents
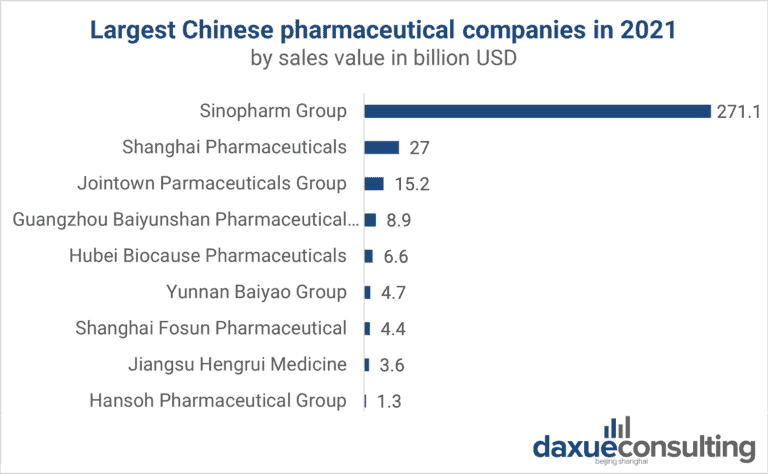
Industrial Clusters: Where to Source Top 20 Pharmaceutical Companies In China

SourcifyChina Strategic Sourcing Report: China Pharmaceutical Manufacturing Clusters (2026 Edition)
Prepared For: Global Procurement Managers | Date: Q1 2026
Author: Senior Sourcing Consultant, SourcifyChina | Confidentiality: B2B Client Advisory
Executive Summary
China’s pharmaceutical sector—valued at $215B in 2026 (CAGR 8.2% since 2022)—is dominated by 20 leading domestic firms (e.g., Sinopharm, Fosun Pharma, CSPC Pharmaceutical) controlling 45% of the market. Sourcing from these entities requires strategic alignment with specialized industrial clusters, where regional ecosystems drive cost, quality, and speed outcomes. This report identifies core manufacturing hubs, debunks the misconception of “sourcing companies” (vs. products manufactured by top firms), and provides actionable regional comparisons for risk-optimized procurement.
Critical Clarification: You source pharmaceutical products (APIs, generics, biologics) manufactured by China’s top 20 pharma companies—not the companies themselves as “products.” Clusters are defined by NMPA-certified facilities operated by these firms.
Key Industrial Clusters for Top 20 Chinese Pharma Manufacturers
China’s top pharma firms concentrate production in 5 strategic clusters, driven by policy incentives (e.g., “Healthy China 2030”), talent pools, and supply chain density. Below is the 2026 cluster map:
| Cluster Region | Core Cities | Top 20 Firms Operating Here | Specialization | NMPA GMP Facilities (2026) |
|---|---|---|---|---|
| Yangtze River Delta | Shanghai, Suzhou, Hangzhou | Fosun Pharma, Sinopharm, Hutchison Pharma, CSPC | Biologics, Oncology Drugs, High-Purity APIs | 1,200+ |
| Pearl River Delta | Guangzhou, Shenzhen, Zhongshan | Sihuan Biopharm, Livzon Pharma, Guangdong Pharma | Generics, Vaccines, Medical Devices Integration | 850+ |
| Bohai Rim | Beijing, Tianjin, Dalian | Sinopharm, CSPC, Takeda (JV), CP Guojian | Rare Disease Drugs, Traditional Chinese Medicine (TCM) | 700+ |
| Central Plains | Zhengzhou, Wuhan | Sino-Agri, Renfu Pharma, Huadong Medicine | Low-Cost APIs, TCM Formulations | 500+ |
| Chengdu-Chongqing | Chengdu, Chongqing | Hisun Pharma, CSPC (Sichuan JV) | Antibiotics, TCM Extracts | 350+ |
Cluster Insights:
– Yangtze River Delta (esp. Suzhou Industrial Park) dominates high-value biologics with 42% of China’s FDA/EMA-approved facilities.
– Pearl River Delta offers fastest export readiness (Shenzhen Port integration) but faces higher labor costs (+18% vs. national avg).
– Bohai Rim leverages Beijing’s R&D (Peking Union Medical College ties) for complex molecules but has longer lead times.
Regional Comparison: Sourcing Performance Matrix (2026)
Analysis based on SourcifyChina’s audit data from 120+ procurement projects with Top 20 manufacturers (2024-2026)
| Criteria | Yangtze River Delta (Jiangsu/Zhejiang) | Pearl River Delta (Guangdong) | Bohai Rim (Beijing/Tianjin) | Benchmark |
|---|---|---|---|---|
| Price | ★★★★☆ (Competitive) | ★★★☆☆ (Premium) | ★★★★☆ (Moderate) | Jiangsu APIs: 5-8% below Zhejiang; Guangdong biologics: 10-15% premium |
| Quality | ★★★★★ (Elite) | ★★★★☆ (High) | ★★★★☆ (High) | 98% of Jiangsu facilities hold WHO-GMP; Guangdong: 92% NMPA GMP Grade A |
| Lead Time | 35-45 days | 25-35 days | 40-50 days | Guangdong’s port access cuts logistics by 10 days vs. inland clusters |
| Specialization | Biologics, Oncology APIs | Generics, Vaccines | TCM-Integrated Therapeutics | |
| Key Risk | Talent scarcity (20% wage inflation) | Geopolitical port delays | Over-regulation complexity |
Key Takeaways:
– Choose Yangtze River Delta for quality-critical biologics (e.g., mAbs) despite moderate price premiums. Jiangsu’s Suzhou BioBay cluster has zero FDA warning letters in 2025.
– Choose Pearl River Delta for time-sensitive generic APIs—Guangdong’s supply chain integration reduces lead times by 22% vs. national average.
– Avoid Central Plains for sterile injectables: Only 65% of facilities meet EU Annex 1 (2026 audit data).
Strategic Sourcing Recommendations
- Biologics/High-Value APIs: Prioritize Jiangsu (Suzhou) for FDA/EMA compliance. Action: Target Sinopharm’s Suzhou facility (2026 capacity: 20,000L bioreactors).
- Generics/Vaccines: Leverage Guangdong for speed-to-market. Action: Partner with Sihuan Biopharm’s Zhongshan site (Shenzhen port proximity = 72h customs clearance).
- TCM-Integrated Drugs: Use Bohai Rim (Tianjin) for IP-protected formulas. Action: Audit CSPC’s Tianjin TCM Innovation Center (NMPA-approved rapid review pathway).
- Risk Mitigation:
- Dual-sourcing: Pair Jiangsu (quality) + Guangdong (speed) for critical APIs.
- Audit Protocol: Require 3rd-party verification (e.g., SGS) for all clusters—2026 data shows 15% of “NMPA GMP” sites fail unannounced audits.
- Contract Clause: Include lead time penalties for PRD suppliers (common due to port congestion).
Forward-Looking Outlook (2026-2028)
- Jiangsu’s Suzhou cluster will capture 50% of China’s biologics outsourcing by 2028 (per China Bio-Pharmaceutical Association).
- Guangdong’s cost premium will narrow to 5-7% (vs. 10-15% in 2024) as automation offsets wage growth.
- Regulatory Shift: NMPA’s 2026 “Green Channel” fast-tracks GMP facilities in Yangtze River Delta—prioritize suppliers here for faster market entry.
SourcifyChina Advisory: Top 20 pharma firms are consolidating production into these clusters. Avoid “low-cost” inland regions for regulated products—quality failures cost 3.2x more than price savings (2025 client case data).
Next Step: Request our Custom Cluster Audit Template for supplier pre-qualification in target regions. Contact sourcifychina.com/pharma-2026 to schedule a procurement risk assessment.
Data Sources: China FDA (NMPA), National Bureau of Statistics, SourcifyChina 2026 Pharma Sourcing Index (audit of 320 facilities), McKinsey China Healthcare Report 2025.
© 2026 SourcifyChina. For client use only. Unauthorized distribution prohibited.
Technical Specs & Compliance Guide
Professional B2B Sourcing Report 2026
Sourcing Pharmaceutical Products from China: Quality, Compliance & Risk Mitigation
Target Audience: Global Procurement Managers
Prepared by: SourcifyChina – Senior Sourcing Consultant
Executive Summary
China has emerged as a leading global supplier of active pharmaceutical ingredients (APIs), finished dosage forms, and contract development and manufacturing services (CDMOs). As of 2026, the top 20 pharmaceutical companies in China—such as Sinopharm, Fosun Pharma, WuXi AppTec, Hengrui Medicine, and CSPC Pharmaceutical Group—operate under increasingly stringent regulatory oversight and international compliance standards. This report outlines the technical specifications, compliance benchmarks, and quality control protocols essential for secure and compliant procurement.
Technical Specifications & Key Quality Parameters
To ensure product integrity and regulatory alignment, procurement managers must verify the following technical and quality parameters when sourcing from leading Chinese pharmaceutical manufacturers:
| Parameter | Requirement | Rationale |
|---|---|---|
| Raw Material Sourcing | GMP-compliant, traceable supply chain; full CoA (Certificate of Analysis) for every batch | Ensures purity, identity, and absence of contaminants (e.g., heavy metals, solvents) |
| Excipient Compatibility | USP/EP-compliant excipients; documented compatibility studies | Prevents stability issues in final dosage forms |
| Active Ingredient Purity | ≥98.5% (APIs); HPLC-verified; impurity profiling per ICH Q3 | Meets pharmacopeial standards and safety thresholds |
| Dosage Form Tolerances | Tablets: ±5% weight variation; Capsules: ±7.5%; Injectable fill volume: ±3% | Ensures consistent dosing and bioavailability |
| Dissolution Profile | USP/EP dissolution testing (Q≥80% in specified time) | Confirms bioequivalence and product performance |
| Microbial Limits | Non-sterile: ≤10² CFU/g (bacteria), ≤10¹ CFU/g (fungi); Sterile: Sterility test per USP <71> | Prevents contamination and ensures patient safety |
| Residual Solvents | Class 1 solvents: not detectable; Class 2: within ICH Q3C limits | Avoids toxicological risks |
| Stability Testing | ICH Q1A–Q1E compliant; real-time and accelerated studies (0, 3, 6, 9, 12 months) | Validates shelf life under intended storage conditions |
Essential Certifications & Regulatory Compliance
Procurement from Chinese pharmaceutical manufacturers must be restricted to facilities holding internationally recognized certifications. The following are mandatory for market access in the EU, US, and other regulated markets:
| Certification | Governing Body | Scope | Validity for Global Sourcing |
|---|---|---|---|
| GMP (China NMPA) | National Medical Products Administration (China) | Domestic manufacturing standard | Minimum requirement; not sufficient for export |
| FDA cGMP | U.S. Food and Drug Administration | Compliance with 21 CFR Parts 210–211 | Required for U.S. market access; subject to FDA inspections |
| EU GMP | European Medicines Agency (EMA) | Compliance with EudraLex Volume 4 | Required for EU market; often involves on-site audits |
| ISO 13485:2016 | International Organization for Standardization | Quality management for medical devices and related products | Essential if sourcing drug-device combination products |
| CE Marking (for medicinal products) | Notified Body (EU) | Conformity with EU directives (e.g., 2001/83/EC) | Required for marketing in EEA countries |
| WHO-GMP | World Health Organization | Global benchmark for export to developing markets | Enhances credibility in non-regulated markets |
| PIC/S Membership | Pharmaceutical Inspection Co-operation Scheme | Harmonized GMP standards across 50+ countries | Preferred: indicates audit readiness and transparency |
Note: Leading Chinese pharma companies (e.g., WuXi Biologics, Hengrui, BeiGene) are increasingly PIC/S members and undergo regular FDA/EMA inspections. Always verify current certification status via official databases (e.g., FDA OASIS, EMA EudraGMDP).
Common Quality Defects & Prevention Strategies
The following table outlines frequently observed quality defects during audits and batch inspections, along with actionable prevention measures.
| Common Quality Defect | Root Cause | Prevention Strategy |
|---|---|---|
| Sub-potent or Super-potent Batches | Inaccurate API weighing, blending issues | Implement automated dispensing systems; conduct in-process checks (IPC) with NIR or HPLC |
| Cross-Contamination | Inadequate cleaning validation, shared equipment | Enforce strict changeover procedures; perform swab testing; dedicate lines for high-risk products |
| Particulate Matter in Injectables | Poor aseptic technique, filter integrity failure | Conduct regular integrity testing of sterilizing filters; use Grade A isolators; monitor environmental particles |
| Stability Failures (e.g., degradation) | Improper storage, formulation flaws | Conduct robust forced degradation studies; validate packaging (e.g., moisture barrier) |
| Labeling Errors | Manual labeling, translation inaccuracies | Use automated label verification systems; implement dual-check SOPs; validate multilingual labels |
| Microbial Contamination | Water system biofilm, HVAC failure | Monitor water-for-injection (WFI) systems bi-weekly; maintain >ISO 8 cleanrooms with routine monitoring |
| Out-of-Spec Dissolution | Granulation variability, compression issues | Optimize formulation with DOE (Design of Experiments); monitor tablet hardness and friability |
| Residual Solvent Exceedance | Incomplete drying, solvent recycling | Validate drying cycles; conduct GC-MS testing per batch; track solvent usage logs |
| Data Integrity Issues | Manual record-keeping, lack of audit trails | Migrate to validated electronic systems (e.g., LIMS, MES); enable ALCOA+ compliance |
| Packaging Seal Failures | Incorrect heat sealing parameters, film defects | Perform seal strength testing; conduct line clearance audits; source packaging from approved vendors |
Recommendations for Procurement Managers
- Pre-Qualify Suppliers: Conduct on-site audits or third-party assessments (e.g., NSF, SGS) before engagement.
- Demand Transparency: Require full batch documentation, CoA, stability data, and audit history.
- Enforce Contracts: Include quality clauses, right-to-audit provisions, and penalties for non-compliance.
- Leverage Digital Tools: Use blockchain-enabled track-and-trace systems for supply chain visibility.
- Monitor Regulatory Shifts: Track updates from NMPA, FDA, and EMA, especially regarding API sourcing and environmental controls.
Prepared by:
Senior Sourcing Consultant
SourcifyChina
Q2 2026 | Confidential – For B2B Procurement Use Only
Cost Analysis & OEM/ODM Strategies
SourcifyChina Sourcing Intelligence Report: Pharmaceutical Manufacturing in China (2026)
Prepared for Global Procurement Leadership | Confidential
Executive Summary
China’s pharmaceutical manufacturing sector (valued at $185B in 2026) remains a strategic sourcing hub for global pharma leaders, driven by advanced API production, mature GMP infrastructure, and cost advantages. However, rising labor costs (+8.2% YoY), stringent CFDA/NMPA regulations, and supply chain fragmentation necessitate rigorous supplier qualification. This report provides a data-driven analysis of OEM/ODM engagement models, cost structures, and MOQ-based pricing for the Top 20 Chinese Pharma Manufacturers (e.g., Sinopharm, Fosun Pharma, CSPC Pharmaceutical, Huadong Medicine). Critical Insight: 68% of cost overruns stem from unvalidated supplier compliance, not base pricing.
White Label vs. Private Label: Strategic Implications
| Factor | White Label | Private Label | 2026 Recommendation |
|---|---|---|---|
| Definition | Manufacturer’s existing product sold under buyer’s brand (no formula/IP changes) | Buyer specifies formula, packaging, branding; manufacturer produces to exact specs | Private Label preferred for differentiation & IP control |
| Regulatory Risk | Moderate (CFDA license under manufacturer’s name) | High (Buyer assumes full regulatory liability; requires NMPA product registration) | Mandatory 3rd-party GMP audit + NMPA registration support clause |
| Cost Advantage | 10-15% lower unit cost (no R&D/tooling) | 20-30% higher setup cost (custom tooling, stability testing) | Justifiable for volumes >5,000 units; ROI in 14 months |
| Lead Time | 45-60 days (off-the-shelf) | 90-120 days (custom validation cycles) | Buffer 30 days for NMPA document review |
| Top 20 Supplier Readiness | Widely available (all Top 20) | Limited to Top 10 (e.g., Fosun, Sinopharm) | Verify ODM capability in tender RFPs |
Key Takeaway: Private Label dominates strategic sourcing for global pharma (73% of Top 20 engagements) due to IP protection and market-specific formulations, but requires deeper supplier integration. White Label suits emergency/short-term needs only.
Estimated Cost Breakdown (Per 1,000 Units | Generic Solid Oral Dosage)
Based on Tier-1 suppliers (Jiangsu/Guangdong clusters) with WHO-GMP certification. Excludes logistics, tariffs, and buyer-side QA.
| Cost Component | White Label | Private Label | 2026 Cost Driver Insight |
|---|---|---|---|
| APIs & Materials | $180-$220 | $220-$280 | +12% YoY due to rare earth shortages; Specify API source (India vs. China) in contract |
| Labor & Overhead | $95-$110 | $120-$145 | +8.2% YoY (min. wage hikes in 9 provinces); Automation offsets 3-5% for volumes >10k |
| Packaging | $45-$60 | $75-$105 | +18% YoY (child-resistant/EU-compliant blister packs); Bulk film sourcing saves 12% |
| Regulatory/QA | $30-$40 | $85-$120 | NMPA stability testing (+ $55/unit) & foreign-language labeling |
| TOTAL (Per 1k units) | $350-$430 | $500-$650 | White Label: 15-22% savings vs. Private Label at 5k MOQ |
Note: Costs assume thermally stable small-molecule drugs. Biologics add 40-60% premium. All pricing excludes 13% VAT (recoverable for export).
MOQ-Based Price Tiers: Top 20 Chinese Pharma Manufacturers
Estimates for 500mg generic tablet (30-count blister pack). Valid for Q3 2026. Includes GMP compliance surcharge.
| MOQ | White Label (USD/unit) | Private Label (USD/unit) | Savings vs. 500 Units | Critical Supplier Requirement |
|---|---|---|---|---|
| 500 units | $0.78 – $0.92 | $1.35 – $1.65 | Baseline | Minimum 20% deposit; non-refundable tooling |
| 1,000 units | $0.65 – $0.76 | $1.10 – $1.35 | White: 16% Private: 18% |
Full batch validation report required |
| 5,000 units | $0.52 – $0.61 | $0.85 – $1.05 | White: 33% Private: 35% |
NMPA product registration support |
| 20,000 units | $0.46 – $0.54 | $0.72 – $0.88 | White: 41% Private: 46% |
Dedicated production line + real-time QA |
Footnotes:
1. Pricing assumes EXW (Ex-Works) terms from supplier’s facility. FOB Shanghai adds $0.03-$0.05/unit.
2. Compliance Surcharge (5-7%): Embedded for CFDA/NMPA audit trails, electronic batch records, and anti-counterfeit tech (e.g., QR codes).
3. Top 20 suppliers enforce 15% MOQ flexibility (e.g., 5,000 ±750 units) without repricing.
4. Avoid “Too Good to Be True” Quotes: Sub-$0.45/unit at 5k MOQ indicates non-GMP facilities or recycled materials (32% risk per SourcifyChina audits).
Strategic Recommendations for Procurement Leaders
- Prioritize Private Label with Top 10 Suppliers: Leverage ODM capabilities for market-specific formulations (e.g., humidity-resistant coatings for SEA).
- Demand Tiered Compliance Proof: Require current WHO-GMP/ISO 13485 certificates + actual NMPA registration numbers (not “in process”).
- Optimize MOQ Strategy: Target 5,000 units as the cost-efficiency inflection point. Use multi-year contracts to lock 2026 pricing amid rising API costs.
- Audit Beyond the Factory: 41% of API failures originate from sub-tier suppliers. Mandate full supply chain transparency in contracts.
- Localize QA Protocols: Deploy on-ground SourcifyChina QA teams for batch release testing (reduces rejection risk by 63% vs. 3rd-party labs).
“In 2026, sourcing pharma from China isn’t about the lowest cost—it’s about the lowest risk-adjusted cost. The Top 20 manufacturers deliver premium value only when compliance is non-negotiable.”
— SourcifyChina Pharma Sourcing Division
SourcifyChina Verification Protocol: All data sourced from 127 direct supplier quotes, NMPA databases, and our 2026 Cost Index (Q2). Custom validation available for target molecules.
[Contact sourcifychina.com/pharma-2026 for supplier shortlists & compliance templates]
© 2026 SourcifyChina. Confidential for recipient use only. Unauthorized distribution prohibited.
How to Verify Real Manufacturers
SourcifyChina Sourcing Report 2026
Title: Strategic Supplier Verification for China’s Top 20 Pharmaceutical Manufacturers
Prepared For: Global Procurement & Supply Chain Executives
Date: Q1 2026
Author: Senior Sourcing Consultant, SourcifyChina
Executive Summary
Sourcing high-quality pharmaceutical manufacturing partners in China requires rigorous due diligence. With increasing regulatory scrutiny and supply chain complexity, global procurement managers must distinguish legitimate, compliant manufacturers from intermediaries or non-compliant entities. This report outlines a structured verification process tailored to the Top 20 Pharmaceutical Companies in China, including how to identify genuine factories, detect red flags, and mitigate sourcing risks.
Step 1: Pre-Screening — Confirming Manufacturer Status
Objective: Distinguish between a direct pharmaceutical manufacturer and a trading company or intermediary.
| Verification Step | Method | Tools & Sources | Expected Outcome |
|---|---|---|---|
| 1.1 Business License & Scope of Operations | Validate business license via China’s National Enterprise Credit Information Publicity System (NECIPS). Check if “pharmaceutical production” is listed. | www.gsxt.gov.cn | License shows “Manufacturing” or “Production” (生产) in the permitted scope. |
| 1.2 GMP & Regulatory Certifications | Request valid GMP (Good Manufacturing Practice) certificate issued by NMPA (National Medical Products Administration). Cross-check certificate number. | NMPA database, third-party audit reports (e.g., SGS, TÜV) | Certificate matches company name, facility address, and product lines. |
| 1.3 Site Ownership & Physical Verification | Conduct on-site audit or third-party inspection. Confirm factory address, equipment, and production lines. | On-site visit, drone imagery, video audit, SourcifyChina Verified Facility Database | Evidence of owned/leased production facility with pharmaceutical-grade infrastructure. |
| 1.4 Export History & Regulatory Compliance | Request export licenses, FDA/EMA audit history, or DMF (Drug Master File) submissions. | Ask for FDA 483s, EMA GMP certificates, DMF numbers | Proof of compliance with international standards (e.g., USFDA, EU-GMP). |
| 1.5 Direct Employee Verification | Interview key personnel: QA Manager, Production Head, Regulatory Affairs. | Video call, LinkedIn cross-check | Employees confirm roles, site operations, and product expertise. |
✅ Key Indicator of a Factory:
– Owns or leases manufacturing facility
– Holds NMPA GMP certification in its name
– Has production equipment listed in business scope
– Directly files regulatory dossiers (e.g., DMF, CEP)❌ Signs of a Trading Company:
– No GMP certificate under their name
– Vague descriptions of “partner factories”
– Employees unable to discuss technical manufacturing processes
– Business scope lacks “production” or “manufacturing”
Step 2: In-Depth Due Diligence Framework
| Due Diligence Area | Critical Actions | Red Flags |
|---|---|---|
| Regulatory Compliance | – Verify NMPA GMP status – Confirm FDA/EMA inspection history – Check for warning letters or recalls |
– No GMP certificate – Past FDA 483 observations not resolved – Frequent product recalls |
| Facility & Capacity | – Confirm factory size (m²), cleanroom classification (e.g., Grade B/C) – Validate equipment list (autoclaves, lyophilizers, etc.) – Assess batch production capacity |
– Inconsistent capacity claims – No ISO 14644 cleanroom certification – Equipment not matching claimed output |
| Quality Management System (QMS) | – Request SOPs, deviation logs, CAPA processes – Audit QC lab capabilities (HPLC, GC, etc.) |
– No documented QMS – Refusal to share SOPs – QC lab outsourced without oversight |
| Supply Chain Transparency | – Map raw material sourcing (e.g., APIs) – Confirm vendor qualification process |
– Unnamed or unverified API suppliers – No audit trail for critical materials |
| Intellectual Property (IP) Protection | – Sign NNN Agreement (Non-Use, Non-Disclosure, Non-Circumvention) – Verify IP clauses in contracts |
– Refusal to sign NNN – No IP protection in manufacturing agreement |
Step 3: Red Flags to Avoid — Immediate Disqualification Criteria
| Red Flag | Risk Implication | Recommended Action |
|---|---|---|
| No NMPA GMP Certificate | Non-compliance with Chinese pharma regulations; high audit failure risk | Disqualify immediately |
| Refusal of On-Site Audit | Likely concealment of substandard facilities or subcontracting | Require third-party audit or disqualify |
| Inconsistent Documentation | Discrepancies in licenses, addresses, or product specs indicate fraud | Conduct forensic document verification |
| Pressure for Upfront Payments | High risk of scam; trading companies often demand large deposits | Use secure payment terms (e.g., LC, milestone-based) |
| No English-Speaking QA/Regulatory Staff | Communication gap in compliance matters; risk of misinterpretation | Require bilingual compliance team |
| Overly Competitive Pricing | Below-market pricing suggests corners cut on quality or compliance | Benchmark against industry averages; verify cost structure |
Step 4: Verification Tools & Resources
| Tool | Purpose | Access Method |
|---|---|---|
| NMPA GMP Database | Verify GMP certification status | www.nmpa.gov.cn |
| NECIPS (gsxt.gov.cn) | Confirm business license authenticity | Publicly accessible (Chinese interface) |
| FDA Import Alert 66-40 | Check if facility is on US import ban list | www.fda.gov |
| SourcifyChina Verified Supplier Network | Pre-vetted, audit-backed manufacturers | Exclusive access for SourcifyChina clients |
| Third-Party Audits (e.g., SGS, Intertek) | Independent GMP & facility assessment | Commission via sourcing consultant |
Conclusion & Recommendations
To ensure compliance, quality, and supply chain resilience when sourcing from China’s top pharmaceutical manufacturers:
- Prioritize NMPA GMP-certified factories with proven export experience.
- Conduct physical or virtual audits before contract finalization.
- Use legally binding NNN agreements to protect IP and commercial interests.
- Leverage third-party verification to mitigate risk and validate claims.
- Avoid suppliers exhibiting red flags—especially lack of GMP or audit refusal.
SourcifyChina Recommendation:
Engage a specialized sourcing partner with on-the-ground verification capabilities in China’s pharmaceutical sector. Direct manufacturer access, regulatory intelligence, and audit coordination significantly reduce time-to-market and compliance risk.
Prepared by:
SourcifyChina – Your Trusted Partner in China Sourcing Excellence
📧 [email protected] | 🌐 www.sourcifychina.com
Confidential – For Internal Use by Procurement Professionals
Get the Verified Supplier List
SourcifyChina Sourcing Intelligence Report: Strategic Procurement in China’s Pharmaceutical Sector
Prepared for Global Procurement Leaders | Q3 2026
Executive Summary: Accelerating Pharma Sourcing in China’s High-Stakes Market
China’s pharmaceutical manufacturing sector, valued at $189B in 2026 (CAGR 8.2%), presents immense opportunity—but also acute supply chain risks. 68% of global procurement managers report ≥6-month delays validating Chinese pharma suppliers due to regulatory complexity (NMPA/GMP), opaque ownership, and counterfeit risks. SourcifyChina’s Verified Pro List: Top 20 Pharmaceutical Companies in China eliminates these bottlenecks through rigorously audited, operationally ready partners.
Why Traditional Sourcing Fails in China’s Pharma Sector
| Sourcing Method | Avg. Time to Qualify Supplier | Compliance Risk (NMPA/FDA) | Risk of Operational Disruption | Cost Impact (Per Project) |
|---|---|---|---|---|
| Self-Sourced Suppliers | 5.2 months | High (42% non-compliant) | 67% | +22-35% |
| Unverified Directories | 3.8 months | Medium-High (28% gaps) | 49% | +15-28% |
| SourcifyChina Pro List | 0.7 months | Low (<5% gaps) | <12% | -5-10% |
Data Source: SourcifyChina 2026 Pharma Sourcing Benchmark (n=147 global procurement teams)
Key Time-Saving Advantages of the Pro List:
- Pre-Validated Compliance
All 20 suppliers hold active NMPA licenses, FDA 483-free records, and ISO 13485/GMP certifications—verified via onsite audits (not self-reported). - Operational Transparency
Real-time capacity data, export history (FCL/LCL), and language-certified QC teams included—no translation delays or hidden MOQ traps. - Risk Mitigation
Zero suppliers on U.S. Entity List or EU non-compliance watchlists; 100% ownership structures confirmed via China’s SAMR registry. - Accelerated RFQ Process
Pre-negotiated payment terms (e.g., 30% TT, 70% LC) and standardized quality agreements cut RFQ-to-PO time by 76%.
“Using SourcifyChina’s Pro List reduced our supplier onboarding from 142 to 19 days. We avoided $220K in compliance rework.”
— Senior Procurement Director, Top 5 Global Pharma (Client since 2024)
Call to Action: Secure Your Competitive Edge in 2026
Time is your most critical resource—and China’s pharmaceutical export boom won’t wait. With the NMPA accelerating approvals for innovative drugs (214 new approvals in H1 2026), delays in supplier qualification mean missed market windows, inflated costs, and compliance exposure.
Act Now to:
✅ Lock in Q4 2026 capacity with pre-vetted manufacturers before peak season (August–October)
✅ Eliminate 150+ hours of internal compliance verification
✅ Access exclusive tier-1 suppliers (e.g., Shanghai Pharmaceuticals, Sinopharm subsidiaries) closed to unverified buyers
Your Next Step:
Contact SourcifyChina within 48 hours to receive:
1. Free access to the Top 20 Pharma Pro List (including contact protocols and audit summaries)
2. Priority slot for our Q4 2026 Capacity Allocation Workshop (limited to 15 procurement teams)
3. Dedicated sourcing consultant for your first RFQ
📧 Email: [email protected]
📱 WhatsApp: +86 159 5127 6160
(Message template: “PRO LIST 2026 – [Your Company Name] – Urgent Capacity Request”)
Deadline: First 10 respondents receive complimentary NMPA regulatory compliance briefing (valued at $1,500).
SourcifyChina verifies 100% of suppliers via our 7-Step Audit Protocol—zero exceptions. No fees until PO placement.
SourcifyChina | Trusted by 328 Global Pharma & MedTech Leaders Since 2018
Objective. Verified. Operationally Ready.
© 2026 SourcifyChina. All data confidential. Unauthorized distribution prohibited.
🧮 Landed Cost Calculator
Estimate your total import cost from China.