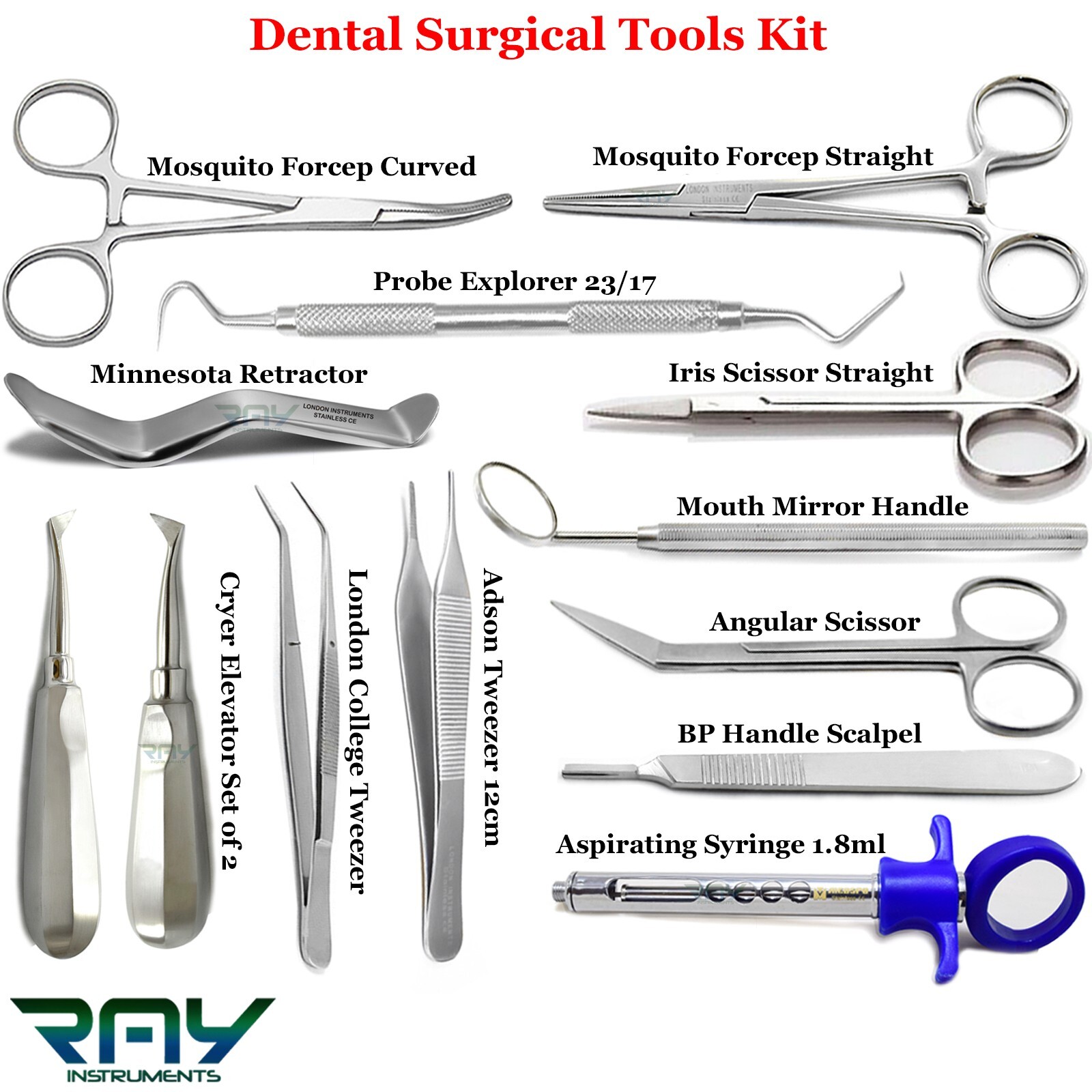
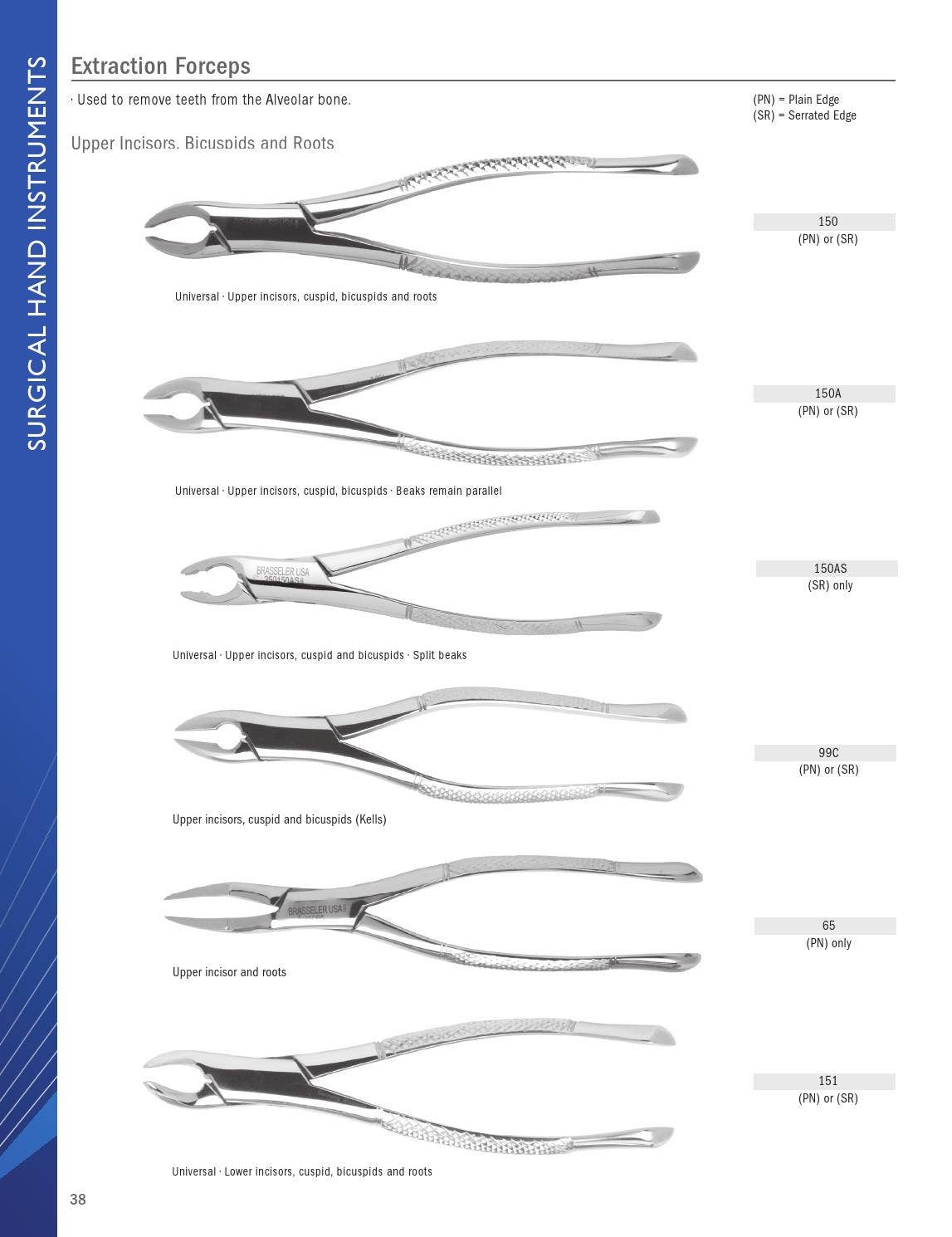
The global dental instruments market is witnessing steady expansion, driven by rising prevalence of dental disorders, increasing demand for dental surgeries, and advancements in dental technology. According to a report by Mordor Intelligence, the global dental instruments market was valued at USD 12.7 billion in 2023 and is projected to grow at a CAGR of 7.2% from 2024 to 2029. Tooth extraction, a common dental procedure, relies heavily on specialized surgical instruments such as elevators, forceps, luxators, and periosteal elevators. This growing procedural volume has spurred demand for high-quality, precision-engineered extraction tools, intensifying competition among manufacturers. As dental practitioners prioritize ergonomics, durability, and infection control, leading companies are investing in innovative designs and sterilization-compatible materials. In this evolving landscape, nine manufacturers have emerged as key players, combining legacy, technological advancement, and global distribution networks to dominate the tooth extraction instruments segment.
Top 9 Tooth Extraction Instruments Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Practicon, Inc.
Domain Est. 1998
Website: practicon.com
Key Highlights: Free delivery 30-day returnsDiscover high-quality dental supplies at Practicon Inc. From innovative tools to everyday essentials, we provide dental professionals with top-tier prod…
#2 W&H Impex Inc
Domain Est. 1995
Website: wh.com
Key Highlights: Supplier. Dental tools & supplies made by W&H. The international W&H group is a global leader in the development and manufacture of high-end dental tools….
#3 Dental Forceps & Extraction Tools
Domain Est. 1995
Website: henryschein.com
Key Highlights: Henry Schein Dental offers a vast array of universal and cowhorn dental forceps for all types of tooth extractions — from upper/lower molars and incisors to ……
#4 to Straumann
Domain Est. 1996
Website: straumann.com
Key Highlights: Straumann® – the world-leading brand for confidence in esthetic dentistry….
#5 Dental Instruments, Devices and Materials
Domain Est. 1997
Website: parkell.com
Key Highlights: Parkell manufactures a variety of high quality, top performing dental instruments, devices and materials for the problem solving dental professional….
#6 Karl Schumacher
Domain Est. 2002
Website: karlschumacher.com
Key Highlights: Karl Schumacher manufactures dental instruments that are known for their precision, durability, and ergonomic design. We use the highest quality materials and ……
#7 Brasseler USA
Domain Est. 2013
Website: brasselerusadental.com
Key Highlights: At Brasseler USA, we design and provide the highest-quality dental instrumentation to healthcare professionals….
#8 Dentsply Sirona USA
Domain Est. 2015
Website: dentsplysirona.com
Key Highlights: Welcome to the Dentsply Sirona site for dental professionals. Discover, shop & learn about world-class dental innovations, products, services and CE ……
#9 TBS Dental
Domain Est. 2019
Website: tbsdental.com
Key Highlights: We create premium, modern instruments designed to improve efficiency in dentistry, with innovative solutions for wide range of dental procedures….
Expert Sourcing Insights for Tooth Extraction Instruments

H2: 2026 Market Trends for Tooth Extraction Instruments
The global tooth extraction instruments market is poised for significant transformation by 2026, driven by advancements in dental technology, increasing prevalence of dental disorders, and growing demand for minimally invasive procedures. As healthcare systems continue to evolve and digital integration becomes standard in dental practices, several key trends are expected to shape the market dynamics for tooth extraction instruments.
-
Technological Advancements and Automation
By 2026, the integration of smart technologies such as robotics and AI-assisted diagnostics is anticipated to revolutionize tooth extraction procedures. Automated extraction systems equipped with precision sensors and digital imaging compatibility are expected to enhance accuracy, reduce surgical time, and minimize patient discomfort. Instruments with ergonomic designs and improved material durability—such as titanium-coated or anti-corrosive stainless steel tools—are gaining traction, particularly in advanced healthcare markets. -
Rising Demand for Minimally Invasive Procedures
There is a growing preference for atraumatic tooth extraction techniques that preserve surrounding bone and soft tissue. This trend is fueling demand for advanced forceps, periotomes, and luxators designed for controlled root elevation and socket preservation. Instruments compatible with piezoelectric surgery devices, which allow precise bone cutting with minimal damage to adjacent tissues, are expected to see increased adoption, especially in cosmetic and implant dentistry. -
Expansion of Dental Care Access in Emerging Markets
Emerging economies in Asia-Pacific, Latin America, and Africa are witnessing rapid growth in dental infrastructure and insurance coverage, contributing to higher volumes of dental surgeries. As a result, the demand for cost-effective, high-quality extraction instruments is rising. Local manufacturing and partnerships with global suppliers are expected to expand the availability of reliable instruments in these regions by 2026. -
Emphasis on Infection Control and Single-Use Instruments
Heightened awareness of infection control, particularly in the post-pandemic era, is accelerating the shift toward single-use or disposable extraction instruments. While reusable instruments remain dominant due to cost and sustainability concerns, innovations in sterilization-resistant materials and tracking systems (e.g., RFID tagging) are enhancing safety and compliance in reusable tool management. -
Growth in Geriatric and Cosmetic Dentistry
The aging global population is experiencing higher rates of periodontal disease and tooth loss, increasing the need for extractions. Concurrently, the rise in dental implant procedures requires precise tooth removal, boosting demand for specialized extraction kits. This dual influence is expected to drive product innovation and market segmentation in extraction instruments by 2026. -
Regulatory Standards and Market Consolidation
Stricter regulatory requirements in North America and Europe are pushing manufacturers to comply with ISO and FDA standards, influencing product design and quality control. Market consolidation through mergers and acquisitions among dental equipment companies is anticipated to intensify competition and foster innovation, particularly in premium instrument segments.
In conclusion, the 2026 tooth extraction instruments market will be characterized by technological sophistication, regional diversification, and a strong focus on precision, safety, and patient comfort. Stakeholders who invest in R&D, digital integration, and emerging market expansion are likely to gain a competitive edge in this evolving landscape.

Common Pitfalls Sourcing Tooth Extraction Instruments (Quality, IP)
Sourcing dental instruments, especially critical tools like tooth extraction forceps, elevators, and luxators, involves navigating several potential pitfalls. Overlooking these can lead to compromised patient safety, legal issues, and reputational damage. Below are key challenges related to quality and intellectual property (IP):
Poor Material Quality and Manufacturing Standards
Many low-cost suppliers use substandard stainless steel or inadequate heat treatment processes, resulting in instruments that corrode, deform, or lose sharpness quickly. Instruments made with inferior alloys may also trigger allergic reactions or fail sterilization protocols. Always verify compliance with international standards such as ISO 7405 (dentistry — evaluation of biocompatibility) and ISO 9001 (quality management systems). Request material certifications and conduct batch testing when scaling up procurement.
Lack of Precision in Design and Functionality
Tooth extraction instruments require precise geometries to match dental anatomy. Poorly manufactured tools may have misaligned tips, incorrect angulations, or inconsistent jaw profiles, increasing the risk of tooth fracture or tissue trauma. Ensure suppliers adhere to original equipment manufacturer (OEM) specifications and conduct functional testing before bulk orders. Engage dental professionals in the evaluation process.
Counterfeit or Imitation Brands Infringing IP Rights
A significant risk when sourcing from certain regions is the prevalence of counterfeit instruments mimicking well-known brands (e.g., Hu-Friedy, Aesculap, or W&H). These copies often infringe design patents and trademarks. Purchasing such products exposes buyers to legal liability, customs seizures, and damage to professional credibility. Conduct due diligence: verify brand authorization, request IP documentation, and work with reputable distributors rather than anonymous online marketplaces.
Inadequate Sterilization and Biocompatibility Documentation
Instruments must withstand repeated autoclaving without degradation. Suppliers may fail to provide evidence of biocompatibility or sterilization validation. Insist on documentation such as IFU (Instructions for Use), biocompatibility test reports (per ISO 10993), and proof of resistance to repeated sterilization cycles (e.g., 134°C, 20+ cycles).
Insufficient Quality Control and Traceability
Unreliable suppliers may lack proper quality control systems, leading to inconsistent instrument performance. Traceability—batch numbers, manufacturing dates, and material logs—is essential for recalls and audits. Ask for QC protocols, sample inspection reports, and whether instruments are individually serialized or batch-tracked.
Hidden Costs from High Failure and Replacement Rates
Though initial prices may be attractive, low-quality instruments often require frequent replacement due to breakage or dulling. This increases long-term costs and disrupts clinical workflow. Factor in total cost of ownership, including maintenance, replacement frequency, and potential clinical complications.
Non-Compliance with Regulatory Requirements
Instruments sold in regulated markets (e.g., U.S. FDA, EU MDR) must meet specific medical device classifications. Sourcing non-compliant tools can result in import bans or fines. Confirm that the supplier can provide CE marking, FDA registration, or other relevant certifications based on your target market.

Logistics & Compliance Guide for Tooth Extraction Instruments
Regulatory Classification and Standards
Tooth extraction instruments, such as forceps, elevators, and luxators, are classified as medical devices and are subject to stringent regulatory oversight. In the United States, the Food and Drug Administration (FDA) categorizes these instruments under Class I or Class II devices, typically requiring adherence to 21 CFR Part 872 (Dental Devices). In the European Union, they must comply with the Medical Device Regulation (MDR) (EU) 2017/745 and bear the CE marking. Manufacturers and distributors must ensure conformity with international standards such as ISO 13485 (Quality Management for Medical Devices) and ISO 7639 (Dental Instruments—General Requirements). Labeling must clearly indicate device classification, intended use, sterilization status, and manufacturer details.
Manufacturing and Quality Control
All tooth extraction instruments must be manufactured using biocompatible materials, typically high-grade stainless steel (e.g., AISI 304 or 316L), resistant to corrosion and suitable for repeated sterilization. Production facilities must operate under certified quality management systems (QMS), with documented procedures for design control, material sourcing, machining, finishing, and inspection. Instruments should undergo rigorous quality checks for dimensional accuracy, surface finish, mechanical strength, and absence of defects. Batch traceability must be maintained throughout the production cycle to support recall management and regulatory audits.
Sterilization and Packaging
Tooth extraction instruments are typically supplied non-sterile and intended for terminal sterilization by the end-user. However, if pre-sterilized, they must be packaged in validated sterile barrier systems compliant with ISO 11607. Packaging must protect against contamination, physical damage, and moisture ingress during transit and storage. Common sterilization methods include steam autoclaving (121°C or 134°C), ethylene oxide (EtO), or hydrogen peroxide plasma. Validated sterilization protocols must be documented, and packaging must clearly display sterilization method, expiry date (if applicable), and instructions for use.
Import and Export Compliance
International shipment of tooth extraction instruments requires compliance with export control regulations, customs documentation, and country-specific import requirements. Exporters must obtain necessary licenses (e.g., U.S. Department of Commerce or FDA export certificates) and ensure instruments meet the regulatory standards of the destination country. Harmonized System (HS) codes, such as 9018.49 (for dental instruments), must be accurately declared. Documentation should include commercial invoices, packing lists, certificates of origin, and regulatory conformity certificates (e.g., CE, FDA 510(k), or Free Sale Certificate). Restricted materials or dual-use technologies must be screened to avoid compliance violations.
Transportation and Storage
Instruments must be transported in secure, tamper-evident packaging to prevent damage and contamination. Temperature and humidity-controlled logistics are recommended, especially for sterile products. Storage facilities should maintain clean, dry environments with controlled access and proper inventory rotation (FIFO – First In, First Out). Shelf life, if applicable, must be monitored, and expired or compromised products must be quarantined and disposed of according to medical waste regulations.
Post-Market Surveillance and Vigilance Reporting
Manufacturers and authorized representatives are responsible for post-market surveillance (PMS) and reporting adverse events or device malfunctions. Under FDA’s Medical Device Reporting (MDR) regulation (21 CFR Part 803) and EU MDR Article 87, incidents such as instrument breakage, corrosion, or patient injury must be reported within specified timeframes. Periodic safety update reports (PSURs) may be required for higher-risk devices. A robust complaint handling system and field corrective action process (e.g., recalls or safety alerts) must be in place.
Labeling and Instructions for Use (IFU)
Each instrument must bear a unique device identifier (UDI) if required by jurisdiction (e.g., FDA UDI rule or EU MDR). Labels must include the device name, model/lot number, manufacturer information, material composition, and reprocessing instructions. The IFU must provide clear guidance on cleaning, disinfection, sterilization, handling, and contraindications. Multilingual IFUs may be necessary for international distribution. Electronic IFUs (eIFUs) are permitted under certain conditions but must remain accessible to users.
Environmental and Disposal Compliance
End-of-life instruments must be disposed of in accordance with local biomedical or hazardous waste regulations. Reusable instruments should be reprocessed according to validated protocols; single-use items must be discarded after one procedure. Manufacturers should consider environmental impact during design and packaging, promoting recyclable materials and minimizing waste. Compliance with directives such as the EU’s Waste Electrical and Electronic Equipment (WEEE) or Restriction of Hazardous Substances (RoHS) may apply depending on construction.
Training and Supply Chain Accountability
Distributors and healthcare providers must be trained on proper handling, storage, and reprocessing of tooth extraction instruments. Supply chain partners should be audited for regulatory compliance and quality assurance. Contracts with suppliers and logistics providers must include clauses ensuring adherence to medical device regulations, data integrity, and traceability requirements.
In conclusion, sourcing high-quality tooth extraction instruments is a critical step in ensuring the success, safety, and efficiency of dental procedures. It requires careful consideration of factors such as instrument material (typically surgical-grade stainless steel), precision in craftsmanship, ergonomics for practitioner comfort, and compliance with international safety and sterilization standards. Sourcing from reputable manufacturers or suppliers who provide authenticity, warranties, and adherence to regulatory requirements—such as FDA or CE certification—is essential. Additionally, building relationships with reliable vendors, evaluating cost-effectiveness without compromising quality, and staying updated on advancements in dental instrumentation contribute to long-term clinical success. Ultimately, the right sourcing strategy not only enhances treatment outcomes but also supports infection control, durability, and overall patient satisfaction in dental practice.