The global titanium mesh market is experiencing robust growth, driven by rising demand in aerospace, medical implants, and industrial filtration. According to Mordor Intelligence, the titanium market is projected to grow at a CAGR of over 6.5% from 2023 to 2028, with titanium mesh applications playing a key role in this expansion due to their high strength-to-weight ratio, corrosion resistance, and biocompatibility. Grand View Research further supports this trend, estimating that the global titanium market size was valued at USD 20.3 billion in 2022 and is expected to grow at a CAGR of 6.8% from 2023 to 2030. This growth is fueled by increased adoption in orthopedic and dental implant sectors, where titanium mesh is critical for bone regeneration, as well as in aerospace and defense for lightweight structural components. As demand escalates, a select group of manufacturers has emerged as leaders in innovation, quality, and global supply—shaping the future of advanced material solutions across industries.
Top 10 Titanium Mesh Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Titanium Mesh Manufacturer in China
Domain Est. 2016
Website: filsonfilters.com
Key Highlights: As a titanium wire mesh factory backed by more than two decades of experience, Filson always provides high-quality and cost-effective titanium wire mesh for ……
#2 Titanium Expanded Metal Manufacturer
Domain Est. 1997
Website: acsindustries.com
Key Highlights: Titanium expanded metal is a durable material created by slitting and stretching sheets of titanium, forming a strong, open mesh with diamond-shaped openings….
#3 Top Titanium Mesh Manufacturer in China
Domain Est. 2021
Website: heletitanium.com
Key Highlights: Hele Titanium is a premier global supplier of titanium mesh. Dive into our diverse offerings, from wire to expanded and perforated designs….
#4 Titanium mesh Manufacturer
Domain Est. 2021
Website: titanuim-anodes.com
Key Highlights: Titanium mesh is valued for its high strength, light weight and corrosion-resistant properties. It is used in a wide variety of Industries, Aerospace, Medical…
#5 Titanium Mesh
Website: chinatitaniumfactory.com
Key Highlights: Explore high-quality titanium mesh for medical, industrial, and aerospace applications. Discover woven, expanded, and surgical grades with superior ……
#6 pfm medical titanium
Domain Est. 2000
Website: pfmmedical.com
Key Highlights: pfm medical titanium gmbh, founded in Nuremberg in 2002, specializes in the development, manufacture and sale of mesh implants….
#7 Titanium Mesh
Domain Est. 2013
Website: samaterials.com
Key Highlights: Stanford Advanced Materials (SAM) has over two decades of experience in the manufacture and sale of titanium mesh, offering customers high-quality titanium ……
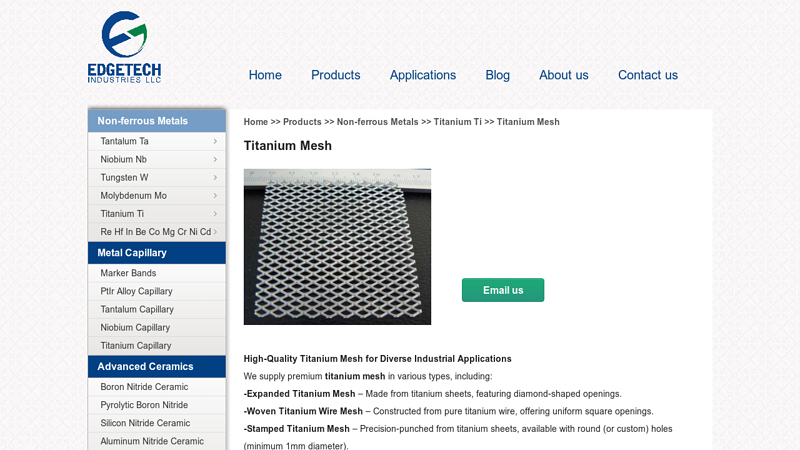
#8 Titanium Mesh
Domain Est. 2015
Website: edge-techind.com
Key Highlights: Our titanium mesh solutions are durable, corrosion-resistant, and tailored for filtration, electrolysis, architecture, and chemical processing….
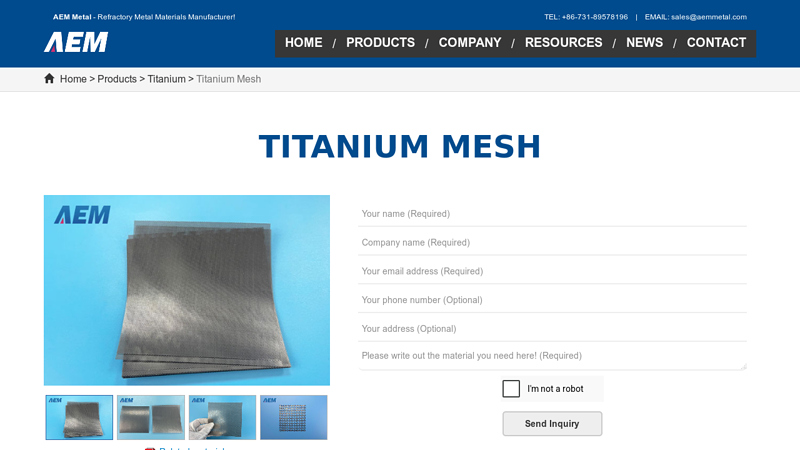
#9 Titanium Mesh
Domain Est. 2018
Website: aemmetal.com
Key Highlights: AEM Metal supplies all kinds of titanium mesh worldwide. Support bulk customization to your specifications at a reasonable price. Request A Quote….
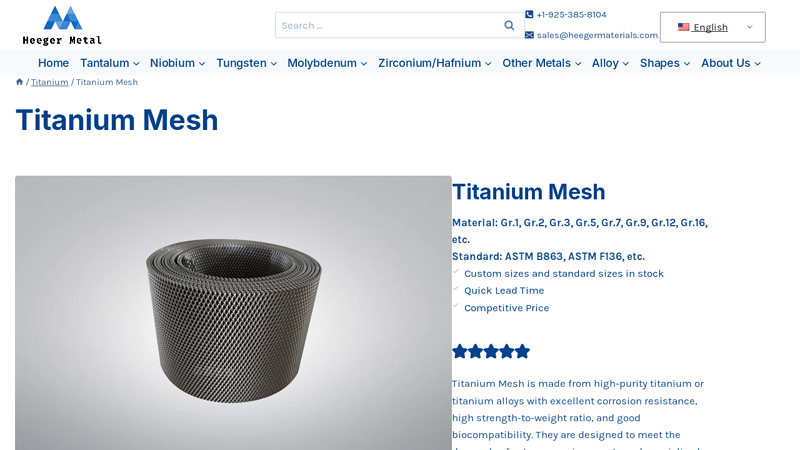
#10 Titanium Mesh
Domain Est. 2024
Website: heegermetal.com
Key Highlights: Titanium Mesh is made of titanium wire woven in different ways, including plain, twill, and Dutch weave. Customized wire diameters, aperture rates, and weaving ……
Expert Sourcing Insights for Titanium Mesh

H2: 2026 Market Trends for Titanium Mesh
The global titanium mesh market is poised for robust growth and significant transformation by 2026, driven by converging technological advancements, expanding applications, and evolving industry demands. Key trends shaping the landscape include:
1. Surge in Medical and Biomedical Applications:
* Orthopedic & Spinal Implants: Demand for titanium mesh in spinal fusion cages, bone grafts, and craniofacial reconstruction will accelerate due to aging populations, rising prevalence of degenerative spine diseases, and the material’s superior biocompatibility, osseointegration, and strength-to-density ratio. 3D-printed customized mesh implants will gain significant traction for complex anatomical requirements.
* Dental Implants & Prosthetics: Increasing adoption of dental implants globally, coupled with the need for immediate loading and guided bone regeneration (GBR), will drive demand for precisely engineered titanium mesh barriers and scaffolds.
* Advanced Wound Healing & Tissue Engineering: Research and development in using titanium mesh as scaffolds for soft tissue repair and regenerative medicine will progress, potentially opening new high-value market segments by 2026.
2. Dominance of Aerospace & Defense Adoption:
* Weight Reduction Imperative: The relentless drive for fuel efficiency and increased payload capacity in commercial and military aircraft will solidify titanium mesh’s role in lightweight, high-strength components like acoustic liners, engine nacelles, radomes, and structural panels.
* Hypersonics & Next-Gen Platforms: Development of hypersonic vehicles and advanced defense systems requiring materials that withstand extreme temperatures and stresses will create new, high-performance applications for specialized titanium mesh configurations.
* Supply Chain Resilience: Geopolitical factors and post-pandemic strategies will push aerospace OEMs towards securing reliable, diversified sources for critical materials like titanium mesh, potentially reshaping sourcing patterns.
3. Technological Advancements in Manufacturing:
* Additive Manufacturing (AM) Revolution: By 2026, AM (especially Electron Beam Melting – EBM and Laser Powder Bed Fusion – LPBF) will be a primary driver, enabling the production of complex, patient-specific medical implants and intricate aerospace components with optimized lattice structures impossible through traditional methods. This reduces waste and enables mass customization.
* Enhanced Process Control & Automation: Increased automation in weaving, sintering, and post-processing will improve consistency, reduce costs, and scale production capacity to meet rising demand across sectors.
* Advanced Coatings & Surface Modifications: Development of bioactive coatings (e.g., hydroxyapatite) for medical implants and specialized corrosion/erosion-resistant coatings for industrial applications will enhance functionality and expand market reach.
4. Industrial & Energy Sector Expansion:
* Filtration & Separation: Growing demand for efficient filtration in harsh environments (chemical processing, oil & gas, power generation) will boost use of titanium mesh filters and screens due to exceptional corrosion resistance and durability at high temperatures.
* Electrochemical Applications: The titanium mesh market for anodes in chlor-alkali production and electro-winning/refining processes will remain stable, with potential growth linked to green hydrogen production (electrolysis) and battery material processing, where titanium’s stability is crucial.
* Emerging Energy Technologies: Applications in fuel cells and next-generation nuclear reactors (as components or filters) will represent niche but potentially high-growth areas.
5. Sustainability and Cost Pressures:
* Recycling Focus: Increased emphasis on circular economy principles will drive investment in efficient titanium scrap recycling technologies, impacting raw material sourcing and potentially moderating long-term price volatility.
* Cost Optimization: High raw material and processing costs remain a challenge. Innovations in powder production (e.g., cheaper methods like AMA, HAMR), yield improvement in AM, and process optimization will be critical competitive factors.
* Supply Chain Localization: Efforts to reduce reliance on single-source suppliers (particularly for sponge) will lead to regional capacity expansion, especially in Asia-Pacific and North America, enhancing supply security but potentially increasing short-term costs.
6. Regional Dynamics:
* Asia-Pacific Leadership: China, Japan, and India will remain dominant markets and manufacturing hubs, driven by massive medical device production, aerospace ambitions (e.g., COMAC), and rapid industrialization.
* North American & European Innovation: These regions will lead in R&D, advanced manufacturing (especially AM for medical/aerospace), and high-value applications, focusing on premium, customized solutions.
* Emerging Markets: Growth in medical infrastructure and industrial development in regions like Latin America and the Middle East will create new demand pockets.
Conclusion for 2026:
By 2026, the titanium mesh market will be characterized by strong growth, technological sophistication, and application diversification. The convergence of additive manufacturing, particularly in medical and aerospace sectors, will be the most transformative trend. While cost and supply chain challenges persist, the unique properties of titanium ensure its irreplaceable role in critical, high-performance applications. Success will favor companies investing in AM capabilities, R&D for new functionalities, sustainable practices, and resilient, geographically diversified supply chains.

Common Pitfalls When Sourcing Titanium Mesh: Quality and Intellectual Property Risks
Sourcing titanium mesh—commonly used in aerospace, medical implants, filtration, and architectural applications—requires careful attention to technical specifications and legal compliance. Overlooking key factors can lead to compromised performance, project delays, or legal exposure. Below are the most common pitfalls related to quality and intellectual property (IP).
Quality-Related Pitfalls
1. Inconsistent Material Composition and Certification
One of the most frequent issues is receiving titanium mesh that does not meet the required alloy specifications (e.g., Grade 2, Grade 5/Ti-6Al-4V). Unverified suppliers may provide material without proper mill test reports (MTRs) or with falsified certifications, leading to substandard performance in critical applications.
Best Practice: Always demand full material traceability and certified test reports conforming to standards like ASTM B265 (for titanium plate, sheet, and strip) or ASTM F136 (for surgical implant applications).
2. Poor Mesh Dimensional Accuracy and Tolerance Control
Titanium mesh often requires precise aperture sizes, wire diameters, and tensile strength. Inaccurate weaving or inconsistent post-processing (e.g., annealing, cutting) can result in non-uniform mesh, affecting filtration efficiency or structural integrity.
Best Practice: Specify tight tolerances in your procurement documents and conduct incoming inspections using calibrated tools or third-party testing.
3. Inadequate Surface Finish and Contamination
Titanium is highly reactive during processing. Poor handling or improper cleaning can introduce surface contaminants (e.g., iron particles from shared equipment), leading to corrosion or biocompatibility issues in medical uses.
Best Practice: Ensure suppliers use dedicated, clean equipment and perform passivation or pickling as needed. Request certification of cleaning procedures.
4. Lack of Traceability and Batch Control
In regulated industries, full traceability from raw material to finished product is essential. Some suppliers fail to maintain lot tracking, making it impossible to investigate failures or comply with audits.
Best Practice: Require batch-specific documentation and ensure the supplier has a documented quality management system (e.g., ISO 9001 or ISO 13485 for medical devices).
Intellectual Property-Related Pitfalls
1. Use of Patented Mesh Designs Without Authorization
Certain titanium mesh configurations—especially those used in spinal implants or advanced filtration systems—are protected by patents. Sourcing from suppliers who replicate patented designs without licensing exposes buyers to infringement claims, even if unintentional.
Best Practice: Conduct due diligence on the design’s IP status. Request indemnification from suppliers and verify they have rights to manufacture and sell the mesh design.
2. Supplier Misrepresentation of Proprietary Technology
Some suppliers claim to use proprietary weaving or sintering techniques to justify premium pricing. However, these claims may lack validity or patent protection, leading to overpayment for unproven or generic technology.
Best Practice: Ask for patents, technical white papers, or third-party validations. Perform background checks on the supplier’s IP portfolio.
3. Grey Market or Diverted Products
Titanium mesh produced under exclusive agreements may appear on the open market through unauthorized channels. Purchasing such products—even unknowingly—can violate distribution agreements and expose the buyer to legal risk.
Best Practice: Source directly from authorized manufacturers or distributors and verify the supply chain path.
4. Inadequate Protection of Custom Designs
If you develop a custom mesh design for a specific application, failing to secure IP rights (e.g., through design patents or confidentiality agreements) may allow suppliers to replicate or resell the design to competitors.
Best Practice: Use robust NDAs and clearly define IP ownership in contracts. File for appropriate protections before disclosing detailed specifications.
By proactively addressing these quality and IP risks, organizations can ensure reliable performance, regulatory compliance, and legal safety when sourcing titanium mesh.

Logistics & Compliance Guide for Titanium Mesh
Titanium mesh, widely used in medical implants, aerospace components, and industrial applications, requires careful handling, documentation, and compliance with international regulations due to its high value, controlled nature, and potential dual-use status. This guide outlines key logistics and compliance considerations for the transportation, storage, and regulatory adherence related to titanium mesh products.
Regulatory Classification and Export Controls
Titanium mesh may be subject to export controls depending on its composition, intended use, and destination. Key regulatory frameworks include:
- ITAR (International Traffic in Arms Regulations): If the titanium mesh is manufactured for or used in defense-related applications (e.g., military implants or aerospace structures), it may be listed on the U.S. Munitions List (USML), requiring ITAR compliance.
- EAR (Export Administration Regulations): Most commercial titanium mesh falls under the Commerce Control List (CCL), typically under ECCN 1C004 (metal alloys in forms such as mesh, wire, or powder) or 9A012 (aerospace-grade materials). A license may be required for certain destinations.
- Dual-Use Considerations: Titanium mesh with high strength-to-density ratios or specific processing may be classified as dual-use, triggering additional scrutiny.
Always verify the correct ECCN or USML category and obtain necessary export licenses before shipment.
Packaging and Handling Requirements
Proper packaging ensures product integrity and regulatory compliance during transit:
- Use anti-static, corrosion-resistant packaging (e.g., vacuum-sealed or VCI-wrapped materials) to prevent oxidation or contamination.
- Label packages with product specifications, batch/lot numbers, and handling instructions (e.g., “Fragile,” “Keep Dry”).
- For medical-grade titanium mesh, maintain sterile packaging and comply with ISO 11607 standards for packaging of terminally sterilized medical devices.
- Include Material Safety Data Sheets (MSDS/SDS) if required, especially for industrial or powdered forms.
Transportation and Shipping
- Mode of Transport: Air, sea, or land—choose based on urgency, cost, and destination. Air freight is common for high-value medical implants.
- Customs Documentation: Prepare commercial invoices, packing lists, certificates of origin, and export declarations. Include detailed descriptions (e.g., “Titanium Alloy Mesh, ASTM F136, for surgical use”).
- Incoterms: Clearly define responsibilities using standard Incoterms (e.g., FOB, DDP) to avoid disputes.
- Cold Chain & Environmental Controls: If applicable, monitor temperature and humidity, especially for sterile medical products.
Import Compliance
- Verify import regulations in the destination country. Some countries require:
- Import licenses or permits
- Product registration (e.g., CE Marking in EU, FDA clearance in USA)
- Conformity assessments (e.g., ISO 13485 for medical devices)
- Pay applicable duties and taxes. Titanium products may be subject to anti-dumping or countervailing duties in certain regions.
Quality and Traceability
- Maintain full traceability from raw material sourcing to final product (mill test reports, heat numbers, inspection certificates).
- Comply with industry standards such as ASTM F136, ASTM F67, or ISO 5832 for medical-grade titanium.
- Implement a documented Quality Management System (QMS) compliant with ISO 9001 or ISO 13485.
Recordkeeping and Audit Preparedness
- Retain export and shipping records for a minimum of 5 years (as required by EAR).
- Store documentation electronically with secure backups.
- Conduct internal audits to ensure ongoing compliance with export controls and logistics procedures.
Special Considerations for Medical Titanium Mesh
- FDA Registration: U.S. importers must ensure the device is registered and listed with the FDA.
- CE Marking: Required for sale in the European Economic Area; involves conformity assessment under MDR (Medical Device Regulation).
- Sterility and Shelf Life: Monitor expiration dates and storage conditions to maintain product efficacy.
Adherence to this logistics and compliance guide ensures safe, legal, and efficient handling of titanium mesh across global supply chains. Always consult legal and regulatory experts to address jurisdiction-specific requirements.
In conclusion, sourcing titanium mesh requires a strategic approach that balances material quality, supplier reliability, cost-efficiency, and compliance with industry standards. Titanium mesh, valued for its exceptional strength-to-weight ratio, corrosion resistance, and biocompatibility, is critical in industries such as aerospace, medical, and industrial filtration. When selecting a supplier, it is essential to verify certifications (such as ISO, ASTM, or ASME), ensure consistent material traceability, and evaluate manufacturing capabilities to meet specific design requirements. Additionally, considering factors such as lead times, scalability, and logistical support will contribute to a resilient supply chain. By conducting thorough due diligence and building strong partnerships with reputable suppliers, organizations can secure high-quality titanium mesh that supports performance, safety, and long-term project success.