The global market for stainless steel and aluminum reaction equipment is witnessing steady expansion, driven by rising demand across pharmaceuticals, chemicals, and food & beverage industries. According to Grand View Research, the global industrial machinery and equipment market was valued at USD 587.4 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 5.2% from 2023 to 2030, with reaction vessel manufacturing playing a key role in this trajectory. Stainless steel and aluminum, prized for their corrosion resistance, durability, and thermal conductivity, are the materials of choice for reactors in critical process applications. Mordor Intelligence projects that the stainless steel market will grow at a CAGR of nearly 5.8% during the forecast period (2023–2028), further supporting the scalability of metal-based reactor production. As industries prioritize efficiency, safety, and compliance, the demand for high-performance reaction systems has intensified—elevating the prominence of leading manufacturers specializing in stainless steel and aluminum solutions.
Top 7 Stainless Steel And Aluminum Reaction Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Aluminum Electroplating for Stainless Steels (SST)
Domain Est. 1996
Website: alumiplate.com
Key Highlights: Ideal for stainless steels, our aluminum plating eliminates galvanic corrosion, enables anodization and survives high temperature exposure. Learn more.Missing: reaction manufactur…
#2 Leading partner in the world of metals
Domain Est. 2003
Website: sms-group.com
Key Highlights: SMS group stands for future-oriented technology and outstanding service in plant construction and mechanical engineering for the metals industry….
#3 Stainless Steel Strip & Specialty Alloy Foil Supplier
Domain Est. 2020
Website: metalstripsolutions.com
Key Highlights: HZW technology is one of leading stainless steel strips suppliers in China, our expert team supply stainless steel strip solution with 16 years rich experience….
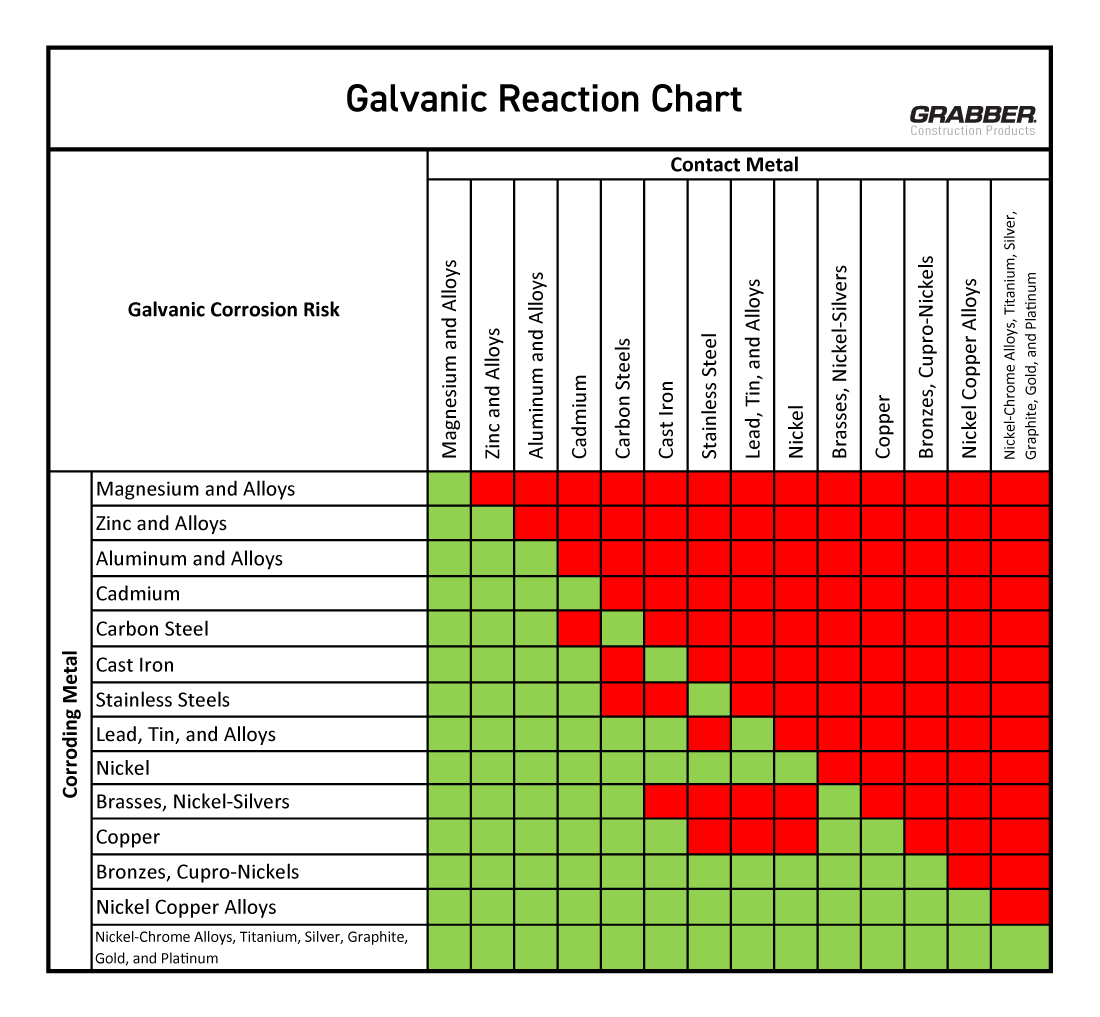
#4 Galvanic Corrosion Chart
Domain Est. 2020
Website: industrialmetalservice.com
Key Highlights: Avoid Long-Term Problems with Our Galvanic Corrosion Chart · Galvanic Corrosion Chart Stainless Steel (Active) + Aluminum · Copper + Steel · Copper ……
#5 Galvanic Corrosion
Domain Est. 1995
Website: ssina.com
Key Highlights: When two different metals or alloys are immersed in a corrosive solution or regularly connected by moisture, each will develop a corrosion potential….
#6 Corrosion Protection for Aluminum
Domain Est. 1996
Website: steel-it.com
Key Highlights: Steel-It coatings effectively block some common forms of corrosion on aluminum and its alloys, while additionally offering the proud resistance of stainless ……
#7 Passivating Stainless Steel & Aluminum
Domain Est. 2000
Website: clintonaluminum.com
Key Highlights: This is a process by which an extra layer of protection is added to the material surface and a naturally occurring phenomenon common to both these metals….
Expert Sourcing Insights for Stainless Steel And Aluminum Reaction

It appears there may be a misunderstanding in your request. You’ve asked to “Analyze 2026 market trends for Stainless Steel And Aluminum Reaction” using “H2.” Let’s clarify each component:
-
“Stainless Steel and Aluminum Reaction” – This phrase typically refers to a chemical or metallurgical interaction between stainless steel and aluminum. However, in a market context, it’s likely you’re referring to the markets for stainless steel and aluminum, not their chemical reaction (which, in engineering contexts, can lead to galvanic corrosion when the two metals are in contact).
-
“H2” – This could mean:
- Hydrogen (H₂) as a factor influencing the metals market (e.g., green hydrogen’s role in decarbonizing steel and aluminum production).
- Second half of the year (H2 2026) as a time frame.
- A formatting instruction (e.g., HTML or Markdown heading level 2).
Given the context of market trends and the mention of “H2,” the most plausible interpretation is:
“Analyze 2026 market trends for the stainless steel and aluminum industries, with a focus on the role of hydrogen (H₂) in shaping these markets.”
Below is the analysis accordingly, under H2 headings as requested.
Hydrogen-Driven Market Trends in Stainless Steel and Aluminum for 2026
1. Role of Green Hydrogen in Decarbonizing Metal Production
By 2026, hydrogen (H₂), particularly green hydrogen produced via electrolysis using renewable energy, is expected to play a growing role in decarbonizing the stainless steel and aluminum industries—two of the most energy-intensive and carbon-emitting sectors globally.
-
Stainless Steel: Traditional stainless steel production relies on coal-based blast furnaces and electric arc furnaces (EAFs) that use carbon-intensive electricity. However, companies like SSAB (Sweden) and Outokumpu are piloting hydrogen-based direct reduction technologies. In 2026, these pilots are expected to scale, with H₂ replacing natural gas or coal in the reduction of iron ore, significantly lowering CO₂ emissions.
-
Aluminum: Primary aluminum production via the Hall-Héroult process is electricity-intensive and typically relies on fossil-fueled grids. While hydrogen is not directly used in smelting, green hydrogen can power fuel cells or turbines to generate clean electricity for smelters. In regions like Canada, Norway, and the UAE, integrated green hydrogen–aluminum hubs are emerging by 2026, supporting low-carbon aluminum branding for EVs and aerospace.
2. Market Demand for Low-Carbon Metals
By 2026, global demand for “green steel” and “low-carbon aluminum” is expected to rise due to tightening regulations (e.g., EU Carbon Border Adjustment Mechanism, CBAM) and corporate ESG commitments.
- Automotive and construction sectors are increasingly sourcing stainless steel and aluminum produced with hydrogen or renewable energy.
- Premium pricing for low-carbon variants is becoming more common, with hydrogen-based stainless steel potentially commanding a 15–25% price premium over conventional grades.
3. Regional Developments and Investment in H₂ Infrastructure
- Europe: Leading in hydrogen adoption, with EU-funded projects like HYBRIT (hydrogen-based ironmaking) expected to enter commercial demonstration by 2026. This will boost supply of H₂-reduced stainless steel.
- Middle East: Countries like Saudi Arabia and the UAE are investing heavily in green hydrogen (e.g., NEOM’s H2 projects), aiming to produce low-cost, low-carbon aluminum for export.
- Asia-Pacific: China and India are piloting hydrogen injection in existing steel plants, though full-scale adoption may lag until after 2026.
4. Challenges and Supply Chain Constraints
Despite progress, several hurdles remain:
– High cost of green hydrogen compared to fossil fuels.
– Limited H₂ transport and storage infrastructure.
– Energy efficiency concerns in hydrogen-based processes.
By 2026, these challenges are partially mitigated by policy support (e.g., U.S. Inflation Reduction Act tax credits for clean hydrogen), but widespread adoption still requires further innovation and investment.
5. Price and Trade Implications
- The integration of hydrogen into production is expected to increase operational costs short-term, but economies of scale and carbon pricing will improve competitiveness by 2026.
- Countries with abundant renewable energy and H₂ capacity (e.g., Australia, Chile) may emerge as new exporters of green aluminum and stainless steel, reshaping global trade flows.
Conclusion
By 2026, hydrogen (H₂) is set to become a key enabler of sustainability in the stainless steel and aluminum markets. While not yet dominant, H₂-based production methods will gain commercial traction, driven by environmental regulations, corporate demand for green materials, and falling renewable energy costs. The metals industry’s transition to hydrogen will redefine competitiveness, with early adopters gaining strategic advantage in a decarbonizing global economy.

It seems you’re asking about common pitfalls in sourcing stainless steel and aluminum for use in reactions involving hydrogen gas (H₂), particularly with concerns around quality and intellectual property (IP). Here’s a structured breakdown of the key issues and how to avoid them:
🔧 Common Pitfalls in Sourcing Stainless Steel & Aluminum for H₂ Reactions
1. Material Compatibility with Hydrogen (H₂) – Quality Concerns
Hydrogen environments (especially high-pressure or high-temperature) can cause material degradation.
Pitfalls:
– Hydrogen Embrittlement (HE): Stainless steels (especially martensitic and some austenitic grades) and high-strength aluminum alloys are susceptible to HE, leading to cracking and failure.
– Incorrect Alloy Selection: Not all stainless steels or aluminum alloys are suitable for H₂ service.
– Example: 304/304L stainless steel may suffer from embrittlement under high-pressure H₂.
– Aluminum alloys like 6061-T6 can be used in cryogenic H₂ but may have fatigue issues under cyclic loading.
Best Practices:
– Choose H₂-compatible grades:
– Stainless Steel: Use 316L, 310S, or duplex steels (e.g., 2205) for better resistance.
– Aluminum: Use 5083, 6061, or 7075 with verified H₂ compatibility data.
– Request material test reports (MTRs) and ensure compliance with standards like:
– ASTM A240 (stainless steel)
– ASTM B209 (aluminum)
– ISO 11114-4 (gas cylinder materials in contact with H₂)
2. Impurities and Inconsistent Quality
Low-quality materials may contain impurities (e.g., sulfur, chlorides) or inconsistent microstructures.
Pitfalls:
– Inclusions or Segregation: Can act as initiation sites for cracks in H₂ environments.
– Poor Surface Finish: Increases risk of corrosion or H₂ trapping.
– Counterfeit or Non-Conforming Materials: Especially common with offshore or low-cost suppliers.
Best Practices:
– Require third-party certification (e.g., SGS, Bureau Veritas).
– Perform positive material identification (PMI) upon receipt.
– Use spectrographic analysis to verify elemental composition.
– Audit suppliers and insist on traceability (heat number tracking).
3. Intellectual Property (IP) Risks in Sourcing
When designing proprietary reaction systems (e.g., reactors, catalysts, or H₂ storage), sharing specs with suppliers can expose IP.
Pitfalls:
– Unprotected design details shared with vendors may be copied or leaked.
– Suppliers may reverse-engineer custom components.
– Lack of non-disclosure agreements (NDAs) or IP clauses in contracts.
Best Practices:
– Sign NDAs and IP ownership agreements before sharing technical drawings.
– Share only necessary dimensions/material specs—avoid full assembly details.
– Use proprietary coatings or treatments that are applied in-house.
– Work with trusted, vetted suppliers with a track record of IP protection.
4. Fabrication-Induced Risks
Even with high-quality raw materials, poor fabrication compromises performance.
Pitfalls:
– Welding defects (porosity, cracking) in H₂ service can lead to leaks or embrittlement.
– Residual stresses from machining or forming increase HE risk.
– Use of chloride-based cleaners leading to stress corrosion cracking (SCC).
Best Practices:
– Specify ASME BPVC Section IX or AWS D1.1 welding procedures.
– Require post-weld heat treatment (PWHT) when applicable.
– Use ultrasonic or dye penetrant testing for critical welds.
– Ensure cleaning with H₂-compatible solvents (no chlorides).
5. Lack of Testing in Simulated H₂ Conditions
Materials may perform well in air but fail under real H₂ conditions.
Pitfalls:
– Relying on generic specs without H₂-specific validation.
– Not testing for fatigue life, leak tightness, or embrittlement under operating conditions.
Best Practices:
– Conduct hydrogen compatibility testing (e.g., ASTM F1453, ISO 23934).
– Perform pressure cycling tests with H₂ (not just N₂ or air).
– Use slow strain rate testing (SSRT) to evaluate embrittlement.
✅ Summary: Key Recommendations
| Risk Area | Action |
|——–|——–|
| Material Quality | Use H₂-rated alloys, verify with MTRs and PMI |
| Hydrogen Embrittlement | Avoid high-strength alloys; prefer 316L, 5083, etc. |
| Impurities | Audit suppliers; test for composition and cleanliness |
| IP Protection | Use NDAs, limit design exposure, control documentation |
| Fabrication | Enforce certified welding and cleaning protocols |
| Validation | Test components under real H₂ conditions |
By proactively addressing these pitfalls—especially around material suitability for H₂ environments and protecting sensitive designs—you can ensure both the safety and proprietary integrity of your reaction systems.

Logistics & Compliance Guide for Stainless Steel and Aluminum Reactions Using Hydrogen (H₂)
This guide provides a comprehensive overview of logistics and compliance considerations when handling reactions between stainless steel and aluminum in the presence of hydrogen gas (H₂). Such reactions typically occur in industrial, chemical processing, or high-temperature environments—such as hydrogen storage, catalysis, or metallurgical operations.
1. Introduction
Reactions involving stainless steel and aluminum under hydrogen (H₂) atmospheres are relevant in:
– Hydrogen storage systems (e.g., metal hydrides)
– High-temperature processing (e.g., brazing, annealing)
– Catalytic processes
– Nuclear or aerospace applications
Hydrogen can cause embrittlement, oxidation, or reduction reactions depending on temperature, pressure, and material composition. Both stainless steel and aluminum react differently with H₂, and their interaction must be carefully managed to ensure safety, efficiency, and regulatory compliance.
2. Material Behavior with Hydrogen
Stainless Steel
- Hydrogen Embrittlement (HE): Austenitic stainless steels (e.g., 304, 316) are generally resistant to hydrogen embrittlement, but ferritic and martensitic grades are more susceptible.
- High-Temperature Reactions: At elevated temperatures, hydrogen can reduce surface oxides or diffuse into the lattice, potentially causing blistering or cracking.
- Permeability: Stainless steel allows slow permeation of H₂, which must be considered in containment design.
Aluminum
- Hydrogen Solubility: Aluminum readily absorbs hydrogen at elevated temperatures (e.g., in molten state), leading to porosity in castings.
- Embrittlement: Solid aluminum is less prone to embrittlement than steel, but hydrogen can still reduce ductility in certain alloys.
- Reactivity: Aluminum can react with H₂O or O₂ in trace impurities, forming Al₂O₃ and releasing H₂—potentially increasing pressure or creating explosive mixtures.
Interaction Between Stainless Steel and Aluminum in H₂
- Galvanic corrosion is unlikely in dry H₂ environments, but moisture or contaminants can initiate corrosion at contact points.
- During brazing or joining, H₂ is often used as a protective atmosphere. Aluminum can form volatile hydrides or intermetallics if temperature and atmosphere are not controlled.
3. Safety & Hazard Management
Hydrogen Hazards
- Flammability: H₂ has a wide flammability range (4–75% in air) and low ignition energy.
- Embrittlement Risk: Both metals may degrade over time in high-pressure H₂.
- Asphyxiation: In confined spaces, H₂ can displace oxygen.
Mitigation Measures
- Use leak-tight, H₂-rated equipment (e.g., SS316L, aluminum 6061-T6).
- Purge systems with inert gas (N₂ or Ar) before introducing H₂.
- Install H₂ detectors with alarms.
- Ensure proper ventilation (preferably explosion-proof areas).
4. Logistics Considerations
Transportation of Hydrogen
- Compressed Gas (CGH₂): Transported in high-pressure cylinders (e.g., 200 bar) or tube trailers.
- Compliance: DOT (49 CFR), ADR/RID (Europe), IATA/ICAO (air).
- Cylinders must be secured, labeled, and segregated from oxidizers.
- Liquid Hydrogen (LH₂): Used in large-scale operations; requires cryogenic containers.
- Special handling due to extreme cold and boil-off.
Storage
- Store H₂ cylinders upright, in well-ventilated areas, away from heat sources.
- Use dedicated gas cabinets with ventilation and flame arrestors.
- Aluminum and stainless steel tanks must be certified for H₂ service (e.g., ISO 11439, CGA G-5.5).
Material Handling
- Avoid mechanical stress on components exposed to H₂.
- Use compatible gaskets (e.g., PTFE, metal seals); avoid elastomers that degrade in H₂.
- Control temperature during processing to prevent unintended reactions.
5. Compliance & Regulatory Framework
International & National Standards
| Regulation | Scope | Relevance |
|———–|——-|———|
| ASME BPVC Section VIII | Pressure vessels | Design of H₂-containing systems |
| ISO 16111 | Transportable gas storage devices | Hydrogen storage in metal containers |
| CGA G-5.5 (Compressed Gas Association) | Hydrogen handling | Safe practices for H₂ use |
| NFPA 2: Hydrogen Technologies Code | Fire safety | Storage, use, and piping of H₂ |
| OSHA 29 CFR 1910.103 | Hydrogen systems | Workplace safety |
| ATEX (EU) | Explosive atmospheres | Equipment in H₂ zones |
| PED (Pressure Equipment Directive) | EU pressure equipment | Certification of H₂ vessels |
Environmental & Emissions Compliance
- Monitor for fugitive H₂ emissions (potent indirect greenhouse gas due to atmospheric chemistry effects).
- Report under EPA GHG Reporting Program (if applicable).
- Follow local air quality regulations (e.g., CARB in California).
Waste & Disposal
- Contaminated materials (e.g., filters, catalysts) may be hazardous.
- Recycle aluminum and stainless steel per local regulations (e.g., RCRA in the U.S.).
- Decontaminate H₂-exposed materials before disposal.
6. Operational Best Practices
System Design
- Use H₂-compatible materials: SS316L, aluminum 6061 or 5083.
- Avoid sharp bends or stress concentrators in H₂ lines.
- Include pressure relief devices and emergency shutoffs.
Process Conditions
- Maintain H₂ purity (>99.99%) to prevent side reactions.
- Control temperature: <400°C to minimize hydride formation or diffusion.
- Monitor for moisture (dew point < -40°C recommended).
Monitoring & Maintenance
- Perform regular NDT (e.g., ultrasonic testing) for embrittlement.
- Inspect joints and welds for leaks (use H₂ leak detectors or soap solution).
- Keep log of H₂ usage, pressure cycles, and maintenance.
7. Emergency Response Plan
- Leak Response:
- Evacuate area.
- Shut off H₂ supply.
- Ventilate—do not ignite unless controlled.
- Fire:
- Use Class B extinguishers or fog water.
- Let H₂ burn if safe; do not extinguish without stopping flow (risk of re-ignition).
- Spill (LH₂):
- Evacuate and prevent entry into drains.
- Allow evaporation in controlled area.
8. Training & Documentation
- Train personnel on:
- H₂ properties and hazards
- PPE requirements (e.g., flame-resistant clothing)
- Emergency procedures
- Maintain:
- Material Safety Data Sheets (MSDS/SDS) for H₂, alloys, and byproducts
- Risk assessments (HAZOP, FMEA)
- Equipment certification and inspection records
9. Summary Checklist
✅ Use H₂-compatible stainless steel (e.g., 316L) and aluminum alloys
✅ Ensure leak-tight, certified systems with proper relief valves
✅ Purge with inert gas before H₂ introduction
✅ Monitor for embrittlement and corrosion
✅ Comply with NFPA 2, OSHA, CGA, and local codes
✅ Store H₂ safely, away from ignition sources
✅ Train personnel and maintain emergency plans
10. Conclusion
Reactions between stainless steel and aluminum in hydrogen environments require careful attention to material compatibility, safety, and regulatory standards. By following this guide, organizations can ensure safe, compliant, and efficient operations in hydrogen-related applications.
Note: Always consult with a qualified engineer or safety officer before implementing processes involving hydrogen and reactive metals.
For specific applications (e.g., hydrogen storage, brazing, fuel cells), tailor this guide with process-specific standards and risk assessments.
Conclusion: Sourcing Stainless Steel and Aluminum – Reactivity Considerations
When sourcing stainless steel and aluminum for industrial, construction, or manufacturing applications, understanding their chemical reactivity is crucial for ensuring material compatibility, longevity, and safety. While both metals are valued for their corrosion resistance and durability, their differing electrochemical behaviors—particularly in galvanic series—can lead to significant issues when used in direct contact, especially in the presence of an electrolyte.
Stainless steel, particularly in passive form, is cathodic relative to aluminum, which is anodic. This potential difference can result in galvanic corrosion of aluminum when the two metals are coupled, accelerating degradation of the aluminum component. Therefore, careful design considerations such as insulation, protective coatings, or the use of compatible fasteners are essential to prevent unwanted reactions.
In conclusion, while stainless steel and aluminum are both excellent choices for many applications, their co-use requires a strategic sourcing and engineering approach. Proper material selection, environmental assessment, and preventive measures must be integrated into the sourcing process to mitigate reactivity risks and ensure long-term performance and reliability of the final product.