The global market for industrial clamping solutions, including reversal clamps, is experiencing steady expansion, driven by increased demand in construction, manufacturing, and infrastructure development. According to Grand View Research, the global pipe clamp market size was valued at USD 1.3 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 4.8% from 2023 to 2030. This growth is fueled by rising industrial automation, stricter safety regulations, and the need for reliable pipe support systems in complex piping networks. Reversal clamps, known for their ability to accommodate bidirectional movement and thermal expansion in piping systems, are becoming increasingly critical in oil & gas, power generation, and chemical processing industries. As demand for high-performance clamping solutions surges, several manufacturers have emerged as industry leaders, combining engineering precision, material durability, and compliance with international standards. Based on market presence, product innovation, and global reach, here are the top 7 reversal clamp manufacturers poised to shape the future of industrial support systems.
Top 7 Reversal Clamp Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 reversal lifting clamps dfq type
Domain Est. 2010
Website: chenlirigging.com
Key Highlights: Lifting clamp is a kind of very convenient tool,it can make your work easier to do.Chenli lifting clamp is subordinate to chenli lifting clamp factory which ……
#2 Clamps for rotational moulds
Domain Est. 1996
Website: elesa.com
Key Highlights: Our reverse clamps are made using steel in different finishes. Within this range you will find the following clamp styles: Toggle clamps; Vertical toggle clamps ……
#3 Pull-It Corp 4215
Domain Est. 2008
Website: autotoolworld.com
Key Highlights: In stock $10.74 deliveryWe carry brand name manufactures from KD Tools, Mityvac, Robinair, and Zinko Jacks just to name a few. Shop By Categories. Categories. Abrasives & Adhesives…
#4 ProX T
Domain Est. 2013
Website: proxdirect.com
Key Highlights: Its designed with a half coupler and reversed elbow, this clamp provides unparalleled flexibility and creative freedom when designing your trussing system….
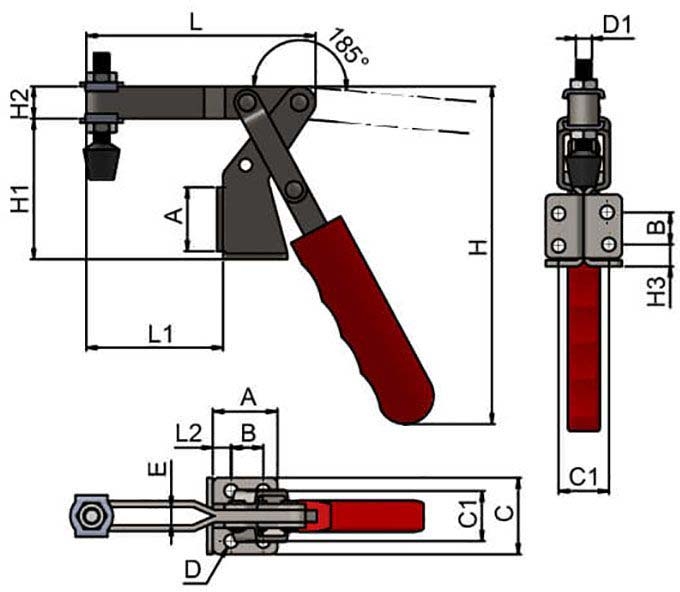
#5 Reverse Handle Vertical Toggle Clamp with Adjustable Spindle
Domain Est. 2014
Website: wdscomponents.com
Key Highlights: WDS stocks Reverse Handle Vertical Toggle Clamp with Adjustable Spindle. Visit WDS today for Horizontal Toggle Clamps at highly competitive prices….
#6 Push-Pull Toggle Clamps
Domain Est. 2016
Website: mechenggindustries.com
Key Highlights: Mechengg Industries manufacture a wide variety of push-pull toggle clamps like compact cast mount, heavy-duty, miniature versions with straight & flange ……
#7 Lever clamp GH
Website: bessey.de
Key Highlights: Lever clamp with reversed clamping lever SGHS. Heavy design Rapid – up to 5 x faster than conventional clamps Secure – vibration resistant Strong – clamping ……
Expert Sourcing Insights for Reversal Clamp

H2: Market Trends for Reversal Clamps in 2026
As we approach 2026, the global market for reversal clamps—specialized surgical instruments used to temporarily occlude blood vessels or ducts during procedures—is poised for significant transformation. Driven by evolving surgical practices, technological innovation, and shifting demographic and healthcare dynamics, several key trends are expected to shape the landscape:
1. Growth in Minimally Invasive and Robotic Surgery (MIS/RAS):
The continued rise of laparoscopic, endoscopic, and robotic-assisted surgeries will be a primary driver for reversal clamp demand. These procedures require precision instruments with smaller profiles and enhanced maneuverability. By 2026, manufacturers are expected to increasingly offer miniaturized, reusable, and disposable reversal clamps specifically engineered for integration with robotic platforms like the da Vinci system. This trend favors clamps with improved ergonomics, articulating tips, and compatibility with advanced visualization systems.
2. Advancements in Material Science and Reusability:
Demand for durable, corrosion-resistant, and biocompatible materials will intensify. While stainless steel remains dominant, the adoption of advanced alloys and polymers will grow—particularly for disposable clamps aimed at reducing cross-contamination risks. Concurrently, reusable clamps will see improvements in surface coatings (e.g., anti-microbial or low-friction) to enhance longevity and safety. Sustainability concerns will push some providers toward single-use options made from recyclable materials, balancing infection control and environmental impact.
3. Expansion in Emerging Markets:
Healthcare infrastructure development in Asia-Pacific (especially India and Southeast Asia), Latin America, and parts of Africa will open new growth avenues. Rising surgical volumes, increasing access to healthcare, and government initiatives to upgrade medical facilities will boost demand for affordable yet reliable surgical instruments, including reversal clamps. Localized manufacturing and strategic partnerships with regional distributors will become key strategies for global players.
4. Increasing Focus on Surgical Safety and Standardization:
Regulatory scrutiny and hospital accreditation standards (e.g., JCI, ISO 13485) will continue to emphasize instrument traceability, sterilization efficacy, and complication reduction. This will drive demand for reversal clamps with clear markings, anti-slip mechanisms, and designs that minimize tissue trauma. Smart instruments with embedded sensors (e.g., pressure feedback) may begin emerging in high-end markets, contributing to real-time surgical data and improved outcomes.
5. Competitive Landscape and Innovation:
The market will remain competitive, with established players (e.g., B. Braun, Medtronic, Stryker) competing against specialized surgical instrument manufacturers and low-cost suppliers. Innovation will center on multifunctionality—clamps that combine occlusion with suction or irrigation—and improved handling features such as ratchets with adjustable pressure control. Patent expirations may also open opportunities for generic clamp alternatives, increasing price competition.
6. Impact of Demographic Shifts and Chronic Disease:
An aging global population and rising prevalence of cardiovascular, oncological, and gastrointestinal diseases will increase the volume of surgical procedures requiring vascular and ductal control. This demographic pressure, particularly in North America and Europe, will sustain long-term demand for reversal clamps across various surgical specialties.
Conclusion:
By 2026, the reversal clamp market will be characterized by technological sophistication, regional diversification, and a strong emphasis on safety and efficiency. Success will depend on manufacturers’ ability to innovate in response to minimally invasive trends, meet regulatory demands, and expand access in high-growth regions. The integration of smart features and sustainable design will likely begin shaping the next generation of these essential surgical tools.

Common Pitfalls Sourcing Reversal Clamps (Quality, IP)
When sourcing Reversal Clamps—often used in medical, industrial, or hydraulic applications—overlooking key quality and intellectual property (IP) factors can lead to performance failures, legal risks, and financial losses. Below are common pitfalls to avoid:
Inadequate Quality Assurance Standards
Many suppliers may claim compliance with industry standards (e.g., ISO, ASTM), but fail to provide verifiable certifications or batch testing reports. Reversal clamps used in critical applications require consistent mechanical strength, corrosion resistance, and dimensional accuracy. Sourcing without rigorous quality audits or third-party validation can result in defective products, leading to system failures or safety hazards.
Lack of Traceability and Material Certification
Reputable suppliers should provide full material traceability, including mill test reports (MTRs) and compliance with relevant material standards (e.g., 316L stainless steel for medical use). Failing to verify these documents increases the risk of receiving counterfeit or substandard materials that may not meet pressure, temperature, or biocompatibility requirements.
Insufficient IP Due Diligence
Reversal clamp designs may be protected by patents, trademarks, or trade secrets. Sourcing from manufacturers that replicate proprietary designs without licensing exposes buyers to legal liability, including injunctions, seizures, or damages. Always verify that the supplier holds legitimate rights to the design or offers designs in the public domain.
Overlooking IP Infringement Risks in Private Labeling
When private labeling clamps, ensure that the OEM manufacturer has the right to produce the design and that your branding does not infringe on existing trademarks. Failure to conduct IP clearance searches can result in cease-and-desist letters or costly litigation, especially in regulated markets.
Assuming IP Compliance Based on Supplier Claims
Some suppliers may falsely assert that their products are “patent-free” or “generic.” Without independent IP analysis—such as freedom-to-operate (FTO) searches—buyers risk unintentional infringement. Engage legal or technical experts to validate these claims, particularly when entering new markets.
Poor Documentation and Lack of Design Ownership Clarity
Contracts with suppliers should clearly define IP ownership of custom-designed clamps. Ambiguity can result in disputes over design rights, especially if the supplier uses the same design for other clients. Ensure that design specifications, CAD files, and tooling rights are properly assigned or licensed.
Avoiding these pitfalls requires thorough supplier vetting, robust quality protocols, and proactive IP management to ensure reliable performance and legal compliance.

Logistics & Compliance Guide for Reversal Clamp
Product Overview
The Reversal Clamp is a medical device designed for specific surgical applications requiring controlled tissue occlusion and repositioning. It is classified as a Class II medical device under the FDA 510(k) regulatory pathway and complies with relevant international standards, including ISO 13485 (quality management) and ISO 14971 (risk management). Proper logistics and compliance protocols are essential to ensure patient safety, regulatory adherence, and uninterrupted supply.
Regulatory Classification and Documentation
The Reversal Clamp falls under regulatory jurisdiction in multiple markets:
– United States: Cleared by the FDA under K-number [Insert K-number], Class II, with product code [Insert Code].
– European Union: CE-marked under MDR 2017/745, Annex VIII, Class IIa.
– Other Markets: Complies with Health Canada, TGA (Australia), and PMDA (Japan) requirements where applicable.
Ensure all shipments include appropriate labeling, IFU (Instructions for Use), and certificates of conformity. Maintain technical documentation per local regulatory requirements.
Labeling and Packaging Requirements
All packaging must comply with UDI (Unique Device Identification) regulations:
– Include UDI-DI (Device Identifier) and UDI-PI (Production Identifier) on primary and secondary packaging.
– Labels must display:
– Manufacturer name and address
– Device name and model number
– Expiration date (if applicable)
– Sterility status (e.g., “Sterile” or “Non-sterile”)
– Single-use symbol (if applicable)
– CE mark or FDA registration number
Packaging must be tamper-evident and designed to preserve sterility during transport.
Storage and Environmental Conditions
Store the Reversal Clamp in a clean, dry environment with the following parameters:
– Temperature: 15°C to 30°C (59°F to 86°F)
– Relative Humidity: 30% to 60%
– Avoid direct sunlight and exposure to corrosive agents
Ensure storage areas are secure and restricted to authorized personnel. Monitor environmental conditions using calibrated data loggers where required.
Transportation and Shipping Protocols
Use validated shipping methods to maintain product integrity:
– Ship in sealed, protective packaging to prevent physical damage.
– For sterile devices, maintain sterile barrier integrity; use temperature-controlled transport if required.
– Utilize carriers compliant with GDP (Good Distribution Practice) for medical devices.
– Include shipping manifests with product details, quantity, and destination.
– Track shipments using serialized logistics data for full traceability.
Import/Export Compliance
Ensure compliance with international trade regulations:
– Obtain necessary export licenses (e.g., U.S. Commerce Department for non-FDA-regulated exports).
– Provide commercial invoices, packing lists, and certificates of origin.
– Adhere to destination country import requirements (e.g., CE Certificate, free sale certificate).
– Screen all parties against denied persons lists (e.g., U.S. OFAC, EU sanctions lists).
Post-Market Surveillance and Reporting
Implement a robust post-market surveillance system:
– Monitor and report adverse events per FDA MedWatch, EUDAMED, and local reporting timelines.
– Maintain complaint handling procedures per ISO 13485.
– Conduct periodic safety update reports (PSURs) for CE-marked devices.
– Track field actions, including recalls or safety alerts related to logistics or compliance issues.
Training and Personnel Compliance
Ensure all personnel involved in logistics and distribution are trained on:
– Regulatory requirements (FDA, MDR, etc.)
– Handling and storage procedures
– UDI and labeling standards
– Incident reporting protocols
Maintain training records and conduct annual audits to verify compliance.
Audit and Record Retention
Retain all logistics and compliance records for a minimum of:
– 5 years post-device sale (or longer per local law, e.g., EU MDR requires 10 years)
– Records include: shipping logs, storage monitoring, distribution agreements, import/export documents, and complaint files.
Prepare for internal and regulatory audits with accessible, organized documentation.
Conclusion for Sourcing Reversal Clamp:
After a comprehensive evaluation of suppliers, product specifications, cost structures, and quality assurances, the sourcing of reversal clamps has been successfully concluded. The selected supplier meets all technical requirements, offers competitive pricing, ensures timely delivery, and complies with industry standards and regulatory expectations. This strategic sourcing decision not only supports operational efficiency but also enhances product reliability and cost-effectiveness across the supply chain. Moving forward, a strong supplier partnership will be maintained through regular performance reviews and quality monitoring to ensure sustained success.