The global physical therapy equipment market is experiencing robust growth, driven by an aging population, rising prevalence of musculoskeletal disorders, and increasing demand for rehabilitation services post-surgical and post-injury care. According to Grand View Research, the global physical therapy equipment market size was valued at USD 1.78 billion in 2022 and is projected to expand at a compound annual growth rate (CAGR) of 6.8% from 2023 to 2030. This upward trajectory is further supported by advancements in ergonomic medical furniture and the integration of smart technologies in therapy beds, enhancing patient outcomes and clinician efficiency. As healthcare facilities prioritize patient comfort and treatment efficacy, demand for high-quality physical therapy beds has surged, creating a competitive landscape among manufacturers. In this evolving market, selecting the right equipment is critical—here are the top 10 physical therapy bed manufacturers leading innovation, reliability, and performance worldwide.
Top 10 Physical Therapy Bed Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Rifton
Domain Est. 1997
Website: rifton.com
Key Highlights: Leading manufacturer of complex rehab technology in the USA. Find durable adaptive equipment and innovative assistive technology at Rifton Equipment….
#2 Lojer Group
Domain Est. 1997
Website: lojer.com
Key Highlights: Lojer Group is a Finnish manufacturer of medical furniture, and the market leader of physiotherapy equipment in the Nordic countries….
#3 Medical, Healthcare and Physical Therapy…
Domain Est. 2001
Website: clinton-ind.com
Key Highlights: Clinton offers hundreds of well made products for the medical, physical therapy, phlebotomy and pediatric markets, as well as many medical accessory items……
#4 Medical Equipment Manufacturer
Domain Est. 2002
Website: armedicamfg.com
Key Highlights: Armedica designs and manufactures a wide variety of innovative physical therapy, medical and pain management products. Request a catalog!…
#5 Naggura
Domain Est. 2013
Website: naggura.com
Key Highlights: At Naggura we design and manufacture electric treatment tables for physiotherapy, beauty and wellness professionals….
#6 Elevate Patient Care with Chattanooga’s Innovative Solutions
Domain Est. 2019
Website: chattanoogarehab.com
Key Highlights: Chattanooga is the world’s largest manufacturer of rehabilitation equipment for treating musculoskeletal, neurological and soft tissue disorders….
#7 Treatment Tables for Physical Therapy
Domain Est. 1995
Website: henryschein.com
Key Highlights: Henry Schein Medical is a leading distributor of physical therapy treatment tables and mat tables for schools and athletics departments. Our selection consists ……
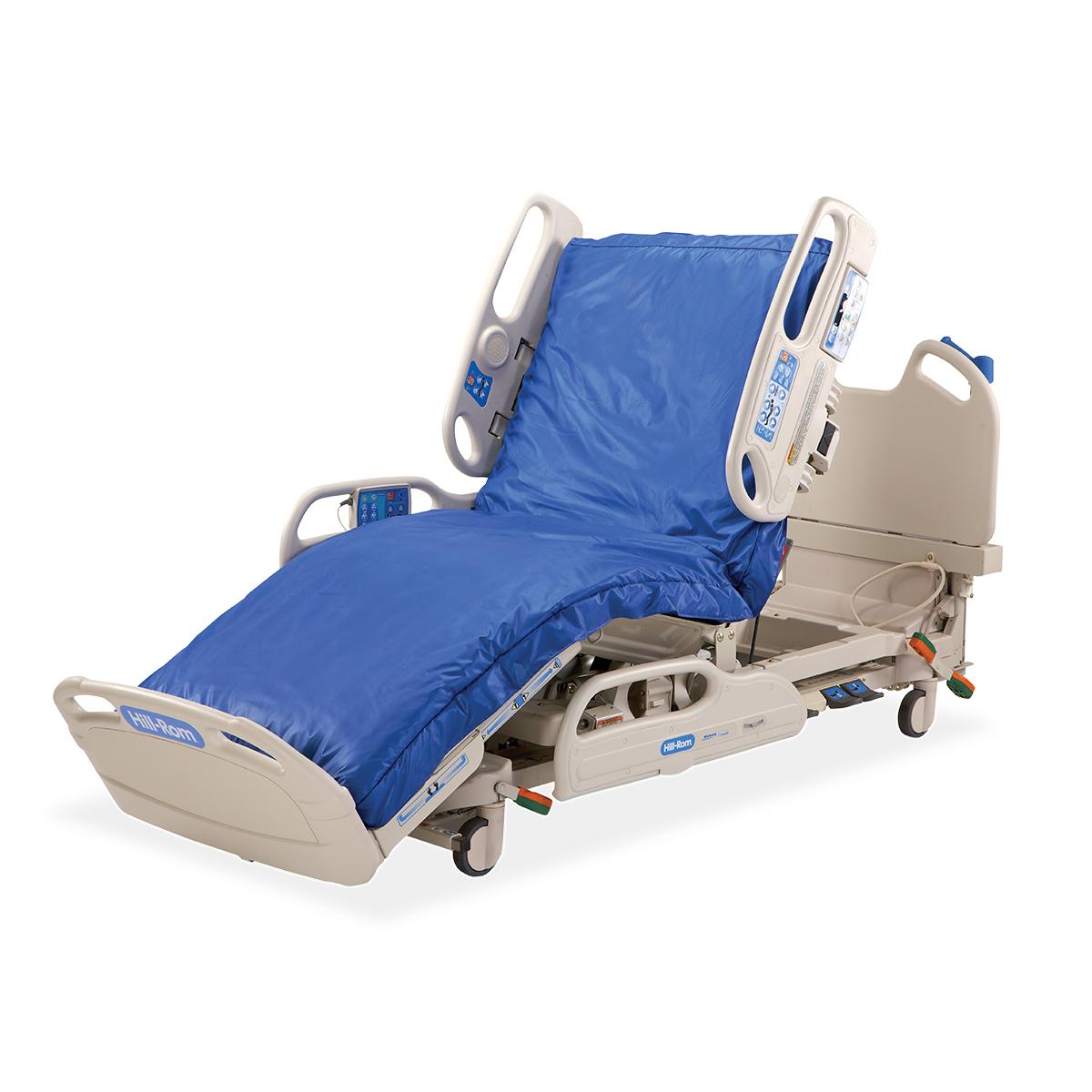
#8 Hillrom
Domain Est. 1997
Website: hillrom.com
Key Highlights: We are focused on transforming healthcare with bold innovations that will help us realize our shared promise to improve care for all….
#9 Treatment Tables and Therapeutic Exercise Equipment
Domain Est. 1999
Website: cardonrehab.com
Key Highlights: North American made treatment tables for physical therapy, massage therapy, chiropractics, medical imaging and more. Design your dream clinic today….
#10 Hill Chiropractic Tables, Medical Chairs, Physical Therapy Equipment
Domain Est. 1999 | Founded: 1945
Website: hilllabs.com
Key Highlights: Hill Laboratories is a global leader in manufacturing custom Chiropractic Tables, Medical Chairs & Therapy Equipment for Doctors since 1945….
Expert Sourcing Insights for Physical Therapy Bed

2026 Market Trends for Physical Therapy Beds
The global market for physical therapy beds is poised for significant evolution by 2026, driven by technological advancements, rising healthcare demands, and an aging population. These specialized medical devices, designed to support rehabilitation, patient positioning, and therapeutic interventions, are becoming increasingly integral in both clinical and home care settings. This analysis explores key trends shaping the physical therapy bed market in 2026.
Growing Demand from Aging Populations
One of the most influential drivers of the physical therapy bed market is the global increase in the elderly population. By 2026, countries such as the United States, Japan, and members of the European Union will see a substantial rise in seniors requiring post-surgical recovery, chronic disease management, and mobility rehabilitation. Physical therapy beds offer essential support for geriatric care, enabling safe patient transfers, pressure ulcer prevention, and enhanced mobility—factors that will sustain high demand.
Technological Integration and Smart Features
By 2026, smart technology integration will be a hallmark of advanced physical therapy beds. Features such as IoT connectivity, real-time patient monitoring, automated pressure redistribution, and AI-driven posture adjustment will become standard in premium models. These innovations not only improve patient outcomes but also reduce caregiver burden, making them highly attractive to hospitals, rehabilitation centers, and home healthcare providers.
Expansion of Home-Based Rehabilitation
The shift toward home-based care is accelerating, fueled by patient preference, cost-efficiency, and telehealth advancements. Physical therapy beds designed for home use—lightweight, easy to assemble, and equipped with remote monitoring—are expected to capture a growing market share. This trend is supported by government initiatives promoting decentralized healthcare and insurance coverage for home medical equipment.
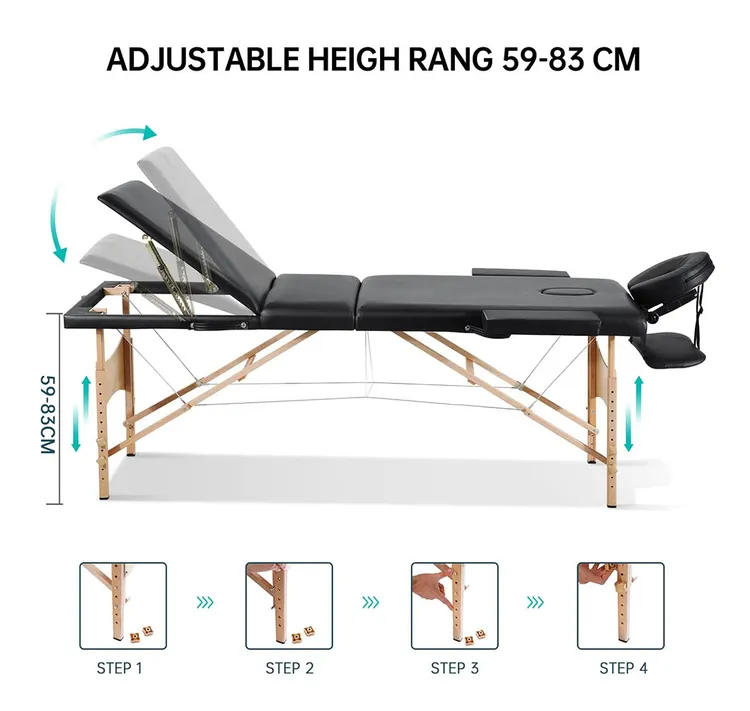
Focus on Ergonomics and Customization
Manufacturers are increasingly focusing on ergonomics and patient-specific customization. In 2026, physical therapy beds will offer modular designs, adjustable articulation, and compatibility with various therapeutic accessories (e.g., traction systems, gait trainers). This enables tailored rehabilitation programs, especially for patients recovering from orthopedic surgeries, neurological conditions, or spinal injuries.
Increased Investment in Emerging Markets
Emerging economies in Asia-Pacific, Latin America, and Africa are expected to show robust growth in the physical therapy bed market by 2026. Rising healthcare infrastructure, increased insurance penetration, and government support for rehabilitation services are creating new opportunities. Local manufacturing and partnerships with global brands will likely expand access and affordability.
Regulatory and Safety Standards
Stringent regulatory requirements related to medical device safety and performance will shape product development. In 2026, compliance with ISO 60601 (medical electrical equipment) and FDA/CE marking standards will be critical for market entry. Emphasis on infection control, fire resistance, and electromagnetic compatibility will drive innovation in materials and design.
Sustainability and Eco-Friendly Design
Environmental concerns are influencing medical equipment manufacturing. By 2026, leading producers of physical therapy beds are expected to adopt sustainable practices—using recyclable materials, minimizing packaging waste, and improving energy efficiency in electric models. This aligns with broader healthcare sustainability goals and appeals to environmentally conscious institutions.
Conclusion
The physical therapy bed market in 2026 will be defined by innovation, personalization, and accessibility. Driven by demographic shifts and digital transformation, the industry is moving toward smarter, safer, and more patient-centered solutions. Stakeholders—from manufacturers to healthcare providers—must adapt to these trends to meet evolving clinical needs and maintain competitive advantage.

Common Pitfalls in Sourcing Physical Therapy Beds: Quality and Intellectual Property Risks
Sourcing physical therapy beds, especially from international or lower-cost suppliers, presents several critical challenges. Overlooking quality assurance and intellectual property (IP) considerations can lead to product failures, legal disputes, reputational damage, and financial losses. Below are the most common pitfalls in these two crucial areas.
Quality-Related Pitfalls
1. Inadequate Compliance with Medical Device Standards
A major risk is sourcing beds that do not meet essential regulatory standards such as ISO 13485, IEC 60601 (electrical safety), or regional requirements like FDA 510(k) clearance in the U.S. or CE marking in Europe. Beds failing these standards may pose safety hazards to patients and therapists and could be barred from sale or use in regulated markets.
2. Substandard Materials and Construction
Some suppliers use lower-grade steel, weak motors, or poor-quality upholstery to cut costs. This leads to premature wear, mechanical failure, or hygiene issues—especially problematic in clinical environments where durability and infection control are paramount.
3. Inconsistent Manufacturing Processes
Lack of process controls during manufacturing can result in variability between units. For example, inconsistent welding or motor alignment may affect bed functionality, safety, or noise levels, undermining reliability and user trust.
4. Insufficient Testing and Validation
Reputable suppliers conduct rigorous testing for load capacity, cycle life, electrical safety, and ergonomics. Sourcing from vendors who skip or falsify these tests increases the risk of field failures, recalls, and patient injury.
5. Poor After-Sales Support and Spare Parts Availability
Even a high-quality bed becomes unusable if spare parts are unavailable or technical support is unresponsive. Sourcing from suppliers without a service network can lead to extended downtime and increased total cost of ownership.
Intellectual Property-Related Pitfalls
1. Supplying Counterfeit or Copycat Designs
Some manufacturers produce beds that closely mimic patented designs from leading brands (e.g., Hill-Rom, Stryker). Purchasing such products exposes the buyer to IP infringement claims, potential seizure of goods, and legal liability—even if the buyer was unaware of the violation.
2. Lack of IP Due Diligence in Supplier Vetting
Failing to verify whether a supplier holds legitimate rights to the design, technology, or brand name used can result in unintentional IP violations. This includes checking for valid patents, trademarks, and design registrations in relevant jurisdictions.
3. Inadequate Contractual IP Protections
Purchase agreements that do not clearly define IP ownership, especially for custom-designed beds or modifications, may lead to disputes. Without proper clauses, the buyer may not have rights to use, modify, or resell the product freely.
4. Risk of Technology Leakage in Custom Development
When collaborating with a supplier to develop a new physical therapy bed, sensitive design or functional specifications may be shared. Without robust non-disclosure agreements (NDAs) and IP assignment clauses, the supplier could reuse or sell the technology to competitors.
5. Trademark and Branding Infringement
Some suppliers may affix well-known brand logos or use similar names to imply endorsement or authenticity. Distributing such products—even unknowingly—can result in cease-and-desist orders, brand damage, and costly litigation.
Mitigation Strategies
- Conduct third-party audits and factory inspections to verify quality systems.
- Require compliance certifications and test reports before purchase.
- Perform IP searches and consult legal counsel to verify freedom to operate.
- Include strong IP clauses in supplier contracts, including indemnification.
- Build relationships with reputable, transparent manufacturers with verifiable track records.
Avoiding these pitfalls requires due diligence, clear contractual terms, and a proactive approach to both product quality and intellectual property integrity.
Logistics & Compliance Guide for Physical Therapy Bed
Product Classification and Regulatory Compliance
Physical therapy beds are classified as medical devices and are subject to regulatory oversight in most countries. In the United States, they typically fall under Class I or Class II medical devices regulated by the U.S. Food and Drug Administration (FDA). These beds must comply with FDA 21 CFR Part 890 (Physical Medicine Devices) and may require 510(k) premarket notification if they include advanced features such as electronic controls, positioning mechanisms, or integrated therapy systems. Manufacturers must register their facilities and list devices with the FDA. In the European Union, physical therapy beds must meet the requirements of the Medical Device Regulation (MDR) (EU) 2017/745 and carry the CE mark after undergoing conformity assessment, typically under Annex IX or via a Notified Body for higher-risk classifications. Other regions, including Canada (Health Canada), Australia (TGA), and Japan (PMDA), have similar regulatory frameworks requiring technical documentation, quality management systems (e.g., ISO 13485), and local registration.
Quality Management System (QMS) Requirements
All manufacturers and distributors of physical therapy beds must implement a compliant Quality Management System (QMS) in accordance with ISO 13485:2016. This international standard outlines requirements for a comprehensive management system for the design, development, production, installation, and servicing of medical devices. Key components include design controls, risk management per ISO 14971, document and record controls, supplier management, internal audits, and corrective and preventive actions (CAPA). A robust QMS ensures consistent product quality, regulatory compliance, and patient safety. Regular audits and continual improvement processes are mandatory to maintain certification and facilitate inspections by regulatory authorities.
Labeling and Packaging Standards
Labeling of physical therapy beds must adhere to jurisdiction-specific requirements. In the U.S., FDA 21 CFR Part 801 mandates that labels include the device name, intended use, manufacturer information, model and serial numbers, and any required warnings or contraindications. The Unique Device Identification (UDI) system requires each unit to bear a UDI barcode on the label and in the device master record. In the EU, labeling must include the CE mark, UDI, manufacturer details, and comply with MDR Annex I general safety and performance requirements. Packaging must protect the device during transit, include essential instructions for use (IFU), and meet environmental and safety standards such as ISTA 3A for transport testing. Multilingual labeling may be required for international distribution.
Import and Export Documentation
Shipping physical therapy beds across borders requires accurate and complete documentation to ensure customs clearance and compliance. Key documents include a commercial invoice, packing list, bill of lading or air waybill, and a certificate of origin. For regulated medical devices, additional documentation such as a Certificate to Foreign Government (CFG) from the FDA (for U.S. exports), CE Certificate of Conformity (for EU exports), or import licenses from destination countries may be required. Exporters must classify the product using the correct Harmonized System (HS) code—commonly 9019.10 or 9019.20 for therapeutic apparatus—and comply with export control regulations such as the U.S. Export Administration Regulations (EAR). Accurate classification prevents delays and ensures proper tariff application.
Transportation and Handling Protocols
Physical therapy beds are often heavy, bulky, and sensitive to impact and environmental conditions. They should be shipped via freight (LTL or FTL) using palletized packaging with protective corner boards, stretch wrapping, and, if necessary, moisture barriers. Use carriers experienced in medical equipment logistics to reduce handling risks. Temperature and humidity should be controlled during transit, especially for beds with electronic components. Handling instructions such as “This Side Up,” “Fragile,” and “Do Not Stack” must be clearly marked. Ensure compliance with transportation safety standards, including those from the International Safe Transit Association (ISTA), to minimize damage risk. Consider freight insurance to mitigate financial loss from transit incidents.
Installation, Training, and Post-Market Surveillance
Upon delivery, physical therapy beds must be properly installed and commissioned by qualified personnel in accordance with manufacturer instructions. Installers should verify electrical safety, mechanical function, and software calibration (if applicable). End-user training on safe operation, maintenance, and emergency procedures is essential for compliance and patient safety. Additionally, manufacturers must establish a post-market surveillance (PMS) system as required by FDA and MDR regulations. This includes monitoring device performance, collecting customer feedback, managing complaints, and reporting adverse events (e.g., via FDA’s MedWatch or EU’s Eudamed). Regular PMS reports support ongoing compliance and inform risk management updates.
Environmental and Disposal Compliance
At the end of life, physical therapy beds must be disposed of in accordance with local environmental and medical waste regulations. Components such as batteries, electronic controls, and motors may contain hazardous materials subject to WEEE (Waste Electrical and Electronic Equipment) directives in the EU or equivalent recycling programs elsewhere. Facilities should partner with certified e-waste recyclers to ensure environmentally responsible disposal. Hospitals and clinics must follow internal biomedical waste protocols, particularly if the bed has been used in infectious environments. Manufacturers should provide disposal guidance in the IFU to support compliance with environmental standards.
In conclusion, sourcing a physical therapy bed requires careful consideration of patient needs, clinical functionality, durability, and cost-effectiveness. It is essential to select a bed that supports therapeutic goals, enhances patient comfort, and facilitates ease of use for both patients and healthcare providers. Key factors such as adjustability, weight capacity, mobility, safety features, and compatibility with ancillary equipment must be evaluated to ensure optimal performance in rehabilitation settings. Additionally, choosing a reputable supplier with strong after-sales support, warranties, and compliance with medical device regulations ensures long-term reliability and patient safety. By conducting thorough research and involving clinical stakeholders in the decision-making process, healthcare facilities can make an informed investment that improves therapy outcomes and operational efficiency.