The global calcium supplement market is experiencing steady expansion, driven by rising awareness of bone health, an aging population, and increased demand for natural and bioavailable mineral sources. According to Grand View Research, the global calcium supplements market was valued at USD 11.3 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 7.2% from 2023 to 2030. This growth trajectory is further amplified by a shift toward natural calcium sources, such as oyster shell calcium, which is gaining traction in both nutraceutical and pharmaceutical applications due to its high calcium content and sustainable sourcing. Amid this demand, manufacturers producing oyster shell calcium and calcium carbonate—two of the most widely used forms—are scaling production and investing in purification and bioavailability enhancement technologies. While calcium carbonate remains the dominant form due to its low cost and high elemental calcium concentration, oyster shell calcium is emerging as a preferred choice in premium and eco-conscious segments. As the industry evolves, evaluating the top manufacturers in both categories becomes critical for formulators, brands, and distributors aiming to balance cost, efficacy, and consumer preference.
Top 8 Oyster Shell Calcium Vs Calcium Carbonate Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Oyster Shell Calcium Carbonate Powder
Domain Est. 2004
Website: caltronclays.com
Key Highlights: Caltron is one of the reputed manufacturers of Oyster Shell Calcium Carbonate. We offer the most authentic and purest form of oyster shell calcium powder….
#2 OYSTER SHELL CALCIUM 500 MG TB – 100EA
Domain Est. 2007
Website: txvendordrug.com
Key Highlights: Brand name: OYSTER SHELL CALCIUM 500 MG TB – 100EA ; Generic Name: calcium carbonate ; Manufacturer: APHENA PHARMA S ; NDC: 43353038065….
#3 Oyster Shell Calcium Carbonate Manufacturer from Mumbai
Domain Est. 2011
Website: calciumproduct.com
Key Highlights: The Calcium Carbonate derived from the Limestone mining has a lot of heavy metals as compared to the Oyster Shell Calcium Carbonate and hence it becomes an ……
#4 SuperTab®
Domain Est. 2021
Website: twghealthandnutrition.com
Key Highlights: SuperTab® Oyster Shell calcium carbonate is produced from shells naturally-harvested from undersea beds in the San Francisco Bay and are believed to be ……
#5 Effect of fortified calcium compounds from oyster shell on the quality …
Domain Est. 1997
Website: sciencedirect.com
Key Highlights: Oyster shells consist of approximately 96 % calcium carbonate (Silva et al., 2019), making them a low-cost natural source of calcium….
#6 World Market for Oyster Shell Calcium Carbonate Tablets
Domain Est. 2002
Website: pr.mono.ipros.com
Key Highlights: It includes information on the overview of the global oyster shell calcium carbonate tablets market, trends of major companies (sales, selling ……
#7 Comparison of Physicochemical Properties of Noodles Fortified with …
Domain Est. 2003
Website: mdpi.com
Key Highlights: Food products fortified with calcium from oyster shells have been demonstrated to prevent and treat osteoporosis among rats and humans [16,17]….
#8 Leading Companies
Domain Est. 2016
Website: coherentmarketinsights.com
Key Highlights: *Definition- Oyster shell calcium refers to a dietary supplement made from ground oyster shells that is rich in calcium carbonate, a mineral essential for bone ……
Expert Sourcing Insights for Oyster Shell Calcium Vs Calcium Carbonate
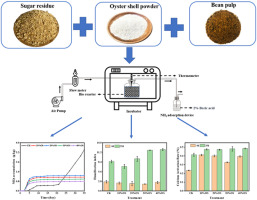
H2: Comparative Market Trends for Oyster Shell Calcium vs. Calcium Carbonate (2026 Outlook)
As the global demand for calcium supplements continues to rise in response to aging populations and increased awareness of bone health, two prominent forms—oyster shell calcium and calcium carbonate—are expected to experience divergent market trajectories by 2026. A detailed analysis highlights key trends in consumer preferences, sustainability concerns, regulatory developments, and technological advancements shaping their respective positions.
1. Market Demand and Consumer Preferences
- Calcium Carbonate: Dominates the market due to its cost-effectiveness, high elemental calcium content (40%), and widespread use in both pharmaceutical and nutraceutical industries. By 2026, it is projected to maintain a significant share—over 60% of the global calcium supplement market—due to its use in mass-market products like antacids and chewable tablets.
- Oyster Shell Calcium: Experiencing moderate but steady growth, driven by increasing demand for natural and organic supplements. Consumers seeking “whole food” or marine-sourced nutrients are increasingly opting for oyster shell calcium, perceiving it as more bioavailable and environmentally connected. The premium pricing associated with this form limits mass adoption but supports growth in niche wellness and eco-conscious markets.
2. Sustainability and Environmental Concerns
- Calcium Carbonate: Sourced primarily from limestone (mined) or as a byproduct of industrial processes, its production raises environmental issues related to quarrying and carbon emissions. However, advancements in carbon capture and sustainable mining practices are expected to mitigate some concerns by 2026.
- Oyster Shell Calcium: Benefits from the circular economy trend—utilizing waste shells from the seafood industry. With rising emphasis on ocean sustainability and shell recycling programs, oyster shell calcium is gaining favor as a sustainable alternative. By 2026, partnerships between aquaculture industries and supplement manufacturers are expected to scale up supply chains, improving availability and reducing ecological footprint.
3. Bioavailability and Health Perceptions
- Calcium Carbonate: Requires stomach acid for optimal absorption, making it less effective for elderly or acid-reduced populations. Despite this, its proven efficacy in clinical settings ensures continued trust among healthcare providers.
- Oyster Shell Calcium: Marketed as having superior bioavailability due to its natural matrix of calcium, magnesium, and trace minerals. While clinical evidence remains comparable to calcium carbonate, consumer perception favors oyster-derived calcium as a more “holistic” option—driving growth in the premium supplement segment.
4. Regulatory and Safety Trends
- Calcium Carbonate: Well-established regulatory approval across major markets (FDA, EFSA, etc.). Concerns over heavy metal contamination are minimal due to standardized purification.
- Oyster Shell Calcium: Faces scrutiny over potential heavy metal contamination (e.g., lead, cadmium) from marine sources. By 2026, stricter global standards and third-party testing certifications (such as USP or NSF) are expected to enhance consumer confidence, but compliance costs may limit smaller producers.
5. Regional Market Dynamics
- Asia-Pacific: Strong growth in both forms. Calcium carbonate dominates in countries like China and India due to affordability. Meanwhile, Japan and South Korea show rising interest in oyster shell calcium, leveraging local seafood byproducts.
- North America and Europe: Increasing demand for clean-label and sustainable products is boosting oyster shell calcium, especially in organic and vegan-certified supplements (when combined with plant-derived binders). However, calcium carbonate remains the default in insurance-covered and OTC health products.
6. Innovation and Product Development
- Calcium Carbonate: Focus remains on improving formulation (e.g., microencapsulation for better absorption, combination with vitamin D3 and K2).
- Oyster Shell Calcium: Innovation in processing technologies—such as low-temperature calcination and nano-sizing—is expected to enhance solubility and absorption, closing the bioavailability gap with synthetic forms.
Conclusion:
By 2026, calcium carbonate will continue to dominate the global market due to scalability, cost, and regulatory familiarity. However, oyster shell calcium is poised for accelerated growth in premium, sustainable, and niche health segments. Its success will depend on overcoming supply chain limitations, ensuring consistent quality, and capitalizing on the global shift toward natural and circular economy models. While not replacing calcium carbonate, oyster shell calcium is emerging as a compelling alternative for environmentally and health-conscious consumers.
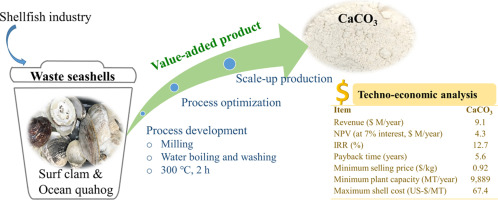
Common Pitfalls Sourcing Oyster Shell Calcium vs. Calcium Carbonate (Quality, IP)
When sourcing calcium supplements, understanding the key differences and potential pitfalls between Oyster Shell Calcium and purified Calcium Carbonate—especially regarding quality and intellectual property (IP) considerations—is critical for manufacturers, formulators, and brand owners. Here are the most common pitfalls to avoid:
Quality-Related Pitfalls
Inconsistent Purity and Contaminant Risk in Oyster Shell Calcium
Oyster Shell Calcium is a natural source derived from ground oyster shells, which introduces inherent variability and contamination risks:
– Heavy Metal Contamination: Oyster shells can accumulate lead, cadmium, arsenic, and mercury from marine environments. Sourcing from polluted waters significantly increases the risk of exceeding safety thresholds (e.g., USP, FDA, or EU limits). Inadequate testing protocols can result in unsafe products.
– Batch-to-Batch Variability: Natural sourcing leads to inconsistent calcium content (typically 30–40% elemental calcium), making precise dosing difficult. This variability impacts product formulation and efficacy.
– Microbial Contamination: Improper handling or drying can introduce pathogens such as Salmonella or E. coli, especially if processing does not include sterilization steps.
– Lack of Standardization: Unlike synthetic calcium carbonate, oyster shell lacks standardized manufacturing processes, increasing the risk of impurities and inconsistent particle size, which affects bioavailability.
Overlooking Processing and Additive Concerns in Calcium Carbonate
While pharmaceutical-grade Calcium Carbonate is highly purified and consistent, pitfalls still exist:
– Source Transparency: Calcium Carbonate can be derived from natural limestone or synthesized. Some consumers prefer “natural” sources, but suppliers may not disclose origin, leading to greenwashing or mislabeling risks.
– Excipients and Flow Agents: Commercial Calcium Carbonate may contain anti-caking agents (e.g., magnesium stearate, silicon dioxide). These additives can affect formulation stability or conflict with clean-label claims.
– Particle Size and Bioavailability: Finer particle sizes improve solubility and absorption. Sourcing low-quality or poorly milled calcium carbonate can reduce efficacy, especially in elderly or low-acid stomach populations.
Intellectual Property (IP) and Regulatory Pitfalls
Misrepresentation and Labeling Compliance
- “Natural” Claims Misuse: Marketing Oyster Shell Calcium as inherently “superior” or “more natural” without scientific substantiation can lead to regulatory scrutiny (e.g., FTC or FDA warning letters). Calcium Carbonate, even if mined, is chemically identical and equally bioavailable when properly processed.
- IP Infringement in Formulations: Proprietary blends or delivery systems (e.g., microencapsulation, chelated forms) using either calcium source may be protected by patents. Sourcing raw materials without reviewing formulation patents can expose companies to litigation.
Supply Chain and Certification Risks
- Lack of Certifications: Oyster Shell Calcium suppliers may lack certifications like GMP, ISO, or heavy metal testing reports. Relying on uncertified suppliers increases regulatory and reputational risk.
- Traceability Gaps: Without clear chain-of-custody documentation, proving the origin and safety of oyster shells becomes difficult, especially under FDA DSHEA or EU Novel Food regulations.
- Patented Purification Processes: Some high-purity Calcium Carbonate grades use patented purification techniques to remove trace metals. Using these without licensing can infringe on process patents.
Best Practices to Mitigate Risks
- Require Full COAs (Certificates of Analysis): Mandate testing for heavy metals, microbial load, and elemental calcium content for both materials.
- Audit Suppliers: Conduct on-site audits of oyster shell processors and calcium carbonate manufacturers to verify GMP compliance.
- Verify IP Status: Conduct freedom-to-operate (FTO) analyses before launching products using novel delivery systems or claims.
- Standardize on Pharmacopeial Grades: Use USP/NF or Ph. Eur. grade Calcium Carbonate when consistency and safety are paramount.
By recognizing these pitfalls, stakeholders can make informed sourcing decisions that balance consumer expectations, regulatory compliance, and product quality.

Logistics & Compliance Guide: Oyster Shell Calcium vs. Calcium Carbonate
Sourcing and Raw Material Acquisition
Oyster Shell Calcium
Sourcing oyster shell calcium involves harvesting or collecting shells from seafood processing facilities, aquaculture farms, or wild-harvest operations. Sustainability and traceability are critical concerns. Suppliers must demonstrate compliance with environmental regulations and ethical sourcing practices. Due to its animal origin, sourcing must adhere to food safety standards for animal by-products, including potential regional restrictions on shellfish harvesting (e.g., red tide zones). Documentation must confirm the geographical origin and processing history of shells.
Calcium Carbonate (Synthetic or Mineral-Based)
Calcium carbonate is typically derived from natural limestone, marble, or chalk through mining operations, or produced synthetically via chemical precipitation (e.g., soda ash process). Mineral sourcing requires adherence to mining regulations and environmental impact assessments. Synthetic production demands strict chemical process controls. Supply chains are generally more stable and scalable than oyster shell sources, with fewer geographic and biological constraints.
Manufacturing and Processing Requirements
Oyster Shell Calcium
Processing involves thorough cleaning, sterilization (e.g., high-temperature kilning), grinding, and micronization of shells to produce a bioavailable powder. Facilities must comply with Good Manufacturing Practices (GMP) for dietary supplements and implement Hazard Analysis and Critical Control Points (HACCP) to mitigate risks of microbial contamination (e.g., Vibrio spp.) and heavy metals (e.g., lead, cadmium). Cross-contamination with allergens must also be controlled due to shellfish origin.
Calcium Carbonate
Manufacturing includes crushing, purification, precipitation (if synthetic), and particle size reduction. The process is highly controlled and scalable. Synthetic calcium carbonate requires compliance with chemical manufacturing regulations (e.g., REACH in the EU). Both forms must meet pharmacopeial standards (e.g., USP, Ph. Eur.) for purity, but mineral-based calcium carbonate generally has lower inherent contamination risk compared to oyster-derived products.
Regulatory Classification and Labeling
Oyster Shell Calcium
Classified as a natural or animal-derived supplement ingredient in most jurisdictions. Labeling must declare “oyster shell calcium” and may require allergen warnings (e.g., “Contains shellfish”) in markets like the U.S. (FDA) and EU (FIC Regulation). Regulatory scrutiny is higher due to potential heavy metal content; products must meet strict limits (e.g., FDA’s guidance on lead in dietary supplements).
Calcium Carbonate
Widely accepted as a food additive (e.g., E170 in the EU) and dietary supplement. Labeling is straightforward, typically listed as “calcium carbonate.” Allergen warnings are not required unless processed in shared facilities with allergens. It is recognized in major pharmacopeias and generally regarded as safe (GRAS) by the FDA when used appropriately.
International Trade and Import/Export Compliance
Oyster Shell Calcium
Subject to agricultural, food safety, and animal by-product regulations. Export/import may require health certificates, proof of processing (e.g., heat treatment), and compliance with International Maritime Organization (IMO) guidelines for seafood by-products. Countries with strict biosecurity laws (e.g., Australia, New Zealand) may impose additional inspections or bans.
Calcium Carbonate
Treated as a chemical or mineral commodity. Trade is governed by chemical safety regulations (e.g., SDS requirements under GHS) and customs classifications (e.g., HS Code 2836.50). Fewer restrictions apply compared to animal-derived products, facilitating smoother cross-border logistics.
Storage, Handling, and Shelf Life
Oyster Shell Calcium
Requires dry, cool storage conditions to prevent moisture absorption and microbial growth. Packaging must be moisture-resistant and sealed to maintain stability. Due to organic residue risks, shelf life may be shorter than synthetic forms. Regular testing for microbial load and heavy metals during storage is recommended.
Calcium Carbonate
Highly stable under normal conditions. Can be stored in bulk silos or sealed bags without special climate controls. Long shelf life with minimal degradation. Less hygroscopic than oyster shell calcium, reducing packaging complexity and storage costs.
Quality Control and Testing Protocols
Oyster Shell Calcium
Mandatory testing includes heavy metals (Pb, Cd, As, Hg), microbial contaminants, calcium content (typically 30–35%), and species verification (to confirm oyster origin). Third-party certification (e.g., NSF, USP Verified) is advisable to build consumer trust. Batch traceability from shell source to final product is essential.
Calcium Carbonate
Testing focuses on purity, particle size, loss on ignition, and heavy metals. Calcium content is higher (~40%), offering more consistent dosing. Compliance with USP-NF or equivalent standards is standard. Synthetic grades may require additional checks for residual chemicals (e.g., sodium, chloride).
Environmental and Sustainability Considerations
Oyster Shell Calcium
Can support circular economy models by repurposing waste from seafood industries. However, overharvesting and habitat disruption are concerns if not managed sustainably. Certifications like MSC (Marine Stewardship Council) or adherence to local fisheries regulations improve compliance and market access.
Calcium Carbonate
Limestone mining has environmental impacts (e.g., habitat loss, CO₂ emissions from calcination). Synthetic production is energy-intensive. However, some manufacturers use carbon capture methods or recycled materials. Environmental Product Declarations (EPD) and carbon footprint assessments are increasingly important for compliance with green procurement policies.
Summary of Key Compliance and Logistics Differences
| Factor | Oyster Shell Calcium | Calcium Carbonate |
|—————————–|————————————-|—————————————|
| Origin | Animal (marine by-product) | Mineral or synthetic |
| Allergen Labeling Required | Yes (shellfish) | No |
| Heavy Metal Risk | Higher (requires rigorous testing) | Lower (controlled sourcing) |
| Regulatory Scrutiny | Higher (food safety & origin) | Moderate (chemical/mineral standards) |
| Import Restrictions | Possible (biosecurity concerns) | Minimal |
| Shelf Life & Stability | Moderate | High |
| Sustainability Profile | Waste-recovery potential | Higher carbon footprint (mining) |
| GMP & HACCP Requirements | Strict (due to biological origin) | Standard chemical GMP |
This guide enables informed decision-making for manufacturers, importers, and distributors navigating the regulatory and logistical landscape of calcium supplement sourcing.
Conclusion: Sourcing Oyster Shell Calcium vs. Calcium Carbonate
When evaluating oyster shell calcium versus purified calcium carbonate as sources of dietary calcium, several factors—including bioavailability, sustainability, purity, cost, and environmental impact—must be considered.
Oyster shell calcium is a natural form derived from ground oyster shells, often marketed as a more “whole food” or “natural” source. It contains calcium carbonate as its primary component, along with trace minerals like magnesium and phosphorus. However, its bioavailability is comparable to purified calcium carbonate, and it carries potential risks related to environmental contaminants such as lead, cadmium, and heavy metals, depending on the source waters. Sustainable sourcing is also a concern due to the ecological impact of oyster harvesting and shell collection.
In contrast, purified calcium carbonate is a widely used, cost-effective supplement derived from limestone or other mineral sources. It is highly concentrated and rigorously refined to meet pharmaceutical or food-grade standards, ensuring high purity and consistency. Its bioavailability is well-established, especially when taken with food, and it has a strong safety profile when sourced from reputable manufacturers.
Ultimately, while oyster shell calcium may appeal to consumers seeking natural or marine-based supplements, the benefits over purified calcium carbonate are minimal. The latter offers greater assurance of purity, consistent potency, and lower contamination risks. For most individuals seeking an effective and safe calcium supplement, purified calcium carbonate remains the more reliable and responsible choice, both from a health and environmental standpoint. Careful sourcing, transparency in production, and third-party testing are crucial—regardless of the calcium source chosen.