The global hydrothermal autoclave market has seen consistent expansion, driven by rising demand for advanced materials synthesis in research, pharmaceuticals, and nanotechnology applications. According to a 2023 report by Grand View Research, the global autoclave market size was valued at USD 2.1 billion and is expected to grow at a compound annual growth rate (CAGR) of 7.8% from 2023 to 2030, fueled by increasing adoption in material science and biotechnology sectors. Mordor Intelligence projects similar momentum, highlighting strong growth in R&D investments—particularly in Asia-Pacific—as a key driver for hydrothermal autoclave utilization. As industries prioritize precision, scalability, and contamination-free processing, the role of reliable manufacturers becomes increasingly critical. This growing demand has elevated the prominence of leading hydrothermal autoclave producers who combine engineering excellence with innovative design. Below are the top 9 manufacturers shaping this evolving landscape, selected based on technological innovation, customer reviews, global footprint, and market presence.
Top 9 Hydrothermal Autoclave Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
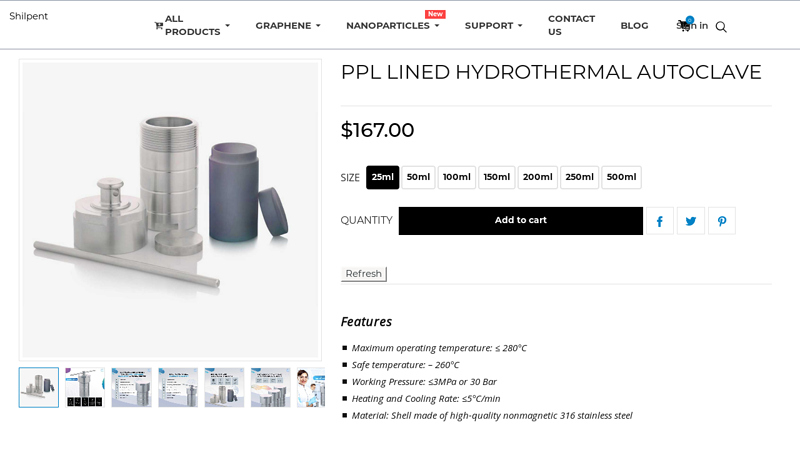
#1 PPL Lined Hydrothermal Autoclave Manufacturer and Supplier
Domain Est. 2011
Website: shilpent.com
Key Highlights: In stock Free delivery over $1,000Discover the PPL-lined hydrothermal autoclave from Shilpent—stainless steel shell, robust inner liner, up to 280 °C & 3 MPa, ideal for high-pressu…
#2 hydrothermal autoclave
Domain Est. 2014
Website: tobmachine.com
Key Highlights: Looking for best hydrothermal autoclave products? We are best hydrothermal autoclave suppliers,manufacturers and wholesalers from China….
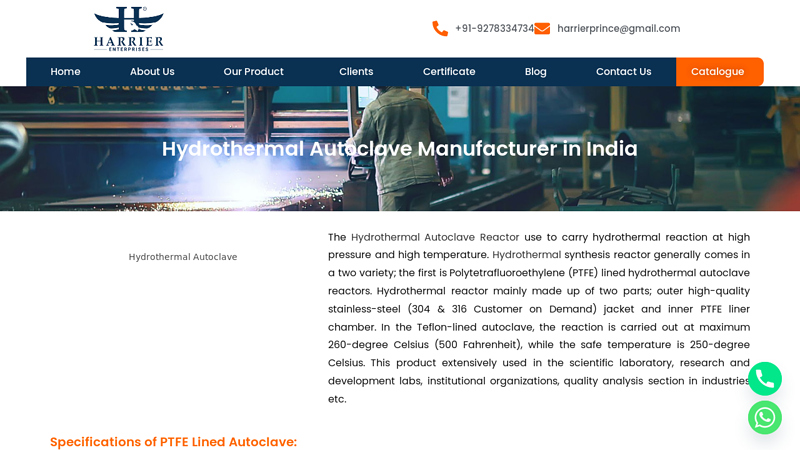
#3 Hydrothermal Autoclave Manufacturer in India
Domain Est. 2022
Website: harriergroups.com
Key Highlights: Harrier Enterprises is a leading manufacturer of Hydrothermal Autoclave in Delhi NCR, Gurugram, Noida, Greater Noida, Faridabad in India, For more details ……
#4 Hydrothermal Synthesis
Domain Est. 1995
Website: parrinst.com
Key Highlights: Parr Instrument Company manufactures a range of pressure vessels that completely encapsulate the sample within a PTFE environment….
#5 Hydrothermal Autoclave FM
Domain Est. 1999
Website: fison.com
Key Highlights: Rating 4.9 (728) · Free deliveryHydrothermal Autoclave is used for hydrothermal reactions, chemical reactions, biology, biochemistry, soil sciences, and catalysis studies. Acce…
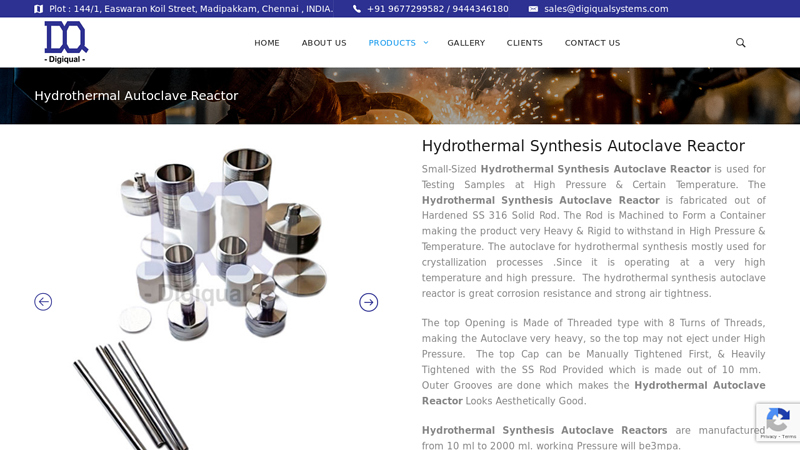
#6 Hydrothermal Synthesis Autoclave Reactor
Domain Est. 2008
Website: digiqualsystems.com
Key Highlights: Rating 4.9 Small-Sized Hydrothermal Synthesis Autoclave Reactor is used for Testing Samples at High Pressure & Certain Temperature….
#7 Hydrothermal Autoclave Reactor
Domain Est. 2013
Website: techinstro.com
Key Highlights: We offer high pressure and customized Hydrothermal Autoclave Reactor with standard capacities. Capacity such as 25ml, 50ml, 100ml, 150ml, ……
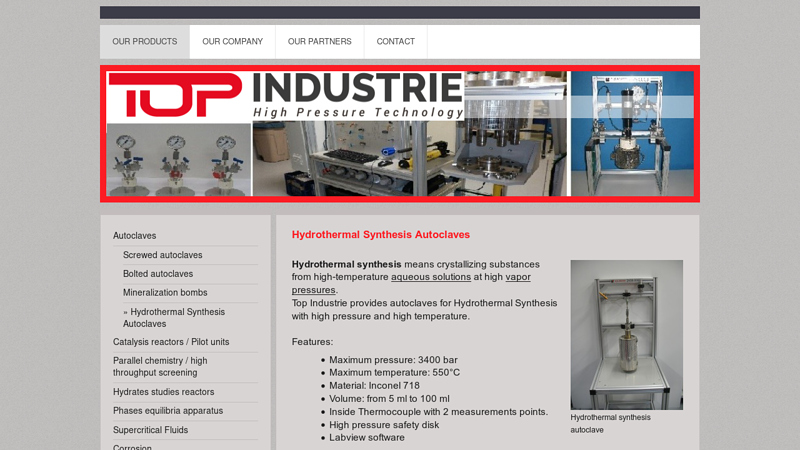
#8 Hydrothermal Synthesis Autoclaves
Domain Est. 2013
Website: topautoclave.com
Key Highlights: Top Industrie provides autoclaves for Hydrothermal Synthesis with high pressure and high temperature. Features: Maximum pressure: 3400 bar; Maximum temperature: ……
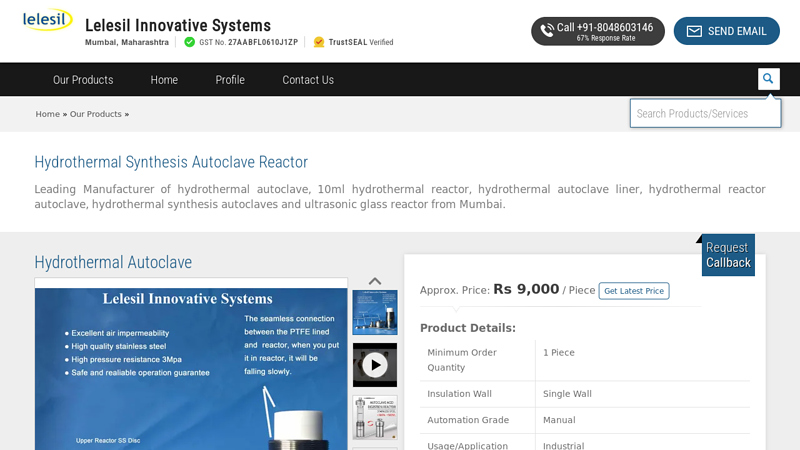
#9 Hydrothermal Synthesis Autoclave Reactor
Domain Est. 2023
Website: lelesil.co.in
Key Highlights: The Hydrothermal Autoclave Reactor use to carry hydrothermal reaction at high pressure and high temperature….
Expert Sourcing Insights for Hydrothermal Autoclave

H2: Market Trends for Hydrothermal Autoclaves in 2026
The global market for hydrothermal autoclaves is projected to experience significant growth and transformation by 2026, driven by increasing demand for advanced materials, sustainability initiatives, and technological innovation across key industries. Hydrothermal autoclaves—specialized pressure vessels used to synthesize materials under high temperature and pressure in aqueous environments—are critical in the production of nanomaterials, advanced ceramics, energy storage components, and sustainable chemical processes. Below is an analysis of the key market trends expected to shape the hydrothermal autoclave sector in 2026:
1. Growing Demand in Nanomaterials and Advanced Ceramics
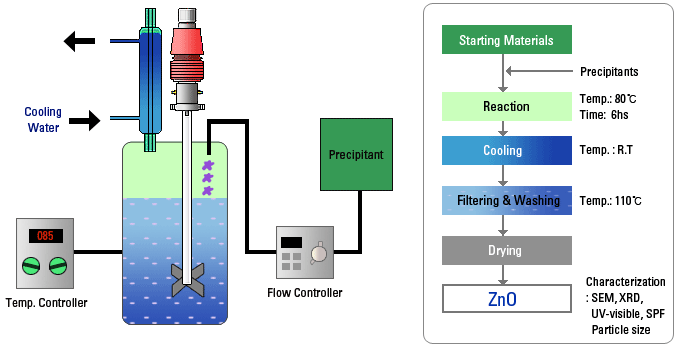
By 2026, the nanotechnology and advanced ceramics sectors are anticipated to be primary growth drivers for hydrothermal autoclaves. These industries rely heavily on hydrothermal synthesis to produce high-purity, uniform nanoparticles such as titanium dioxide, zinc oxide, and metal-organic frameworks (MOFs). The expansion of applications in electronics, coatings, and biomedical devices will increase adoption of autoclaves capable of precise temperature and pressure control.
2. Rise in Green Chemistry and Sustainable Manufacturing
With heightened focus on sustainable practices, hydrothermal methods are gaining favor over traditional high-temperature solid-state synthesis due to lower energy consumption and reduced emissions. Governments and private enterprises are investing in green chemistry solutions, promoting the use of water-based reactions. Hydrothermal autoclaves are central to this shift, enabling eco-friendly synthesis of catalysts, pigments, and battery materials without toxic solvents.
3. Expansion in Energy Storage and Battery Technologies
The booming lithium-ion and next-generation battery markets (e.g., solid-state, sodium-ion) are creating demand for cathode and anode materials synthesized via hydrothermal processes. By 2026, manufacturers will increasingly adopt hydrothermal autoclaves for scalable production of lithium iron phosphate (LFP), nickel-rich NMC, and other electrode materials with precise morphology and high electrochemical performance.
4. Advancements in Autoclave Design and Automation
Innovation in autoclave technology—such as enhanced corrosion resistance, modular designs, real-time monitoring, and integration with IoT and AI—is expected to improve process efficiency and reproducibility. Automated systems with feedback control for temperature, pressure, and pH will become standard, allowing for continuous operation and reducing human error in research and industrial settings.
5. Increased R&D Investment in Academia and Industry
Universities, government research labs, and private companies are investing heavily in materials science R&D, particularly in Asia-Pacific and North America. This trend will drive procurement of hydrothermal autoclaves for experimental and pilot-scale applications. Collaborative initiatives between academia and industry will accelerate the commercialization of new materials developed through hydrothermal synthesis.
6. Regional Market Growth
The Asia-Pacific region—led by China, Japan, and South Korea—is expected to dominate the hydrothermal autoclave market in 2026 due to robust industrialization, government support for high-tech manufacturing, and strong presence of electronics and battery producers. North America and Europe will follow, driven by innovation in clean energy and nanotechnology.
7. Customization and Scalability Needs
As industries transition from lab-scale experiments to commercial production, demand for scalable hydrothermal systems will rise. Equipment manufacturers will focus on offering customizable autoclaves with larger volumes (up to 100+ liters) and improved safety features to meet industrial requirements while maintaining synthesis quality.
8. Regulatory and Safety Standards
By 2026, stricter safety and compliance standards for high-pressure equipment are expected, especially in Europe and North America. This will push manufacturers to adopt certified designs, advanced pressure relief mechanisms, and remote operation capabilities, influencing purchasing decisions and product development.
Conclusion:
By 2026, the hydrothermal autoclave market will be shaped by technological innovation, sustainability imperatives, and rising demand from high-growth industries such as energy storage, nanotechnology, and green chemistry. Companies that invest in scalable, smart, and eco-efficient autoclave systems will be best positioned to capitalize on these trends. Continued collaboration between material scientists, equipment manufacturers, and industrial end-users will be critical to unlocking new applications and driving market expansion.
H2: Common Pitfalls When Sourcing Hydrothermal Autoclaves (Quality & Intellectual Property)
Sourcing hydrothermal autoclaves involves navigating significant risks related to both equipment quality and intellectual property (IP) protection. Failing to address these pitfalls can lead to safety hazards, operational failures, financial losses, and legal disputes. Key pitfalls include:
H3: Quality-Related Pitfalls
-
Inadequate Material Selection & Corrosion Resistance:
- Pitfall: Suppliers may use substandard alloys (e.g., inferior grades of stainless steel like 304 instead of 316L, or insufficient Hastelloy/Inconel) that cannot withstand the harsh, high-temperature, high-pressure, and often corrosive (acidic/alkaline) environments. This leads to premature vessel failure, leaks, contamination, and catastrophic safety risks.
- Mitigation: Specify exact material grades (ASTM/ASME standards), demand certified material test reports (MTRs), and verify compatibility with specific process chemicals and temperature/pressure cycles. Require corrosion resistance data.
-
Insufficient Pressure & Temperature Rating & Safety Certification:
- Pitfall: Autoclaves may be rated only for nominal conditions, lacking sufficient safety margins (e.g., 4:1 safety factor) or proper certification (ASME BPVC Section VIII, PED, etc.). This increases the risk of over-pressurization and explosion. Seals and gaskets may also be inadequate.
- Mitigation: Demand full ASME or equivalent certification with stamped data reports. Verify the Maximum Allowable Working Pressure (MAWP) and temperature ratings exceed your process requirements by a significant margin. Inspect safety features (burst disks, pressure relief valves, interlocks).
-
Poor Manufacturing Tolerances & Welding Quality:
- Pitfall: Poor welding (lack of fusion, porosity, cracks) or machining errors create weak points prone to failure under cyclic stress. Misaligned flanges or ports cause leaks.
- Mitigation: Require detailed fabrication procedures (WPS/PQR), non-destructive testing (NDT) reports (RT, PT, UT, VT), and third-party inspection (API 510/570, or authorized inspector).
-
Inferior Sealing Systems:
- Pitfall: Using low-quality gaskets (e.g., standard PTFE instead of reinforced PTFE, PEEK, or specialized elastomers) or poorly designed sealing mechanisms (e.g., single O-ring, inadequate clamping force) leads to leaks of high-pressure fluids or gases, contaminating experiments and posing safety hazards.
- Mitigation: Specify high-temperature/pressure rated gasket materials and proven sealing designs (e.g., metal C-rings, double O-rings with backup rings, capillary seals). Verify clamping mechanism integrity.
-
Lack of Traceability & Documentation:
- Pitfall: Absence of a complete “birth certificate” (material certs, weld logs, NDT reports, pressure test records, calibration certificates) makes it impossible to verify quality, ensure compliance, or troubleshoot issues later. Voided warranties.
- Mitigation: Contractually require a comprehensive Dossier or Data Package as part of the delivery.
H3: Intellectual Property (IP)-Related Pitfalls
-
Unprotected Design & Reverse Engineering:
- Pitfall: Suppliers, especially in regions with weak IP enforcement, may reverse engineer your unique autoclave design (e.g., specialized liner, port configuration, heating/cooling system) and sell identical or similar units to your competitors without your consent.
- Mitigation: File patents and design registrations before disclosing detailed specifications. Use robust, jurisdiction-specific Non-Disclosure Agreements (NDAs) covering design details. Limit disclosure to only essential information. Consider manufacturing in regions with stronger IP protection.
-
Insufficient Contractual IP Clauses:
- Pitfall: Standard purchase orders often lack clear language defining ownership of improvements, modifications, or tooling developed during the manufacturing process. The supplier may claim rights to enhancements made to your design.
- Mitigation: Negotiate and include explicit IP clauses in the contract stating that all designs, specifications, modifications, tooling, and improvements based on your input remain your sole and exclusive property.
-
Co-Mingling of IP in Custom Designs:
- Pitfall: When collaborating on a custom autoclave, it can be unclear where your proprietary IP ends and the supplier’s standard technology begins. Suppliers might incorporate your ideas into their standard product line.
- Mitigation: Clearly define IP boundaries in the contract. Use a “Background IP” (yours) vs. “Foreground IP” (developed for the project) framework, stipulating that Foreground IP related to your requirements defaults to your ownership. Audit supplier catalogs post-delivery.
-
Lack of Control Over Production & Tooling:
- Pitfall: The supplier retains exclusive control over the molds, jigs, or CNC programs used to make your custom autoclave. They can potentially replicate it without your knowledge.
- Mitigation: Specify in the contract that ownership of custom tooling/jigs/fixtures created for your project transfers to you upon payment, or at least grant you exclusive rights to its use. Require physical control or secure storage.
-
Inadequate Enforcement in High-Risk Jurisdictions:
- Pitfall: Even with contracts and patents, enforcing IP rights can be prohibitively expensive, slow, or ineffective in certain countries, making legal remedies impractical.
- Mitigation: Prioritize sourcing from suppliers in jurisdictions with reputable IP courts and enforcement mechanisms. Factor in the cost and risk of potential IP infringement when selecting suppliers. Consider using trusted intermediaries or joint ventures with local partners who have a vested interest in protecting IP.
Conclusion: Successfully sourcing hydrothermal autoclaves requires rigorous due diligence focused on verifiable quality assurance and proactive, legally sound IP protection strategies. Never compromise on safety certifications and material integrity, and always secure your proprietary designs through robust contracts and appropriate IP filings before engaging suppliers.

H2: Logistics & Compliance Guide for Hydrothermal Autoclave
The safe and compliant use, transport, storage, and maintenance of hydrothermal autoclaves—pressure vessels used in laboratories for high-temperature and high-pressure chemical reactions—requires strict adherence to logistics and regulatory standards. This guide outlines key considerations under the H2 heading for institutions, researchers, and operators.
H2.1 Regulatory Classification and Standards
- Pressure Equipment Directive (PED) 2014/68/EU (Europe): Hydrothermal autoclaves are classified as pressure equipment. Compliance with CE marking and conformity assessment procedures is mandatory for operation within the EU.
- ASME Boiler and Pressure Vessel Code (Section VIII, Division 1) (USA): Autoclaves must conform to ASME standards for design, fabrication, and inspection. Use of the ASME “U” stamp is required for commercial units.
- ISO 21930:2017: Applies to sustainability and environmental declarations relevant to laboratory equipment lifecycle.
- Local Jurisdictional Regulations: Check state/provincial codes (e.g., OSHA in the U.S., HSE in the UK) for workplace safety and pressure system regulations.
H2.2 Transport and Shipping Requirements
- UN Recommendations on the Transport of Dangerous Goods:
- Empty autoclaves: Generally non-hazardous if fully depressurized, cleaned, and purged of residual chemicals.
- Autoclaves containing residual solvents or reactive materials: May fall under Class 4 (flammable solids), Class 6.1 (toxic), or Class 8 (corrosive) depending on content.
- Packaging:
- Use robust, shock-resistant packaging with internal supports to prevent movement.
- Secure lids and seals; use protective caps on threaded ports.
- Documentation:
- Safety Data Sheets (SDS) for any residual substances.
- Declaration of non-hazardous status (for empty units) or proper hazardous material shipping papers.
- Carrier Compliance: Ensure carriers are certified for handling scientific equipment or hazardous materials as applicable.
H2.3 Import/Export Controls
- Dual-Use Goods: Autoclaves capable of operating above 500°C and 100 MPa may be subject to export controls under the Wassenaar Arrangement.
- Licensing: Verify if export licenses are required (e.g., U.S. Department of Commerce, Bureau of Industry and Security – EAR99 or specific ECCN).
- Country-Specific Restrictions: Some nations restrict import of high-pressure equipment for industrial or security reasons.
H2.4 Installation and Site Requirements
- Structural Support: Ensure floor loading capacity meets the combined weight of the autoclave, vessel, and contents.
- Ventilation: Operate in a well-ventilated fume hood or ventilated enclosure, especially when handling volatile precursors (e.g., ammonia, organic solvents).
- Utilities:
- Stable power supply (voltage, frequency, grounding).
- Access to cooling water (if equipped with cooling jacket).
- Clearance: Maintain minimum clearance (per manufacturer) around the unit for heat dissipation and emergency access.
H2.5 Operational Safety & Compliance
- Pressure and Temperature Monitoring: Use calibrated sensors with high-limit cutoffs and audible alarms.
- Personal Protective Equipment (PPE): Heat-resistant gloves, face shield, lab coat, and closed-toe shoes are mandatory.
- Standard Operating Procedures (SOPs): Documented protocols for loading, operation, depressurization, and emergency shutdown.
- Training: Personnel must be trained in pressure system safety, chemical handling, and emergency response.
H2.6 Maintenance & Inspection
- Routine Checks:
- Inspect seals, gaskets, bolts, and safety valves before each use.
- Verify integrity of pressure relief devices (burst discs, pressure relief valves).
- Scheduled Maintenance:
- Follow manufacturer-recommended intervals for servicing.
- Recertification of pressure vessels every 2–5 years (depending on jurisdiction and usage).
- Record Keeping:
- Maintain logs of inspections, repairs, pressure tests, and operational incidents.
H2.7 Waste and Decommissioning
- Decontamination: Thoroughly clean and neutralize internal components before servicing or disposal.
- Disposal:
- Metal components may be recycled as scrap metal.
- Contaminated parts (e.g., Teflon liners with heavy metals) must be disposed of as hazardous waste.
- End-of-Life Certification: Document decommissioning and ensure safe destruction of pressure-containing parts if not reused.
H2.8 Emergency Preparedness
- Emergency Shutdown: Clearly labeled emergency stop button and isolation valves.
- Spill Response: Kits for chemical spills (acid/base/organic) must be nearby.
- Evacuation Plan: Integrate autoclave risks into lab emergency plans, including pressure rupture or thermal runaway scenarios.
- Reporting: Report all incidents (leaks, overpressure events) per institutional and regulatory requirements (e.g., OSHA, RIDDOR).
Adherence to this H2 logistics and compliance framework ensures the safe, legal, and efficient operation of hydrothermal autoclaves in research and industrial environments. Always consult the manufacturer’s manual and local regulatory authorities for site-specific requirements.
Conclusion for Sourcing Hydrothermal Autoclave
Sourcing a hydrothermal autoclave requires careful consideration of several key factors including material compatibility, pressure and temperature ratings, safety features, volume capacity, and intended application. It is essential to select a supplier with a proven track record of quality manufacturing, adherence to international safety standards, and reliable after-sales support. Whether for research, nano-materials synthesis, or industrial processing, investing in a high-quality hydrothermal autoclave ensures operational efficiency, reproducibility of results, and long-term safety. By evaluating technical specifications against project requirements and conducting due diligence on potential vendors, organizations can make informed procurement decisions that support both current and future experimental or production needs. Ultimately, a well-sourced hydrothermal autoclave serves as a critical and durable asset in advancing scientific and industrial innovation.