The global cardiovascular devices market, including surgical instruments like Debakey clamps, is witnessing steady expansion driven by rising cardiovascular disease prevalence and increasing surgical volumes worldwide. According to Grand View Research, the global cardiovascular devices market size was valued at USD 54.7 billion in 2023 and is expected to grow at a compound annual growth rate (CAGR) of 6.3% from 2024 to 2030. This sustained growth underscores the critical demand for high-precision surgical instruments such as the Debakey clamp, widely used in vascular and cardiothoracic procedures for atraumatic vessel manipulation. As hospitals and surgical centers prioritize instrument quality, durability, and ergonomic design, selecting reliable manufacturers becomes paramount. Based on market presence, product quality, and innovation, the following four companies have emerged as leading Debakey clamp manufacturers, consistently meeting the rigorous standards of modern surgical practice.
Top 4 Debakey Clamp Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
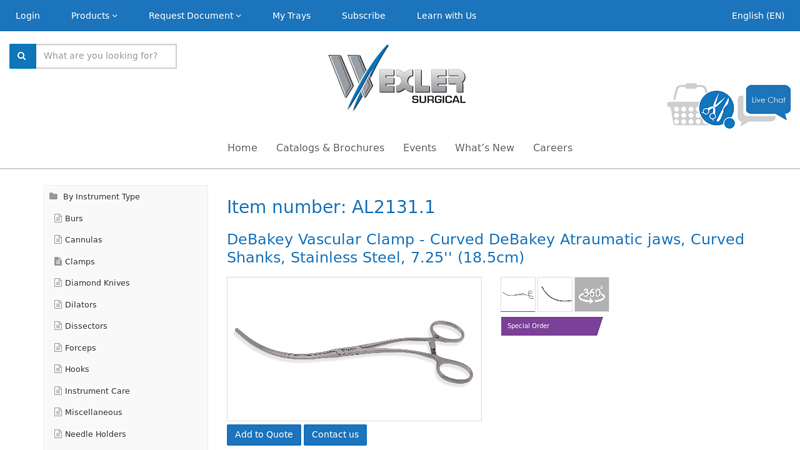
#1 DeBakey Vascular Clamp
Domain Est. 1997
Website: wexlersurgical.com
Key Highlights: The DeBakey Vascular Clamp from Wexler Surgical is made of Stainless Steel. The clamp has curved DeBakey Atraumatic Jaws along with a curved shank….
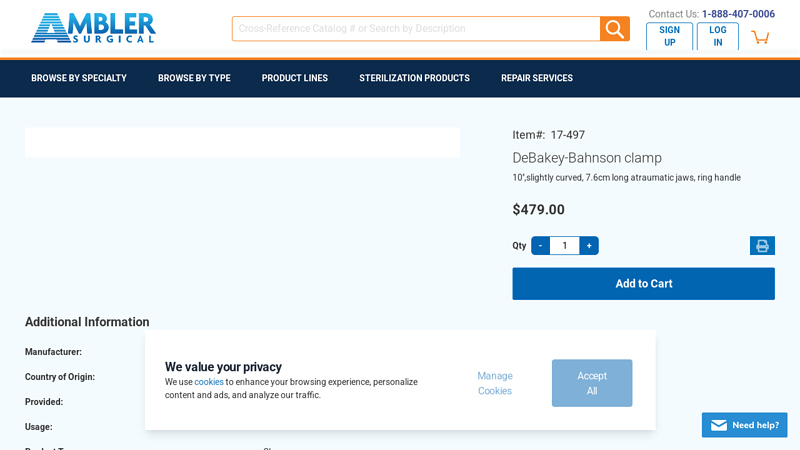
#2 DeBakey
Domain Est. 2003
Website: amblersurgical.com
Key Highlights: In stock $38.22 deliveryDeBakey-Bahnson clamp 10”,slightly curved, 7.6cm long atraumatic jaws, ring handle. $479.00. Item# 17-497….
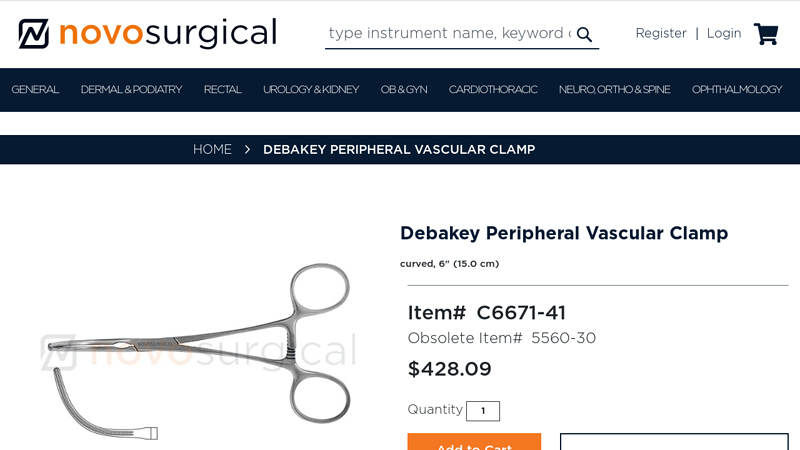
#3 Debakey Peripheral Vascular Clamp
Domain Est. 2009
Website: novosurgical.com
Key Highlights: In stock $23.30 deliveryNovo Surgical’s DeBakey Peripheral Vascular Clamp is designed for use in cardiothoracic surgical procedures where it is necessary to block or occlude blood …
#4 DeBakey® Coarctation Clamps
Domain Est. 2020
Website: us.myteleflex.com
Key Highlights: These are multipurpose clamps, full length but lightweight, with Debakey-type Jaws and spring handles including 1 x 9 ratchets….
Expert Sourcing Insights for Debakey Clamp
H2: Market Trends for the Debakey Clamp in 2026
As the global healthcare landscape evolves, the market for cardiovascular surgical instruments, including the Debakey clamp, is poised for significant transformation by 2026. The Debakey clamp—atraumatic vascular clamp commonly used in cardiovascular and reconstructive surgeries—continues to play a critical role in procedures such as coronary artery bypass grafting (CABG), aortic surgery, and vascular anastomosis. Several key trends are expected to shape its market dynamics in 2026, driven by technological advancements, demographic shifts, and evolving surgical practices.
1. Rising Prevalence of Cardiovascular Diseases (CVDs)
The increasing global burden of cardiovascular diseases remains a primary growth driver. According to the World Health Organization (WHO), CVDs are the leading cause of death worldwide, with an estimated 23.6 million deaths annually by 2030. This surge in CVD incidence directly correlates to higher demand for cardiovascular surgical interventions, thereby boosting the need for precision instruments like the Debakey clamp. Aging populations in North America, Europe, and parts of Asia-Pacific are particularly contributing to this trend.
2. Technological Integration and Product Innovation
By 2026, manufacturers are expected to focus on enhancing the functionality and ergonomics of the Debakey clamp through advanced materials and smart instrumentation. Developments such as titanium-based lightweight designs, antimicrobial coatings to reduce infection risks, and integration with robotic-assisted surgical systems (e.g., da Vinci Surgical System) are gaining traction. These innovations aim to improve surgical precision, reduce tissue trauma, and support minimally invasive procedures, which are increasingly favored in modern cardiac surgery.
3. Growth in Minimally Invasive and Robotic-Assisted Surgeries
The shift toward minimally invasive cardiac surgery (MICS) and robotic platforms is reshaping instrument demand. While traditional open-heart procedures still dominate, the adoption of smaller, specialized versions of the Debakey clamp compatible with endoscopic and robotic systems is growing. Companies are responding by developing micro-sized or articulated variants that maintain atraumatic properties while fitting through smaller incisions.
4. Expansion in Emerging Markets
Healthcare infrastructure improvements in emerging economies—particularly in India, China, Brazil, and Southeast Asia—are expanding access to advanced cardiac care. Rising disposable incomes, government healthcare initiatives, and increased medical tourism are fueling demand for high-quality surgical instruments. Local manufacturing and partnerships with global medtech firms are expected to enhance market penetration of Debakey clamps in these regions by 2026.
5. Stringent Regulatory Standards and Quality Assurance
Regulatory frameworks such as the EU Medical Device Regulation (MDR) and FDA guidelines are pushing manufacturers to ensure higher standards of sterility, biocompatibility, and traceability. This trend is leading to increased investment in quality control and documentation, especially for reusable surgical instruments like the Debakey clamp. Compliance is becoming a competitive differentiator in global tenders and hospital procurement processes.
6. Sustainability and Reusability Focus
Environmental concerns and cost-efficiency are driving a renewed emphasis on reusable surgical instruments. Unlike single-use alternatives, high-quality Debakey clamps made from durable stainless steel or titanium offer long-term value. Hospitals are increasingly adopting reprocessing protocols to extend instrument lifespan, aligning with sustainability goals and reducing operational costs.
Conclusion
In 2026, the Debakey clamp market will be shaped by the confluence of medical necessity, technological innovation, and global healthcare expansion. While remaining a staple in cardiovascular surgery, the instrument is evolving to meet the demands of modern surgical environments. Market players that prioritize innovation, regulatory compliance, and accessibility in emerging regions are likely to capture significant growth opportunities in the coming years.

Common Pitfalls When Sourcing Debakey Clamps (Quality and Intellectual Property)
Sourcing Debakey clamps—precision surgical instruments used in vascular and cardiac procedures—requires careful attention to both quality assurance and intellectual property (IP) compliance. Overlooking these aspects can lead to compromised patient safety, legal liabilities, and reputational damage. Below are key pitfalls to avoid:
Poor Manufacturing Quality and Material Standards
One of the most significant risks is acquiring clamps made from substandard materials or with inconsistent craftsmanship. Low-quality Debakey clamps may use inferior stainless steel that lacks corrosion resistance, leading to instrument degradation, increased infection risk, or failure during surgery. Poor machining can result in misaligned jaws, weak ratchets, or rough surfaces that damage delicate tissues. Always verify that suppliers adhere to international standards such as ISO 13485 and provide material certification (e.g., ASTM F899) for surgical-grade stainless steel.
Lack of Regulatory Compliance
Sourcing from manufacturers or distributors that do not comply with regional medical device regulations—such as FDA 510(k) clearance in the U.S., CE marking in Europe, or other local approvals—can result in legal penalties and import restrictions. Non-compliant instruments may not meet sterility, biocompatibility, or performance requirements, posing serious risks in clinical settings.
Counterfeit or Unlicensed Replicas
Debakey clamps are often subject to intellectual property protections, including design patents and trademarks. Sourcing from unauthorized suppliers increases the risk of purchasing counterfeit or “look-alike” instruments that imitate the original design without proper licensing. These replicas not only infringe on IP rights but are frequently of lower quality and lack traceability. Always confirm that the supplier is an authorized distributor or holds proper licensing agreements with the IP holder.
Inadequate Documentation and Traceability
Reliable sourcing requires full traceability, including lot numbers, sterilization records, and certificates of conformance. Suppliers that fail to provide detailed documentation make it difficult to validate quality or respond effectively to recalls or audits. This lack of transparency can compromise patient safety and regulatory compliance.
Insufficient Due Diligence on Suppliers
Relying solely on price or convenience without vetting the supplier’s reputation, manufacturing processes, or quality control systems can lead to long-term issues. Conduct thorough due diligence, including on-site audits or third-party certifications, to ensure the supplier maintains consistent quality and ethical manufacturing practices.
By addressing these pitfalls proactively, healthcare providers and procurement teams can ensure they source genuine, high-quality Debakey clamps that meet both clinical and legal standards.

Logistics & Compliance Guide for DeBakey Clamp
Proper logistics and compliance handling are essential to ensure the safe, effective, and legally compliant distribution and use of medical devices such as the DeBakey Clamp. This guide outlines key considerations for manufacturers, distributors, healthcare providers, and logistics partners.
Regulatory Classification and Approval
The DeBakey Clamp is classified as a Class II medical device under the U.S. FDA’s regulatory framework, falling under cardiovascular surgical instruments (21 CFR 870.4350). It requires 510(k) premarket notification demonstrating substantial equivalence to a legally marketed predicate device. Equivalent classifications apply in other regions:
- European Union: Must bear the CE mark under the Medical Device Regulation (MDR) (EU) 2017/745.
- Canada: Requires a Medical Device License (MDL) from Health Canada.
- Australia: Listed on the Australian Register of Therapeutic Goods (ARTG).
- Other Regions: Local approvals may be required based on national regulatory authorities.
Ensure all devices are distributed with up-to-date regulatory documentation and conformity certificates.
Labeling and Packaging Requirements
Proper labeling and packaging are critical for compliance and patient safety.
- Sterile Packaging: The DeBakey Clamp must be provided in single-use, sterile, tamper-evident packaging compliant with ISO 11607 standards.
- Labeling Elements:
- Device name and model number
- “Sterile” and method of sterilization (e.g., EO or gamma)
- Expiration date
- Single-use symbol (if applicable)
- Manufacturer name and address
- UDI (Unique Device Identifier) barcode compliant with FDA and EU MDR requirements
- IFU (Instructions for Use) included or accessible
Ensure labeling is accurate, multilingual where required, and adheres to local language regulations in target markets.
Storage and Handling Conditions
To maintain device integrity:
- Temperature: Store in a dry, climate-controlled environment between 15°C and 30°C (59°F to 86°F).
- Humidity: Relative humidity below 60% to prevent corrosion or packaging degradation.
- Light: Avoid prolonged exposure to direct sunlight or UV radiation.
- Orientation: Store upright or flat; avoid stacking heavy items on packages to prevent damage.
Monitor and document environmental conditions in warehouses using calibrated sensors and data loggers.
Transportation and Distribution
Transportation must preserve sterility and structural integrity.
- Shipping Containers: Use validated, shock-absorbent packaging designed for medical devices.
- Cold Chain (if applicable): While not typically required for metallic clamps, ensure ambient conditions are maintained during transit.
- Carrier Compliance: Use carriers experienced in medical device logistics with GDP (Good Distribution Practice) compliance.
- Tracking: Implement real-time shipment tracking with temperature and humidity monitoring if required.
- Import/Export Documentation:
- Commercial invoice
- Packing list
- Certificate of Conformity
- Bill of Lading/Air Waybill
- FDA Prior Notice (U.S. imports)
- Customs clearance forms per destination country
Import/Export and Customs Compliance
Ensure compliance with international trade regulations:
- HS Code: Typically classified under 9018.90 (Other medical/surgical instruments).
- Export Controls: Verify no dual-use or ITAR restrictions apply.
- Duty and Tax Optimization: Leverage trade agreements and bonded warehousing where applicable.
- Documentation Accuracy: Mislabeling or incorrect classification may lead to delays, fines, or seizure.
Engage customs brokers familiar with medical device regulations in target markets.
Post-Market Surveillance and Reporting
Manufacturers are responsible for ongoing compliance after device distribution.
- UDI Implementation: Record and report device implantation or use where required (e.g., in electronic health records).
- Adverse Event Reporting:
- FDA: Report to MedWatch within 30 days of becoming aware of an incident.
- EU: Report to EUDAMED under MDR vigilance requirements.
- Other regions: Follow local incident reporting timelines.
- Field Safety Notices: Issue corrective actions promptly for recalls or safety alerts.
- Audit Readiness: Maintain records of distribution, complaints, and corrective actions for at least 10 years (per FDA and EU MDR).
Reusable vs. Single-Use Considerations
Clarify the intended use model:
- Most DeBakey Clamps are reusable surgical instruments made of stainless steel.
- If reusable:
- Include detailed reprocessing instructions (cleaning, disinfection, sterilization).
- Validate reprocessing per AAMI ST79 and FDA guidance.
- Track device lifecycle and retirement criteria.
- If single-use:
- Clearly label “For Single Use Only.”
- Prevent reprocessing through design and regulatory compliance.
Training and Stakeholder Communication
- Healthcare Staff: Provide training on proper handling, use, and reprocessing (if applicable).
- Distribution Partners: Share compliance requirements and emergency procedures.
- Regulatory Updates: Subscribe to regulatory alerts (e.g., FDA, EMA) to remain current on changes.
Conclusion
Effective logistics and compliance management for the DeBakey Clamp ensures patient safety, regulatory adherence, and supply chain integrity. Stakeholders must collaborate to uphold standards from manufacturing through end-use, leveraging documentation, technology, and regulatory expertise.
Conclusion for Sourcing DeBakey Clamp
In conclusion, sourcing a DeBakey clamp requires careful consideration of quality, regulatory compliance, and supplier reliability. As a precision surgical instrument widely used in vascular and cardiothoracic procedures, it is essential to procure the clamp from reputable manufacturers or distributors that adhere to international standards such as ISO 13485 and FDA regulations. Factors such as material quality (typically surgical-grade stainless steel), design accuracy, ergonomic handling, and sterilization compatibility must be evaluated to ensure patient safety and surgical efficacy. Additionally, establishing relationships with certified medical device suppliers, comparing costs across vendors, and verifying proper documentation will help achieve a balance between affordability, availability, and performance. Ultimately, a well-informed sourcing strategy ensures that healthcare facilities are equipped with reliable, high-quality DeBakey clamps that meet clinical requirements and support optimal surgical outcomes.