The global copper citrate market is experiencing steady expansion, driven by rising demand across pharmaceutical, nutritional supplement, and agricultural sectors. According to Grand View Research, the global copper supplements market—of which copper citrate is a key component—was valued at USD 1.1 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 6.3% from 2023 to 2030. This growth is fueled by increasing awareness of micronutrient deficiencies, expanding applications in animal feed, and the compound’s superior bioavailability compared to other copper salts. As regulatory standards tighten and demand for high-purity, pharmaceutical-grade copper citrate rises, manufacturers with robust quality control, scalable production, and global compliance certifications are emerging as leaders in this niche yet critical segment of the mineral supplement industry. Below are the top 7 copper citrate manufacturers shaping this evolving landscape.
Top 7 Copper Citrate Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Copper Citrate
Domain Est. 1998
Website: wegochem.com
Key Highlights: Wego Chemical Group is an industrial distributor and supplier of Copper Citrate, providing supply chain, logistics and warehousing solutions across the ……
#2 Copper And Citrate Combined
Domain Est. 2005
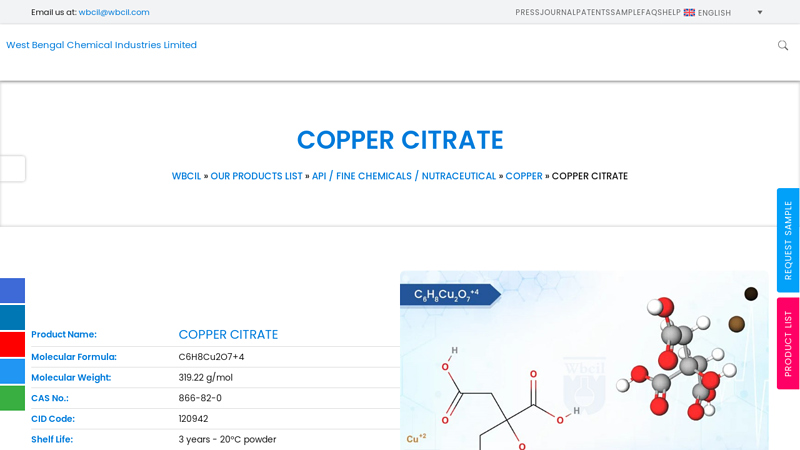
Website: wbcil.com
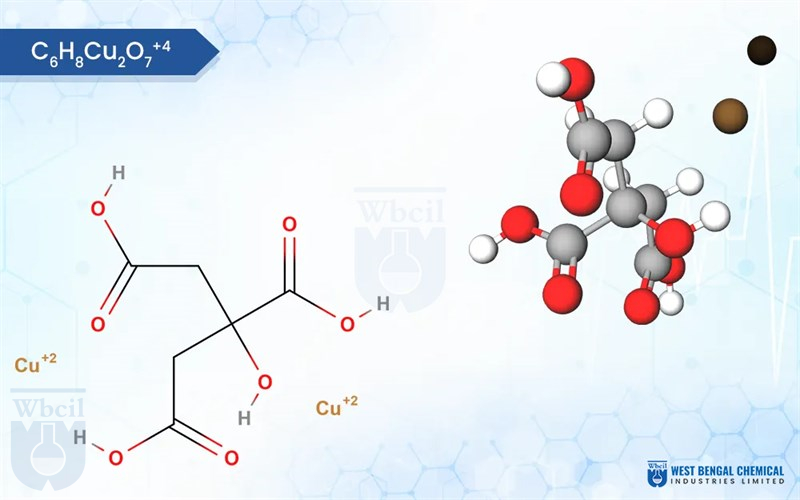
Key Highlights: Copper Citrate is a chemical compound that consists of copper ions (Cu²⁺) bonded to citrate ions (C₆H₅O₇³⁻). It is a copper salt of Citric Acid. Copper Citrate ……
#3 copper citrate
Domain Est. 2005
Website: manusaktteva.com
Key Highlights: Manus Aktteva Biopharma LLP is an ISO 9001:2015 Certified Global Supplier based in India for the product copper citrate, 10402-15-0….
#4 Copper citrate
Domain Est. 1996
Website: biosynth.com
Key Highlights: Copper citrate is a copper salt used as an antimicrobial agent in the food industry. It is often used in combination with citric acid and sodium citrate….
#5 Copper Citrate Hemitrihydrate Powder, CAS Number 866
Domain Est. 1997
Website: jostchemical.com
Key Highlights: Copper Citrate Hemitrihydrate Powder can be used as a dietary ingredient and as a nutrient. Copper aids in the absorption of iron, in the formation of red ……
#6 Copper Citrate (Kupzit) 1kg
Domain Est. 2000
Website: vigoltd.com
Key Highlights: In stockKupzit is a copper citrate preparation developed to remove sulphide off-flavours, especially hydrogen sulphide flavours in wine and cider….
#7 Copper (citrate) 60’s
Domain Est. 2021
Expert Sourcing Insights for Copper Citrate

H2: Projected Market Trends for Copper Citrate in 2026
By 2026, the global market for copper citrate is expected to experience steady growth, driven by increasing demand across key industries such as animal nutrition, pharmaceuticals, and agriculture. As a highly bioavailable form of copper, copper citrate is gaining preference over traditional copper supplements due to its enhanced absorption and lower environmental impact.
1. Rising Demand in Animal Feed:
The animal nutrition sector is anticipated to remain the largest consumer of copper citrate. Regulatory shifts in regions like the European Union, which are phasing out high levels of inorganic copper in livestock feed due to environmental concerns, are promoting the adoption of organic copper sources such as copper citrate. This transition supports improved animal health and reduced copper excretion, aligning with sustainability goals.
2. Growth in Pharmaceutical and Nutraceutical Applications:
Copper citrate is increasingly being used in dietary supplements and therapeutic formulations due to its role in supporting immune function, connective tissue formation, and antioxidant activity. With growing consumer awareness of micronutrient deficiencies and preventive healthcare, demand for copper citrate in fortified foods and supplements is expected to rise significantly by 2026.
3. Expansion in Agricultural Use:
In agriculture, copper citrate is being explored as a micronutrient fertilizer, especially in organic farming systems. Its solubility and plant availability make it effective in correcting copper deficiencies in soils, thereby improving crop yields. As precision farming and sustainable agriculture practices gain traction, the use of chelated minerals like copper citrate is projected to grow.
4. Regional Market Dynamics:
North America and Europe are expected to lead the market due to stringent regulations favoring organic trace minerals and advanced livestock management practices. Meanwhile, the Asia-Pacific region, particularly China and India, will witness rapid growth, driven by expanding livestock production, rising healthcare expenditure, and government initiatives to improve nutritional standards.
5. Supply Chain and Production Trends:
Manufacturers are investing in greener synthesis methods and scalable production technologies to meet rising demand while minimizing environmental impact. Additionally, strategic partnerships between chemical suppliers and feed/pharmaceutical companies are likely to increase, ensuring consistent supply and quality control.
6. Price and Competitive Landscape:
Prices for copper citrate may experience moderate upward pressure due to fluctuations in raw material costs (citric acid and copper salts) and increased demand. However, competitive pressures and technological advancements in production efficiency are expected to balance cost increases.
In conclusion, by 2026, the copper citrate market will be shaped by regulatory trends, sustainability demands, and health-focused applications. Companies that innovate in product formulation, expand into emerging markets, and emphasize environmental stewardship are likely to gain a competitive edge in this evolving landscape.
H2: Common Pitfalls in Sourcing Copper Citrate – Quality and Intellectual Property (IP) Concerns
Sourcing copper citrate, whether for pharmaceutical, nutritional, or industrial applications, presents several challenges related to product quality and intellectual property (IP) considerations. Being aware of these pitfalls is essential to ensure regulatory compliance, product efficacy, and legal safety.
1. Inconsistent Product Quality
- Variable Purity Levels: Suppliers may offer copper citrate with differing levels of purity. Impurities such as heavy metals (e.g., lead, arsenic, cadmium) or residual solvents can compromise safety, especially in human health applications.
- Hydration State Differences: Copper citrate can exist in various hydrated forms (e.g., trihydrate). Inconsistent hydration affects dosing accuracy and stability if not properly specified.
- Lack of Standardized Testing: Some suppliers may not provide comprehensive Certificates of Analysis (CoA) or may use non-standardized analytical methods, making it difficult to verify quality claims.
2. Mislabeling and Adulteration
- Incorrect Chemical Identification: Some suppliers may misrepresent basic copper carbonate or other copper salts as copper citrate. This can lead to formulation failures or regulatory non-compliance.
- Dilution with Fillers: Economic adulteration—such as diluting with inert materials—can occur, especially with less reputable suppliers.
3. Regulatory and Compliance Gaps
- Non-Compliance with Pharmacopeial Standards: For use in supplements or pharmaceuticals, copper citrate should comply with standards such as USP, Ph. Eur., or JP. Sourcing from suppliers who do not adhere to these standards increases regulatory risk.
- Inadequate Documentation: Missing or incomplete regulatory documentation (e.g., DMF filings, REACH/SFDA registration) can delay product approvals or market entry.
4. Intellectual Property (IP) Risks
- Patented Formulations or Processes: Some advanced forms of copper citrate (e.g., stabilized, nano-formulated, or chelated variants) may be protected by patents. Sourcing such materials without proper licensing can lead to IP infringement.
- Proprietary Manufacturing Methods: Even if the compound itself is not patented, the method of synthesis or purification might be. Using a supplier that employs patented processes can expose the buyer to indirect liability.
- Geographical IP Enforcement Variability: Suppliers in regions with weak IP enforcement may offer products that infringe on patents recognized in target markets (e.g., the U.S. or EU), creating legal exposure upon import or use.
5. Supply Chain Transparency Issues
- Opaque Sourcing: Lack of visibility into raw material origins (e.g., citric acid and copper source) can affect quality consistency and raise concerns about sustainability or ethical sourcing.
- Multiple Intermediaries: Relying on brokers or trading companies without direct access to the manufacturer increases the risk of receiving substandard or misrepresented materials.
6. Stability and Shelf Life Misrepresentation
- Some suppliers may not provide accurate data on degradation profiles or proper storage conditions, leading to compromised product performance over time.
Best Practices to Avoid Pitfalls:
– Conduct thorough supplier audits and request full CoAs with every batch.
– Verify compliance with relevant pharmacopeial or food-grade standards.
– Perform independent third-party testing upon receipt.
– Conduct freedom-to-operate (FTO) analyses when sourcing for commercial products to avoid IP conflicts.
– Establish direct relationships with manufacturers where possible to ensure traceability and accountability.
By proactively addressing these quality and IP-related pitfalls, organizations can mitigate risks and ensure the safe, compliant, and effective use of copper citrate in their applications.

H2: Logistics & Compliance Guide for Copper Citrate
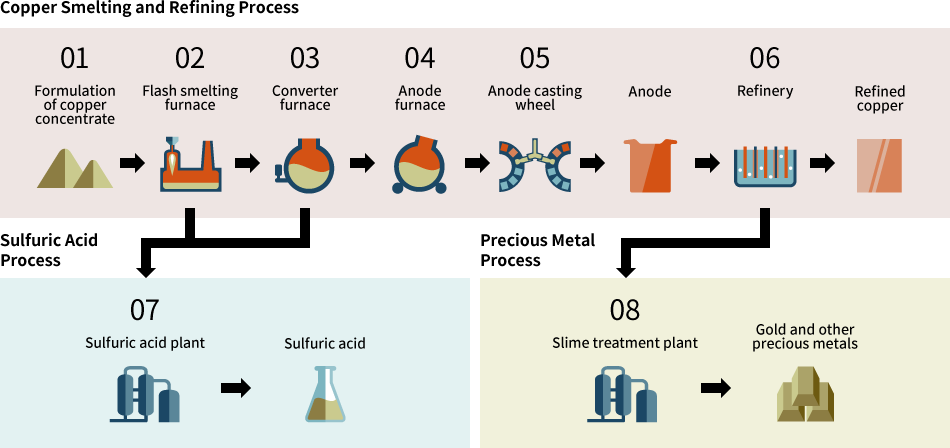
Copper Citrate is a coordination compound of copper and citric acid, commonly used in dietary supplements, agriculture, and industrial applications. Proper logistics and compliance handling are essential due to its chemical nature and regulatory requirements. Below is a comprehensive guide to ensure safe and compliant transportation, storage, and handling.
1. Chemical Identification
- Chemical Name: Copper(II) Citrate
- CAS Number: 11042-39-8
- Molecular Formula: C₆H₆CuO₇
- Appearance: Blue to bluish-green powder or crystalline solid
- Uses: Nutritional supplement, animal feed additive, fungicide, catalyst, and micronutrient in agriculture
2. Hazard Classification (GHS)
According to the Globally Harmonized System (GHS), Copper Citrate may be classified as:
– Acute Toxicity (Oral), Category 4 – Harmful if swallowed
– Skin Irritation, Category 2 – May cause skin irritation
– Serious Eye Damage, Category 2A – May cause eye irritation
– Specific Target Organ Toxicity (Single Exposure), Category 3 – May cause respiratory irritation
– Hazardous to the Aquatic Environment, Chronic, Category 2 – Harmful to aquatic life with long-lasting effects
Note: Classification may vary based on concentration and formulation. Always refer to the Safety Data Sheet (SDS) provided by the supplier.
3. Regulatory Compliance
International Regulations
- GHS/CLP: Must be labeled and classified in accordance with GHS standards (adopted by OSHA in the U.S. as HazCom 2012 and by EU as CLP Regulation).
- REACH (EU): Registered under REACH; ensure compliance with registration, evaluation, and restrictions.
- TSCA (USA): Listed on the TSCA Inventory; no significant restrictions for general industrial use.
Transportation Regulations
- IMDG Code (Sea): Not classified as dangerous goods for transport when in solid form and not meeting criteria for hazardous categories (typically Class 9 not applicable unless in solution).
- IATA (Air): Usually not regulated as dangerous goods under IATA DGR for solid forms in limited quantities. Confirm with SDS and packaging.
- 49 CFR (USA Ground): Generally not regulated as hazardous material when shipped in solid form under normal conditions.
Always confirm current classification with the latest SDS and consult carrier-specific requirements.
4. Packaging & Labeling
- Packaging: Use sealed, moisture-resistant containers (e.g., HDPE bags inside fiber drums or sealed plastic containers).
- Labeling Requirements:
- Product identifier
- Supplier information
- GHS pictograms (e.g., exclamation mark, environment)
- Signal word: “Warning” or “Danger” (based on concentration)
- Hazard and precautionary statements
- Net weight and batch number
5. Storage Requirements
- Conditions: Store in a cool, dry, well-ventilated area.
- Containers: Keep tightly closed when not in use.
- Compatibility: Store away from strong acids, bases, and oxidizing agents.
- Shelf Life: Typically 2–3 years when stored properly. Monitor for discoloration or caking.
6. Transportation Guidelines
- Mode of Transport: Suitable for road, rail, sea, and air when packaged correctly.
- Documentation: Include Safety Data Sheet (SDS), commercial invoice, and packing list.
- Temperature Control: No special temperature requirements, but avoid prolonged exposure to heat or moisture.
- Spill Kits: Recommended for bulk shipments.
7. Handling & Personal Protection
- Engineering Controls: Use local exhaust ventilation if dust is generated.
- PPE Requirements:
- Gloves (nitrile or neoprene)
- Safety goggles or face shield
- Dust mask (NIOSH-approved N95 or equivalent) if powder handling
- Lab coat or protective clothing
8. Environmental & Disposal Compliance
- Environmental Precautions: Prevent release into waterways or soil. Copper compounds are toxic to aquatic organisms.
- Spill Response: Collect spillage mechanically; avoid creating dust. Dispose of contaminated material as hazardous waste.
- Waste Disposal: Dispose in accordance with local, state, and federal regulations (e.g., RCRA in the U.S.). May require treatment as hazardous waste due to copper content.
9. Safety Data Sheet (SDS)
- Ensure an up-to-date SDS is available for each shipment.
- SDS Sections to Review:
- Section 2: Hazards Identification
- Section 7: Handling and Storage
- Section 8: Exposure Controls / PPE
- Section 13: Disposal Considerations
- Section 14: Transport Information
10. Import/Export Considerations
- Customs Codes (HS): Typically 2918.15 or 2833.30 (varies by country; confirm locally).
- Permits: May require registration or notification for agricultural or feed use (e.g., EPA, FDA, or EMA approvals depending on end-use).
- Restricted Countries: Some nations regulate copper compounds in fertilizers or supplements—verify import rules.
Conclusion:
Copper Citrate is generally low-hazard in solid form but requires careful attention to labeling, storage, and environmental protection due to its metal content. Compliance with GHS, transportation regulations, and local environmental laws is critical. Always consult the manufacturer’s SDS and regulatory authorities prior to shipping or industrial use.
Disclaimer: This guide is for informational purposes only. Regulatory requirements may vary by jurisdiction and formulation. Users must verify compliance with current laws and SDS specifications.
In conclusion, sourcing copper citrate requires careful consideration of several key factors to ensure quality, safety, and suitability for the intended application—whether in dietary supplements, agricultural formulations, or industrial processes. It is essential to partner with reputable suppliers that adhere to regulatory standards (such as FDA, USP, or FCC) and provide certificates of analysis for raw materials. Evaluating purity, solubility, and bioavailability is crucial, particularly for nutritional or pharmaceutical uses. Additionally, sustainability, ethical sourcing practices, and transparent supply chains are increasingly important in responsible procurement. By conducting thorough due diligence and maintaining consistent quality control, organizations can secure a reliable supply of copper citrate that meets their technical, regulatory, and environmental standards.