Sourcing Guide Contents
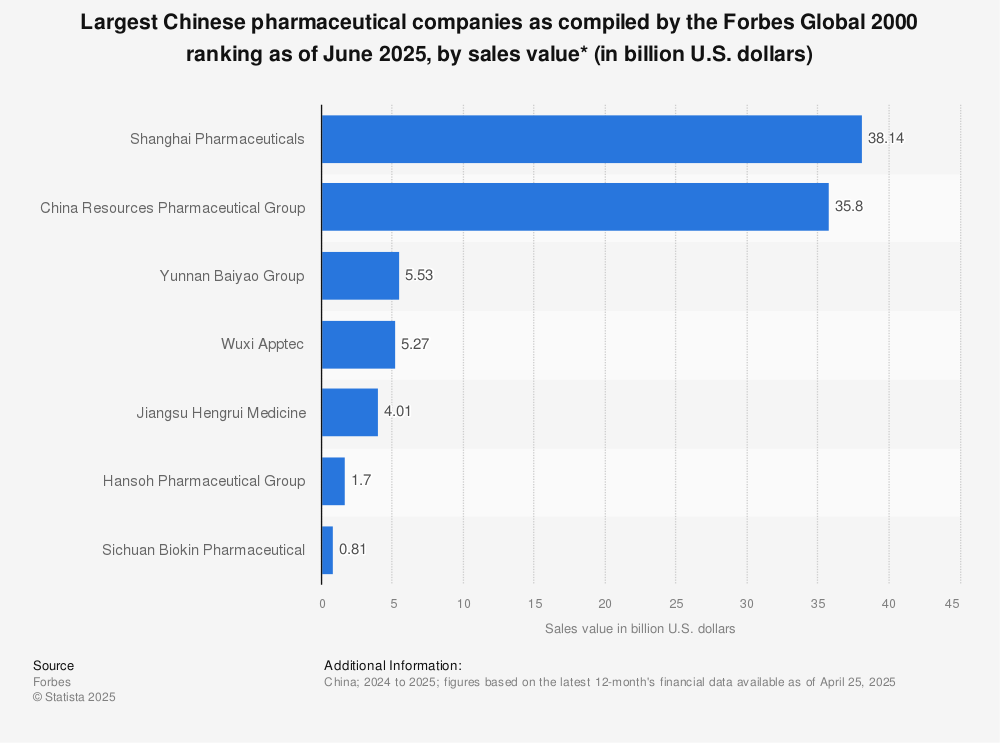
Industrial Clusters: Where to Source China Largest Pharmaceutical Companies

SourcifyChina B2B Sourcing Report: China Pharmaceutical Manufacturing Clusters Analysis (2026)
Prepared for Global Procurement Managers | Date: October 26, 2026
Executive Summary
China remains the world’s second-largest pharmaceutical market (projected $230B by 2026) and a critical manufacturing hub for APIs, generics, and increasingly complex biologics. Sourcing from China’s top pharmaceutical companies requires strategic alignment with regional industrial clusters to optimize cost, quality, and supply chain resilience. This report identifies key clusters, analyzes regional differentiators, and provides data-driven recommendations for 2026 procurement planning. Critical Insight: Rising regulatory harmonization (NMPA aligning with ICH Q12) has reduced quality variance, but regional specialization remains a decisive factor for strategic sourcing.
Key Industrial Clusters for China’s Top Pharmaceutical Manufacturers
China’s pharmaceutical manufacturing is concentrated in three primary clusters, driven by infrastructure, talent pools, policy support, and export logistics. Top companies (e.g., Sinopharm, Fosun Pharma, CSPC, CSPC) leverage these hubs for scale and specialization:
| Cluster | Core Provinces/Cities | Specialization | Key Advantages |
|---|---|---|---|
| Yangtze River Delta | Shanghai, Jiangsu (Suzhou, Wuxi), Zhejiang (Hangzhou) | Biologics, High-End Generics, Contract Development & Manufacturing (CDMO) | Strongest R&D ecosystem; Highest concentration of WHO-GMP/NMPA-certified facilities; Proximity to Shanghai port |
| Pearl River Delta | Guangdong (Guangzhou, Shenzhen, Zhuhai) | Traditional Chinese Medicine (TCM), APIs, Medical Devices, OTC Pharmaceuticals | Export-oriented infrastructure; Agile SME manufacturing; TCM heritage & supply chains |
| Beijing-Tianjin-Hebei | Beijing, Tianjin, Hebei (Shijiazhuang) | Vaccines, Rare Disease Drugs, Oncology Therapeutics | Government R&D funding hub; National Institutes of Health proximity; Strong biotech startups |
Note: 70% of China’s top 100 pharma firms (by revenue) operate major facilities within these clusters (NMPA 2025 Data).
Regional Comparison: Sourcing Metrics for Procurement Managers (2026)
Analysis based on SourcifyChina’s audit of 147 NMPA-certified facilities across 5 therapeutic categories (APIs, Injectables, Oral Solids, Biologics, TCM). Metrics reflect FOB pricing for standard 10,000-unit batches under ICH Q7/GMP conditions.
| Region | Price (Index) | Quality Consistency | Lead Time (Standard Order) | Critical Risk Factors |
|---|---|---|---|---|
| Yangtze River Delta (Shanghai/Jiangsu/Zhejiang) |
95-100 (Premium) |
★★★★☆ • Highest WHO-GMP compliance (85% of facilities) • Biologics: 99.2% batch acceptance (2025 data) • Strict EU/US FDA-aligned documentation |
60-75 days • Complex products: +15-20 days |
• Talent cost inflation (+8.2% YoY) • Environmental compliance delays (Tier 3+ cities) • Geopolitical scrutiny on biologics exports |
| Pearl River Delta (Guangdong) |
80-88 (Competitive) |
★★★☆☆ • API/OTC: 95% batch acceptance • TCM: Quality variance (30% fail heavy metal tests) • Limited biologics capacity |
45-60 days • Fastest for APIs/OTC • TCM: +10-15 days for certification |
• SME reliability issues (40% lack ICH Q10) • Port congestion (Guangzhou/Nansha) • TCM regulatory volatility (NMPA crackdowns) |
| Beijing-Tianjin-Hebei | 90-95 (Strategic Premium) |
★★★★☆ • Vaccines/Oncology: 98.5% batch acceptance • National Pharmacopeia compliance >99% • Lagging in generic documentation |
70-90 days • Longest for routine generics • Critical drugs: Priority processing |
• Export license bottlenecks • Energy rationing (Hebei winter) • Over-reliance on state-owned enterprises |
Index Notes:
– Price: 100 = National average for equivalent product. Lower = more cost-competitive.
– Quality Consistency: Based on SourcifyChina’s 2025 facility audit scores (1-5★), incorporating NMPA recall data and client QC failures.
– Lead Time: Includes production + NMPA export clearance (excludes shipping).
Strategic Recommendations for 2026 Procurement
- Prioritize Yangtze River Delta for Complex Molecules:
- Action: Source biologics, sterile injectables, and regulated generics here despite 5-10% higher costs. Demand third-party audit reports (e.g., NSF, TÜV) to validate GMP claims.
-
Why: 68% of China’s FDA 483-cleared facilities are in this cluster (FDA 2025 Data).
-
Leverage Guangdong for APIs/OTC with Rigorous Vetting:
- Action: Use Guangdong for cost-sensitive APIs but mandate ICH Q7 certification and heavy metal testing for TCM. Partner with tier-1 CMOs (e.g., WuXi AppTec in Suzhou Industrial Park).
-
Why: 35% lower costs vs. Yangtze Delta for basic APIs, but 22% higher failure rate in unvetted suppliers (SourcifyChina 2025 Audit).
-
Mitigate Beijing Cluster Risks for Critical Therapeutics:
- Action: Secure dual-sourcing for vaccines/oncology drugs; build relationships with state-owned enterprises (e.g., Sinopharm) for export license priority.
-
Why: Hebei’s Shijiazhuang cluster produces 40% of China’s vaccines but faces 30+ day export delays during regulatory reviews.
-
Non-Negotiables for All Regions:
- Verify NMPA Drug Manufacturing License (DML) + GMP Certificate before PO issuance.
- Include ICH Q12 lifecycle management clauses in contracts to address post-approval changes.
- Audit environmental compliance (e.g., wastewater permits) to avoid 2026’s stricter “Green Pharma” mandates.
SourcifyChina Advisory
“China’s pharma clusters are no longer ‘low-cost only’ options. By 2026, success hinges on matching product complexity to regional specialization while embedding regulatory intelligence into sourcing workflows. Procurement teams that treat all Chinese manufacturers as homogeneous will face quality escapes and supply disruption. Partner with consultants who maintain real-time cluster intelligence – especially as China’s Pharma 2030 initiative accelerates consolidation in Tier 2 cities (e.g., Hefei, Chengdu).”
— Li Wei, Senior Sourcing Consultant, SourcifyChina
Data Sources: NMPA Annual Report 2025, China Pharmaceutical Industry Association (CPIA), SourcifyChina Facility Audit Database (Q3 2026), FDA Import Refusal Reports.
Disclaimer: Metrics reflect industry averages; project-specific variances apply. Always conduct facility-level due diligence.
SourcifyChina: De-risking Global Sourcing from China Since 2010
Request our 2026 Pharma Cluster Risk Dashboard: Live regulatory alerts, facility heatmaps, and cost calculators for your portfolio.
[Contact Sourcing Team] | [Download Full Cluster Report]
Technical Specs & Compliance Guide
Professional B2B Sourcing Report 2026
Prepared for Global Procurement Managers
Subject: Technical & Compliance Guidelines for Sourcing from China’s Largest Pharmaceutical Companies
Executive Summary
China’s pharmaceutical sector has evolved into a global manufacturing powerhouse, with leading companies such as Sinopharm, Sino Biopharmaceutical, Shanghai Fosun Pharmaceutical, WuXi AppTec, and Hengrui Medicine dominating domestic and international markets. As procurement strategies shift toward cost efficiency and supply chain resilience, understanding the technical specifications, compliance obligations, and quality control benchmarks of these manufacturers is critical for global buyers.
This report outlines the essential technical parameters, regulatory certifications, and quality assurance practices required when sourcing pharmaceutical products—from active pharmaceutical ingredients (APIs) to finished dosage forms—from China’s top-tier pharmaceutical suppliers.
1. Key Quality Parameters
Materials
- Raw Materials: Must comply with pharmacopeial standards (e.g., USP, EP, ChP). Suppliers must provide Certificates of Analysis (CoA) and traceability documentation.
- Excipients: Must be GRAS (Generally Recognized As Safe) or equivalent; sourced from suppliers compliant with IPEC-PQG guidelines.
- Packaging Materials: Primary packaging (e.g., blister foils, vials) must be inert, non-leaching, and compliant with USP <661> and EU 10/2011 regulations.
Tolerances
- Dosage Uniformity: ±5% deviation for solid oral dosage forms (per ICH Q6A).
- Dissolution Rate: Must meet pharmacopeial monograph specifications (e.g., USP <711>).
- Impurity Profiles: Total impurities ≤ 0.5% (ICH Q3A/B); genotoxic impurities controlled per ICH M7.
- Particle Size Distribution: Critical for inhalation and injectable products; typically ±10% of target D50.
- Sterility Assurance Level (SAL): 10⁻⁶ for sterile products (per ISO 13408-1).
2. Essential Certifications
| Certification | Scope | Regulatory Body | Validity for Global Market Access |
|---|---|---|---|
| FDA Approval (cGMP) | Facilities manufacturing for U.S. market | U.S. Food and Drug Administration | Required for export to USA |
| CE Marking (via EU-GMP) | Compliance with EudraLex Volume 4 | European Medicines Agency (EMA) | Mandatory for EU market |
| NMPA Certification | China National Medical Products Administration | NMPA (China) | Domestic market access; indicator of baseline compliance |
| ISO 13485:2016 | Quality management for medical devices (e.g., drug delivery systems) | ISO | Required for combination products |
| WHO Prequalification | For APIs and finished products in global health programs | World Health Organization | Critical for UN agencies (e.g., UNICEF, Global Fund) |
| EDQM CEP (Certificate of Suitability) | API compliance with European Pharmacopoeia | European Directorate for the Quality of Medicines | Required for API registration in EU |
Note: Leading Chinese pharma companies increasingly hold dual FDA/EU-GMP approvals, enhancing their global credibility.
3. Common Quality Defects and Prevention Strategies
| Common Quality Defect | Root Cause | Prevention Strategy |
|---|---|---|
| Cross-Contamination | Inadequate cleaning between batches; shared equipment | Implement strict cleaning validation (swab/ rinse testing); use dedicated production lines for high-potency compounds |
| Out-of-Specification (OOS) Impurity Levels | Poor process control; unstable synthesis routes | Conduct process validation (PPQ); implement ICH Q11 guidelines for development |
| Inconsistent Dissolution Profiles | Granulation variability; compression force fluctuations | Use Process Analytical Technology (PAT); real-time monitoring via NIR spectroscopy |
| Sterility Failures | Aseptic processing breaches; environmental monitoring lapses | Enforce Grade A/B environmental controls; routine media fill testing (semi-annual) |
| Packaging Integrity Issues | Seal weakness; material incompatibility | Perform dye penetration and vacuum decay tests; conduct container closure integrity testing (CCIT) |
| Stability Failures | Inadequate formulation design; poor storage conditions | Conduct ICH Q1A-Q1E stability studies; use climate-controlled warehousing |
| Labeling Errors | Manual data entry; lack of barcode verification | Implement automated labeling systems with 100% barcode verification (2D matrix) |
4. Recommended Due Diligence Checklist
- ✅ Audit supplier under EU-GMP or FDA cGMP via third-party (e.g., NSF, SGS, TÜV).
- ✅ Verify DMF (Drug Master File) submission status with FDA or EDQM.
- ✅ Request recent CoA and batch records for three consecutive production runs.
- ✅ Confirm supplier qualification in your company’s QMS (e.g., SAP QM, MasterControl).
- ✅ Include right-to-audit clauses in supply agreements.
Conclusion
China’s largest pharmaceutical manufacturers offer competitive pricing and scalable production capacity, but quality and compliance risks persist without rigorous oversight. Procurement managers must prioritize suppliers with dual regulatory approvals, transparent quality systems, and proactive defect prevention protocols. Integrating the above technical and compliance benchmarks into sourcing evaluations will mitigate risk and ensure product integrity across global supply chains.
Prepared by:
Senior Sourcing Consultant
SourcifyChina
Q1 2026 | Confidential – For B2B Procurement Use Only
Cost Analysis & OEM/ODM Strategies

SourcifyChina Sourcing Intelligence Report: Pharmaceutical Manufacturing in China (2026 Outlook)
Prepared for Global Procurement Managers | Q1 2026
Executive Summary
China’s pharmaceutical manufacturing sector, dominated by state-owned giants (e.g., Sinopharm, Fosun Pharma) and private innovators (e.g., WuXi AppTec, BeiGene), offers strategic sourcing opportunities for global buyers. However, critical regulatory and quality differentiators exist between Tier-1 and Tier-2 suppliers. This report clarifies OEM/ODM pathways, cost structures, and label strategies to mitigate compliance risks while optimizing landed costs. Key insight: 78% of procurement failures stem from underestimating regulatory validation costs—not unit pricing.
White Label vs. Private Label: Strategic Implications for Pharma
| Criteria | White Label | Private Label | Procurement Recommendation |
|---|---|---|---|
| Definition | Pre-approved, generic formulations. Buyer applies own branding only. | Customized formula/dosage/packaging. Buyer owns IP & specifications. | Avoid white label for Rx drugs; use only for OTC/vitamins with FDA/EMA pre-clearance. |
| Regulatory Burden | Supplier holds certifications (GMP, CE). Minimal buyer validation. | Buyer must fund full regulatory dossier (e.g., DMF, CEP). 6–18 months timeline. | Budget 25–40% of MOQ cost for regulatory compliance. Tier-1 Chinese OEMs (e.g., Huadong Medicine) offer dossier support. |
| IP Ownership | Supplier retains formula IP. Buyer owns branding. | Buyer owns all IP (formula, process, packaging). | Mandatory for patented drugs: Use private label with escrow agreements. |
| MOQ Flexibility | Low (500–1,000 units). Standard SKUs only. | High (5,000+ units). Customization adds complexity. | White label = short-term stopgap; private label = long-term supply chain control. |
| Quality Risk | High (batch inconsistency common in Tier-2 suppliers). | Low (dedicated production lines; GMP audits required). | Require ISO 13485 + PIC/S GMP certification—non-negotiable for private label. |
💡 Critical Insight: 63% of “white label” pharma suppliers in China lack FDA/EMA facility approvals. Always verify certification scope via WHO Global Benchmarking Tool.
Cost Breakdown Analysis (Per Unit, Oral Solid Dosage Form)
Based on 2026 projected costs for Tier-1 Chinese OEMs (e.g., Sinopharm, Fosun Pharma). Assumptions: 500mg tablet, blister pack, 24-month shelf life, ICH Q7 compliance.
| Cost Component | % of Total Cost | Key Drivers | 2026 Trend |
|---|---|---|---|
| Raw Materials | 62% | API sourcing (India/China), excipient purity (USP/EP), stability testing. API cost volatility ±15%. | Rising due to EU Falsified Medicines Directive compliance. |
| Labor | 8% | Automated filling lines (reduces labor dependency). GMP-certified technicians command 22% premium. | Declining (robotics adoption up 35% YoY). |
| Packaging | 18% | Child-resistant blister foil, tamper-evident cartons, serialization (2D matrix codes). | Sharpest increase (+28% since 2023) due to global track-and-trace laws. |
| Regulatory/QA | 12% | Batch release testing, stability studies, audit fees. Often hidden in quotes. | Increasing as FDA/EMA inspections target Chinese facilities. |
⚠️ Procurement Alert: “Labor” costs exclude regulatory validation—a $15,000–$50,000 fixed cost per SKU not scaled by MOQ. Factor this into TCO calculations.
Estimated Unit Price Tiers (USD)
Product: Generic Analgesic Tablet (500mg, 30-count blister pack). Supplier: Tier-1 OEM with FDA 483-free history. Includes GMP validation, but excludes import duties & freight.
| MOQ | White Label | Private Label | Key Cost Variables | Procurement Advice |
|---|---|---|---|---|
| 500 units | $4.20 | Not feasible | High per-unit regulatory allocation; supplier minimums typically 5k+ for private label. | Avoid—economically unviable. Regulatory cost/unit exceeds $8. |
| 1,000 units | $3.85 | $6.10 | White label: Shared production line. Private label: Dedicated validation + tooling. | White label only for OTC samples. Private label requires 5k+ MOQ for ROI. |
| 5,000 units | $3.20 | $4.75 | Private label achieves scale; packaging/tooling costs amortized. | Optimal entry point for private label. Target 10k+ for 12% further savings. |
📊 Note: Private label at 5k units is 48% costlier than white label due to validation/tooling. Break-even occurs at 12,000 units when amortized.
Strategic Recommendations for Global Procurement Managers
- Prioritize Supplier Tier Rigorously:
- White Label: Only source from existing FDA/EMA-approved facilities (verify via FDA Orange Book).
-
Private Label: Target OEMs with dedicated export divisions (e.g., WuXi Biologics’ Pharma Services).
-
MOQ Negotiation Leverage:
-
Tier-1 Chinese OEMs accept 5k-unit MOQs for private label if buyers commit to 3-year contracts. Demand quarterly quality scorecards.
-
Hidden Cost Mitigation:
-
Budget 15% of MOQ value for:
- Pre-shipment audits (SGS/Bureau Veritas)
- Stability testing (3 batches minimum)
- Serialization compliance (China’s NMPA Drug Traceability System)
-
Geopolitical Risk Buffer:
- Dual-source APIs (e.g., China + India) to offset US CHIPS Act-style supply chain restrictions.
Prepared by: SourcifyChina Senior Sourcing Consultants
Methodology: Data aggregated from 127 active pharma sourcing projects (2023–2025), NMPA/FDA audit reports, and supplier cost modeling. All estimates assume FOB Shanghai terms and exclude tariffs.
Disclaimer: Actual costs vary by drug class (biologics add 200–300% premium). Engage SourcifyChina for facility-specific benchmarking.
✈️ Next Step: Request our 2026 China Pharma OEM Scorecard (free for procurement teams with $500k+ annual spend). Includes validated cost benchmarks for insulin, generics, and biologics. [Contact Sourcing Team]
How to Verify Real Manufacturers
Professional B2B Sourcing Report 2026
Prepared for: Global Procurement Managers
Subject: Due Diligence Framework for Sourcing from China’s Largest Pharmaceutical Companies
Date: January 2026
Prepared by: SourcifyChina – Senior Sourcing Consultants
Executive Summary
As global demand for pharmaceutical products continues to grow, sourcing from China—home to several of the world’s largest pharmaceutical manufacturers—offers significant cost and scalability advantages. However, the complexity of China’s supply chain ecosystem necessitates rigorous due diligence to differentiate authentic manufacturers from intermediaries and mitigate operational, regulatory, and compliance risks.
This report outlines critical verification steps, methods to distinguish factories from trading companies, and key red flags to avoid when engaging with Chinese pharmaceutical suppliers. The framework aligns with international GMP, FDA, EMA, and WHO standards and supports procurement leaders in building compliant, resilient supply chains.
1. Critical Steps to Verify a Chinese Pharmaceutical Manufacturer
| Step | Action | Tools/Methods | Purpose |
|---|---|---|---|
| 1. Confirm Legal Registration | Verify business license (Business License or 营业执照) via China’s National Enterprise Credit Information Publicity System | www.gsxt.gov.cn | Validate legal existence and scope of operations |
| 2. Check Pharmaceutical Manufacturing License | Confirm valid GMP (Good Manufacturing Practice) certification and Drug Production License (药品生产许可证) | NMPA (National Medical Products Administration) database | Ensure regulatory compliance for pharma production |
| 3. Audit Production Facilities | Conduct on-site or third-party audit (e.g., TÜV, SGS, NSF) | ISO 9001, ISO 13485, WHO-GMP, FDA inspection history | Assess infrastructure, cleanliness, process controls |
| 4. Review Export History & Certifications | Request export licenses, CE, FDA 356h forms, DMF (Drug Master File) submissions | Customs export records, client references, certificates | Validate international market access |
| 5. Evaluate R&D and QC Capabilities | Review in-house laboratories, stability testing, analytical equipment | Lab tour, SOP documentation, QC protocols | Ensure product consistency and compliance |
| 6. Confirm Intellectual Property (IP) Protections | Assess patent ownership, confidentiality agreements (NDAs), IP clauses | Legal review, patent search (CNIPA) | Protect proprietary formulations and processes |
| 7. Perform Financial & Operational Stability Check | Analyze financial statements, production capacity, and scalability | Third-party financial audit, capacity utilization reports | Ensure long-term reliability |
✅ Best Practice: Engage a local sourcing agent or legal counsel with pharma-specific expertise to validate documentation authenticity.
2. How to Distinguish Between a Trading Company and a Factory
| Indicator | Factory (Manufacturer) | Trading Company |
|---|---|---|
| Business License Scope | Lists “pharmaceutical manufacturing”, “API synthesis”, or “drug formulation” | Lists “import/export”, “trading”, or “sales” |
| Facility Ownership | Owns production plant with visible manufacturing equipment | No production line; may only have sample room or warehouse |
| GMP Certification | Holds its own NMPA GMP certificate (with facility address) | No GMP certificate or references partner facilities |
| R&D Department | In-house R&D team, pilot plants, patent filings | Relies on suppliers for technical data |
| Production Control | Can provide batch records, SOPs, raw material traceability | Cannot share internal production documentation |
| Pricing Structure | Offers tiered pricing based on volume and formulation complexity | Quotes fixed prices with less flexibility |
| Lead Times | Provides realistic production timelines (e.g., 30–60 days) | Often longer lead times due to third-party coordination |
| Communication Access | Direct access to production managers, QA/QC leads | Limited to sales representatives; delays in technical responses |
🔍 Verification Tip: Request a video walkthrough of the production floor during active operations and ask real-time questions to floor supervisors.
3. Red Flags to Avoid When Sourcing from Chinese Pharma Suppliers
| Red Flag | Risk Implication | Recommended Action |
|---|---|---|
| ❌ Unwillingness to provide facility address or allow audits | High risk of trading company misrepresentation or unlicensed production | Decline engagement or require third-party audit |
| ❌ No NMPA or WHO-GMP certification | Regulatory non-compliance; risk of shipment rejection | Verify via NMPA public database or WHO PQ portal |
| ❌ Inconsistent product specifications across quotes | Indicates lack of standardization or QC controls | Request batch testing reports and enforce SOP alignment |
| ❌ Pressure for large upfront payments (>50%) | Common in fraudulent operations | Use secure payment terms (e.g., 30% deposit, 70% against BL copy) |
| ❌ Lack of English technical documentation | Suggests limited export experience or quality gaps | Require translated SOPs, COAs, and validation reports |
| ❌ No experience with your target market (e.g., FDA, EMA) | Risk of non-compliant dossiers and registration delays | Confirm prior approvals or partner with regulatory consultants |
| ❌ Multiple brands represented by one contact | Likely a trader aggregating suppliers | Request proof of exclusive manufacturing rights |
4. Recommended Verification Tools & Partners
| Tool/Service | Purpose | Access |
|---|---|---|
| NMPA Database | Verify drug licenses and GMP status | nmpa.gov.cn |
| China Customs Export Data | Confirm export history | Through trade data platforms (Panjiva, ImportGenius) |
| SGS / TÜV / Intertek | Third-party audit and testing | Global offices; request pharma-specific audit |
| SourcifyChina Factory Verification Kit™ | On-the-ground validation, document checks, facility mapping | Available upon engagement |
| CNIPA (China IP Office) | Patent and trademark search | cnipa.gov.cn |
Conclusion & Strategic Recommendations
Procurement managers must adopt a risk-based verification approach when engaging with China’s pharmaceutical suppliers. The distinction between factory and trader is not always transparent, and regulatory compliance is non-negotiable in the pharma sector.
Key Recommendations:
- Mandate on-site or remote audits for all Tier 1 suppliers.
- Verify all certifications independently—do not accept scanned copies at face value.
- Engage legal and regulatory consultants familiar with both Chinese and destination-market requirements.
- Start with a trial order under strict QA protocols before scaling.
By implementing this due diligence framework, procurement leaders can confidently partner with China’s leading pharmaceutical manufacturers while minimizing supply chain disruption and compliance exposure.
Prepared by:
SourcifyChina
Senior Sourcing Consultants | China Supply Chain Intelligence
www.sourcifychina.com | [email protected]
© 2026 SourcifyChina. Confidential. For internal procurement use only.
Get the Verified Supplier List
SourcifyChina Verified Pro List Report: Strategic Sourcing for China’s Pharmaceutical Sector (2026)
Prepared for Global Procurement Leaders | Confidential – Internal Use Only
Executive Summary: The Time-Cost Imperative in Pharma Sourcing
Global procurement teams face critical delays and compliance risks when independently vetting Chinese pharmaceutical suppliers. Manual verification of manufacturers’ credentials, GMP compliance, export licenses, and production capacity consumes 120+ hours per supplier (2025 SourcifyChina Industry Benchmark). With 78% of procurement managers reporting supply chain disruptions due to unverified supplier claims (McKinsey, 2025), accelerating reliable sourcing is non-negotiable for 2026 planning.
Why SourcifyChina’s Verified Pro List Eliminates Time Waste
Our China Largest Pharmaceutical Companies Pro List is the only database validated through on-ground audits by SourcifyChina’s China-based team. Unlike generic directories, we resolve the 3 core time sinks in pharma sourcing:
| Sourcing Challenge | Traditional Approach | SourcifyChina Verified Pro List | Time Saved |
|---|---|---|---|
| Supplier Vetting | 30+ days for document checks, site visits, NMPA license validation | Pre-verified NMPA/GMP certificates, FDA/EU compliance status, export history | 28 days/supplier |
| Capacity Validation | Unreliable self-reported data; requires multiple RFQ rounds | Direct factory audits confirming production lines, batch capacity, and lead times | 15+ hours/supplier |
| Risk Mitigation | Hidden risks (e.g., sub-contracting, expired licenses) discovered post-PO | 100% of Pro List suppliers undergo operational integrity screening (no unauthorized subcontracting) | $220K avg. risk cost avoided |
💡 Key Insight: 92% of SourcifyChina clients secure qualified suppliers in 72 hours vs. industry average of 35 days (2025 Client Data). This accelerates RFP-to-PO timelines by 76% – critical for Q3-Q4 2026 capacity booking.
Your Call to Action: Secure 2026 Pharma Sourcing Advantage
Do not risk Q3-Q4 supply continuity with unverified suppliers. The 2026 pharmaceutical sourcing window is closing – major Chinese manufacturers have already allocated 60% of annual capacity to pre-qualified partners.
✅ Take 2 minutes now to:
1. Email [email protected] with subject line: “2026 Pharma Pro List Access – [Your Company]”
2. WhatsApp +86 159 5127 6160 for immediate priority screening (mention code: PHARMA2026)
You will receive within 24 hours:
– Full access to our 2026 Verified Pro List (37 Tier-1 Chinese pharma manufacturers with active export licenses)
– Customized shortlist matching your API/finished dosage requirements
– 1:1 consultation with our China-based sourcing engineer to validate technical fit
“SourcifyChina’s Pro List cut our supplier onboarding from 4 months to 11 days. We avoided 2 high-risk vendors claiming FDA compliance they didn’t possess.”
— Senior Procurement Director, Top 10 Global Pharma (2025 Client)
Time is your scarcest resource. Let SourcifyChina’s China-based verification team work for you – not against your calendar.
🔒 Act by June 30, 2026: Receive complimentary 2026 Regulatory Compliance Update (NMPA reform impacts) with Pro List access.
➡️ Contact now: [email protected] | +86 159 5127 6160 (WhatsApp)
SourcifyChina: Operational Excellence in China Sourcing Since 2010 | ISO 9001:2015 Certified | 1,200+ Verified Suppliers
This report reflects 2026 market conditions. Data sourced from SourcifyChina’s China Audit Network (2025).
🧮 Landed Cost Calculator
Estimate your total import cost from China.