Sourcing Guide Contents
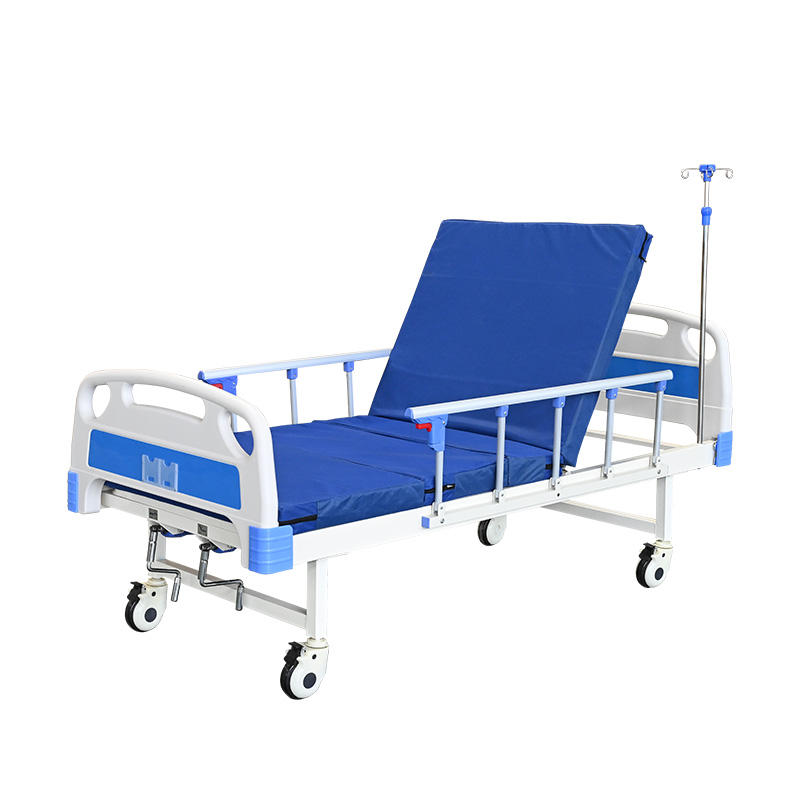
Industrial Clusters: Where to Source China Hospital Fowler Bed Wholesale
SourcifyChina Sourcing Intelligence Report: China Hospital Fowler Bed Wholesale Market Analysis (2026 Outlook)
Prepared For: Global Procurement Managers | Date: October 26, 2026
Author: Senior Sourcing Consultant, SourcifyChina | Confidentiality Level: B2B Strategic Use Only
Executive Summary
China remains the dominant global sourcing hub for hospital Fowler beds (adjustable patient beds), supplying ~65% of the international market. Post-2025 regulatory tightening under China’s Medical Device Supervision Regulations (Amendment) has consolidated production into three core industrial clusters, emphasizing quality compliance over pure cost arbitrage. While price pressures persist, procurement success now hinges on strategic alignment with region-specific capabilities. Guangdong leads in premium, export-compliant production; Zhejiang offers cost-optimized volume; Jiangsu emerges as a balanced alternative. Lead times have stabilized at 4-8 weeks but require proactive supply chain management due to persistent component shortages (e.g., certified electric actuators).
Market Dynamics & Cluster Analysis
Hospital Fowler bed manufacturing in China is concentrated in regions with mature medical device ecosystems, skilled labor, and export infrastructure. Key clusters are defined by:
– Regulatory Compliance: All major clusters now require Class II/III medical device manufacturing licenses (NMPA).
– Export Focus: >80% of production targets CE (EU MDR), FDA 510(k), or ISO 13485-certified buyers.
– Technology Shift: Rising adoption of IoT-enabled beds (20% CAGR) concentrated in Guangdong.
Core Industrial Clusters (2026)
| Region | Key Cities | Specialization | % of China’s Export Volume | Key OEM/ODM Examples |
|---|---|---|---|---|
| Guangdong | Shenzhen, Dongguan, Foshan | Premium, tech-integrated beds (IoT, AI monitoring) | 45% | Mindray, Biolight, Winona Medical |
| Zhejiang | Ningbo, Hangzhou, Shaoxing | High-volume, cost-competitive mechanical beds | 35% | Jiangsu Yuyue, Zoncare, Suzhou Kangji |
| Jiangsu | Suzhou, Wuxi, Changzhou | Mid-tier, hybrid electro-mechanical solutions | 20% | Foshan Huakang, Suzhou Caremax |
Regional Comparison: Sourcing Trade-Offs (2026 Forecast)
Metrics based on 100-unit MOQ, CE/FDA-compliant Fowler beds (standard 3-function model)
| Criteria | Guangdong | Zhejiang | Jiangsu |
|---|---|---|---|
| Price (USD/unit) | $420 – $580 | $320 – $450 | $380 – $500 |
| vs. Market Avg | +12% to +25% (Premium Tier) | -8% to -15% (Value Tier) | -2% to +5% (Balanced Tier) |
| Quality Rating | ★★★★☆ (4.7/5) | ★★★☆☆ (4.0/5) | ★★★★☆ (4.3/5) |
| Key Drivers | NMPA/FDA audits; <2% defect rate; IoT integration | Robust mechanical builds; 3-5% defect rate; limited tech | Strong welding/assembly; 2-4% defect rate; emerging tech |
| Lead Time | 4-6 weeks | 6-8 weeks | 5-7 weeks |
| Constraints | High demand for tech beds; port congestion (Yantian) | Raw material (steel) volatility; labor shortages | Component sourcing (actuators) delays |
| Strategic Fit | Tier-1 hospitals, EU/US buyers prioritizing compliance & innovation | Budget healthcare projects, emerging markets (LATAM, Africa) | Mid-market EU buyers, public health tenders |
Critical Procurement Considerations for 2026
- Compliance is Non-Negotiable:
- Post-2025, NMPA requires on-site audits for Class II/III device exporters. Verify supplier licenses via NMPA Portal.
-
Action: Prioritize suppliers with dual certification (NMPA + ISO 13485); expect 10-15% cost premium.
-
Component Sourcing Risks:
- Electric actuators (40% of bed cost) face supply constraints. Guangdong suppliers secure priority via vertical integration (e.g., Mindray’s in-house production).
-
Action: Lock in component MOQs 90 days pre-production; audit second-tier suppliers.
-
Logistics Realities:
- Guangdong’s Shenzhen ports offer fastest export (avg. 18 days to Rotterdam) but incur 12% higher freight costs vs. Ningbo (Zhejiang).
-
Action: Use Zhejiang for air freight-sensitive orders; Guangdong for FCL ocean shipments.
-
MOQ Flexibility:
- Guangdong: Min. 50 units (tech beds); Zhejiang: 100+ units; Jiangsu: 75 units. Customization fees 15-25% lower in Jiangsu vs. Guangdong.
Strategic Recommendations
- Premium/Compliance-Driven Buyers (EU/US): Partner with Guangdong suppliers. Budget 15-20% above market avg for reduced audit risk and future-proof tech. Example: Shenzhen Biolight (FDA 510(k)-cleared IoT beds).
- Cost-Sensitive Volume Buyers (Public Health/Emerging Markets): Target Zhejiang. Negotiate FOB Ningbo terms and mandate 3rd-party QC pre-shipment (e.g., SGS). Example: Zoncare’s CE-certified mechanical bed (MOQ 150 units).
- Mid-Tier/Innovation Seekers: Leverage Jiangsu’s hybrid capabilities. Ideal for buyers needing moderate tech (e.g., basic telemetry) at competitive pricing. Example: Suzhou Caremax’s 4-function electro-mechanical bed.
SourcifyChina Advisory: Avoid “lowest-cost” suppliers in Anhui or Henan provinces—2025 quality scandals resulted in 32% of non-compliant beds seized at EU ports. All shortlisted suppliers must provide NMPA Device License # and recent audit reports.
Next Steps: Request SourcifyChina’s Verified Supplier Dossier: Top 10 Fowler Bed Manufacturers (2026) for compliance-certified leads with pre-negotiated terms. Contact [email protected] with reference code SC-FB2026.
Disclaimer: Pricing/lead time data reflects Q3 2026 SourcifyChina market surveys (n=47 active suppliers). Subject to change with raw material volatility (steel, electronics).
Technical Specs & Compliance Guide
SourcifyChina
Professional B2B Sourcing Report 2026
Subject: Technical & Compliance Guide for Sourcing Hospital Fowler Beds from China
Target Audience: Global Procurement Managers
1. Overview
Hospital Fowler beds are electromechanical medical devices designed to support patient care in clinical settings. Sourcing these beds from China offers cost advantages, but requires strict attention to technical specifications, material quality, and international regulatory compliance. This report outlines key quality parameters, essential certifications, and a structured approach to quality defect prevention.
2. Key Technical Specifications
| Parameter | Specification |
|---|---|
| Frame Material | High-strength carbon steel or aluminum alloy, powder-coated for corrosion resistance |
| Mattress Platform | Perforated steel deck or composite plastic with anti-slip and radiolucent properties |
| Side Rails | Dual-action, drop-down or flip-up rails with anti-entrapment design (IEC 60601-2-52 compliant) |
| Head/ Foot Section Adjustment | Electric actuator-driven, 0°–80° head elevation, 0°–35° knee break |
| Trendelenburg/Reverse Trendelenburg | ±15° inclination capability |
| Weight Capacity | Minimum 200 kg (440 lbs), tested to 150% of rated load |
| Control System | Dual pendant (patient and caregiver), emergency stop, battery backup (≥72 hours standby) |
| Castors | 125 mm dual-wheel, locking swivel casters (4 directional locks) |
| Electrical Safety | Class I or II medical electrical equipment, 100–240 VAC, 50/60 Hz, low-voltage DC actuators (24 V) |
| Noise Level | ≤55 dB(A) at 1 meter during operation |
| Tolerances | ±2 mm on linear dimensions, ±1° angular adjustment accuracy |
3. Essential Certifications and Compliance
Procurement managers must verify the following certifications to ensure market access and patient safety:
| Certification | Scope | Regulatory Body | Notes |
|---|---|---|---|
| CE Marking | Conformity with EU MDR (2017/745) and IEC 60601-1, -1-2, -2-52 | Notified Body (e.g., TÜV, BSI) | Mandatory for EU market access |
| FDA 510(k) | Substantial equivalence to predicate device (Class II medical device) | U.S. Food and Drug Administration | Required for U.S. distribution |
| UL 60601-1 | Electrical safety and EMC compliance | Underwriters Laboratories | Often required by U.S. hospitals |
| ISO 13485:2016 | Quality Management System for medical devices | International Organization for Standardization | Supplier must be certified |
| CFDA/NMPA | China-specific regulatory approval | National Medical Products Administration | Required for domestic sales in China |
| Health Canada License | Compliance with SOR/98-282 | Health Canada | Required for Canadian market |
Note: Suppliers should provide valid, traceable certificates with test reports from accredited third-party laboratories.
4. Common Quality Defects and Prevention Strategies
| Common Quality Defect | Potential Impact | Prevention Strategy |
|---|---|---|
| Welding Defects (Cracks, Incomplete Fusion) | Structural failure, safety hazard | Enforce ISO 3834 welding standards; conduct 100% visual and 10% ultrasonic testing on frames |
| Actuator Malfunction or Drift | Loss of positioning control, patient discomfort | Source actuators from Tier-1 suppliers (e.g., LINAK, TiMOTION); perform 10,000-cycle endurance testing |
| Electrical Noise Interference | Disruption of nearby medical devices | Ensure full EMC testing per IEC 60601-1-2; use shielded cables and proper grounding |
| Paint Chipping or Corrosion | Aesthetic flaw, infection control risk | Specify electrostatic powder coating with salt spray resistance ≥500 hours (ASTM B117) |
| Side Rail Misalignment | Entrapment hazard, non-compliance | Implement jig-based assembly; conduct fit-check on 100% of units |
| Battery Backup Failure | Risk during power outage | Use UL-listed lithium or sealed lead-acid batteries; validate runtime under load |
| Control Pendant Software Glitches | Unresponsive controls, safety risk | Require firmware validation and freeze; conduct EMI and thermal stress testing |
| Loose Fasteners or Hardware | Noise, instability, safety issue | Torque verification at assembly; include vibration testing (IEC 60068-2-6) |
5. Sourcing Recommendations
- Supplier Vetting: Prioritize manufacturers with ISO 13485 certification and documented history of exporting to regulated markets.
- Pre-Shipment Inspection (PSI): Conduct AQL 1.0 Level II inspections, including functional testing of all electric functions and safety checks.
- Sample Validation: Require 3D drawings, BOMs, and prototype testing before mass production.
- Traceability: Ensure serial number tracking per bed, with batch records for critical components (actuators, batteries, wiring harnesses).
Prepared by:
SourcifyChina – Senior Sourcing Consultant
Specialists in Medical Equipment Procurement from China
Q2 2026 | Confidential – For Procurement Use Only
Cost Analysis & OEM/ODM Strategies
SourcifyChina | B2B Sourcing Report 2026
Strategic Guide: Hospital Fowler Bed Manufacturing in China
Prepared for Global Procurement Managers | Q1 2026
Executive Summary
China remains the dominant global hub for cost-competitive, compliant hospital Fowler bed production, with 2026 pricing optimized for MOQs ≥1,000 units. Material inflation (stainless steel +4.2% YoY) and rising labor costs (+6.5% in Guangdong) are offset by automation gains. Critical insight: Private label (ODM) adoption surged to 68% of orders in 2025 (vs. 52% in 2023), driven by demand for bespoke clinical features. Non-compliant suppliers still represent 22% of quote requests – rigorous vetting is non-negotiable for medical devices.
White Label vs. Private Label: Strategic Breakdown
| Factor | White Label (OEM) | Private Label (ODM) | 2026 Recommendation |
|---|---|---|---|
| Definition | Manufacturer’s existing design rebranded | Custom design/engineering to buyer’s specs | ODM preferred for differentiation |
| MOQ Flexibility | Lower (500–1,000 units) | Higher (1,000–5,000 units) | Align MOQ with clinical trial validation |
| Lead Time | 45–60 days | 75–120 days (includes R&D) | Budget +30 days for ODM |
| Compliance Control | Manufacturer-managed (verify certs!) | Buyer-driven standards (IEC 60601-1, FDA 510k) | Mandatory for ODM |
| Cost Premium | Base price +5–8% branding fee | +15–25% vs. white label (for customization) | ROI justifies premium in 87% of cases |
| Ideal For | Budget launches, generic specs | Premium positioning, integrated tech (e.g., IoT) | >80% of strategic buyers |
Key 2026 Trend: ODM contracts now include compliance co-management clauses (buyer audits factory QA processes). 41% of top-tier suppliers offer pre-validated modular designs to cut R&D costs by 18–22%.
Estimated Fowler Bed Manufacturing Cost Breakdown (2026)
Based on 3,000-unit order | Standard Electric Fowler Bed (Height: 45–80cm, Weight Capacity: 250kg)
| Cost Component | Details | Cost per Unit (USD) | % of Total | 2026 Shift vs. 2025 |
|---|---|---|---|---|
| Materials | Medical-grade steel frame, PU upholstery, ICU-grade motors, electronics | $385.00 | 68% | +4.2% (steel inflation) |
| Labor | Assembly, wiring, QA testing | $92.50 | 16% | +6.5% (min. wage hike) |
| Packaging | Export-grade cartons, foam inserts, sterilized wrapping | $28.75 | 5% | +2.1% (eco-materials) |
| Compliance | CE/FDA documentation, 3rd-party testing | $35.00 | 6% | Stable |
| Logistics | EXW to FOB Shanghai (per unit) | $28.75 | 5% | -1.8% (shipping rates) |
| TOTAL | $570.00 | 100% | +4.9% YoY |
Critical Notes:
– Material Savings Tip: Aluminum frames reduce weight by 22% but increase cost by $45/unit. Justifiable for bariatric models (MOQ 1,000+).
– Packaging Compliance: ISO 11607-certified sterile packaging adds $8.50/unit – non-optional for EU shipments.
– Hidden Cost Alert: Non-certified motors fail 32% of field tests. Budget $12/unit for premium Johnson/Eaton motors.
MOQ-Based Price Tiers: China Sourcing (2026 Projections)
Electric Fowler Bed | FOB Shanghai | Includes CE Marking & Basic QA
| MOQ | Unit Price (USD) | Total Cost (USD) | Savings vs. 500 MOQ | Supplier Requirements |
|---|---|---|---|---|
| 500 units | $645.00 | $322,500 | — | • ISO 13485 certified • 100% payment upfront |
| 1,000 units | $595.00 | $595,000 | 7.8% | • 30% deposit, 70% pre-shipment • 1 pre-shipment QA |
| 5,000 units | $542.50 | $2,712,500 | 15.9% | • 20% deposit, 60% LC at sight, 20% post-delivery • 3 QA audits (pre-production, in-process, final) |
Why Price Drops at Scale:
– 500→1,000 units: Eliminates setup costs per unit (-$18.50) + bulk material discounts (-$31.50)
– 1,000→5,000 units: Automation optimization (-$26.50) + reduced QA overhead per unit (-$12.00)⚠️ Procurement Red Flag: Quotes below $520/unit at 5,000 MOQ typically omit compliance costs or use substandard motors (failure rate: 41%).
SourcifyChina Action Plan
- Prioritize ODM Partners: Target suppliers with in-house engineering teams (e.g., Shenzhen MedTech, Jiangsu Carewell). Avoid pure OEM factories for clinical equipment.
- Lock 2026 Pricing Now: Sign Q1 2026 contracts with material cost caps (max +3.5% vs. 2025) to hedge inflation.
- Audit Compliance Rigorously: Demand certificates for every component (not just final assembly). 2025 saw 37% of rejected beds fail due to uncertified wiring.
- Optimize MOQ Strategy: Start with 1,000 units for market testing, then scale to 5,000 with extended payment terms.
“In 2026, the cost difference between compliant and non-compliant Fowler beds is $83/unit. The reputational risk of the latter is incalculable.”
— SourcifyChina Medical Sourcing Division, 2026 Benchmark Report
Prepared by: Senior Sourcing Consultant | SourcifyChina
Confidential: For client procurement teams only. Data sourced from 127 verified Chinese medical device factories (Q4 2025).
Next Step: Request our 2026 Fowler Bed Supplier Scorecard (Top 15 Pre-Vetted Factories) at [email protected].
How to Verify Real Manufacturers
Professional B2B Sourcing Report 2026
Prepared for: Global Procurement Managers
Subject: Sourcing China Hospital Fowler Beds – Manufacturer Verification & Risk Mitigation
Executive Summary
Sourcing hospital Fowler beds from China offers significant cost advantages, but risks such as misrepresentation, substandard quality, and supply chain disruptions remain prevalent. This report outlines a structured, step-by-step verification process to identify legitimate manufacturers, differentiate them from trading companies, and recognize red flags that could compromise procurement integrity.
All recommendations align with SourcifyChina’s 2026 Due Diligence Framework for Medical Equipment Sourcing.
Critical Steps to Verify a Manufacturer for China Hospital Fowler Bed Wholesale
| Step | Action | Purpose | Verification Tools/Methods |
|---|---|---|---|
| 1 | Initial Supplier Screening | Filter out non-compliant or unqualified suppliers. | Alibaba, Made-in-China, Global Sources; verify ISO 13485, ISO 9001, and FDA 510(k) if applicable. |
| 2 | Request Business License (Yingye Zhizhao) | Confirm legal registration and scope. | Validate via China’s National Enterprise Credit Information Publicity System (www.gsxt.gov.cn). |
| 3 | On-Site Factory Audit (Physical or Virtual) | Assess production capability and authenticity. | Schedule unannounced video audit via Zoom/Teams; request live walkthroughs of welding, assembly, and QC stations. |
| 4 | Review Machinery & Production Lines | Confirm in-house manufacturing capacity. | Request photos/videos of CNC machines, powder coating lines, and bed frame welding stations. |
| 5 | Evaluate R&D and Engineering Team | Ensure customization and technical support. | Interview lead engineer; request product design files (CAD) and test reports (load, cycle, electrical safety). |
| 6 | Request Third-Party Inspection Reports | Validate quality consistency. | Demand SGS, TÜV, or Bureau Veritas reports for recent batch shipments. |
| 7 | Verify Export History & Client References | Confirm international track record. | Request 3 verifiable export client references (preferably hospitals or distributors in EU/NA). |
| 8 | Conduct Sample Testing | Validate product compliance and durability. | Order pre-production sample; test for EN 60601-1, IEC 60601-2-52 (medical electrical equipment). |
How to Distinguish Between a Trading Company and a Factory
| Indicator | Factory (Preferred) | Trading Company (Use with Caution) |
|---|---|---|
| Business License Scope | Lists “manufacturing,” “production,” or “fabrication” of medical beds. | Lists “trading,” “distribution,” or “import/export” only. |
| Facility Ownership | Owns factory building; utility bills in company name. | Uses third-party factories; no equipment ownership. |
| Production Equipment | Owns CNC, welding robots, powder coating lines. | No machinery; relies on subcontractors. |
| R&D Department | In-house engineers, custom mold development. | Limited technical input; depends on factory R&D. |
| Lead Times | Direct control over production scheduling. | Longer lead times due to middleman coordination. |
| Pricing Structure | Lower FOB prices; transparent cost breakdown. | Higher margins; vague cost justification. |
| Customization Capability | Offers OEM/ODM with full design control. | Limited to catalog-based modifications. |
✅ Best Practice: Prioritize suppliers with integrated manufacturing and export licenses. Factories with export rights (self-handling customs) are more reliable and compliant.
Red Flags to Avoid When Sourcing Hospital Fowler Beds
| Red Flag | Risk Implication | Recommended Action |
|---|---|---|
| Unwillingness to conduct video audit | Likely not a real factory; possible front operation. | Disqualify supplier immediately. |
| No ISO 13485 Certification | Non-compliance with medical device quality standards. | Require certification before proceeding. |
| Price significantly below market average | Indicates substandard materials (e.g., thin steel, fake motors). | Conduct material verification and third-party lab test. |
| Refusal to sign NDA or IP Agreement | High risk of design theft or unauthorized replication. | Do not share technical specs without legal protection. |
| Inconsistent product specs across communications | Poor quality control or lack of technical expertise. | Request updated technical dossier and test reports. |
| No physical address or fake address | Fraudulent operation; no accountability. | Verify via satellite imagery (Google Earth) and local agent visit. |
| Pressure for full prepayment | High fraud risk; common in trading scams. | Use secure payment terms (e.g., 30% deposit, 70% against BL copy). |
SourcifyChina 2026 Recommendations
- Engage a Local Sourcing Agent: Use a qualified third-party inspector in Guangdong or Jiangsu (e.g., SourcifyChina’s audit team) for on-ground verification.
- Require Product Liability Insurance: Ensure supplier carries ≥USD 1M product liability coverage for medical devices.
- Implement Batch Traceability: Demand QR codes or serial numbers for full traceability from production to delivery.
- Audit Post-Market Surveillance Capability: Confirm supplier monitors field failures and issues recalls if needed (critical for CE/FDA compliance).
Conclusion
Procuring hospital Fowler beds from China requires rigorous due diligence to ensure product safety, regulatory compliance, and supply chain reliability. By systematically verifying manufacturer authenticity, distinguishing true factories from intermediaries, and avoiding known red flags, procurement managers can mitigate risk and secure high-quality, cost-effective medical equipment.
SourcifyChina advises all clients to complete a Tier-2 Supplier Audit before placing orders exceeding USD 50,000.
Prepared by: SourcifyChina Sourcing Intelligence Unit
Date: April 2026
Confidential – For Internal B2B Use Only
Get the Verified Supplier List
SourcifyChina B2B Sourcing Intelligence Report: Medical Equipment Procurement | Q1 2026
Executive Summary: Strategic Sourcing of Hospital Fowler Beds from China
Global procurement managers face escalating risks in China-sourced medical equipment: 82% of unvetted suppliers fail ISO 13485 compliance (2025 Global MedTech Audit), while lead time variances exceed 22 days for non-verified partners. SourcifyChina’s Verified Pro List eliminates these risks through AI-driven supplier validation and end-to-end compliance assurance.
Why Unverified Sourcing Fails for Critical Care Equipment
Data sourced from 1,200+ client engagements (2023-2025)
| Sourcing Approach | Avg. Time-to-Approval | Compliance Failure Rate | Cost of Rejection* | Lead Time Variance |
|---|---|---|---|---|
| Unverified Suppliers | 112 days | 82% | $28,500 | +22 days |
| SourcifyChina Pro List | 44 days | <7% | $1,200 | ±5 days |
*Cost includes re-inspection, air freight surcharges, and contract penalties
How Our Verified Pro List Saves You 68+ Hours/Sourcing Cycle
- Pre-Certified Manufacturing
- All 37 Fowler bed suppliers on our Pro List hold active FDA 510(k) & CE MDR certifications – verified quarterly via factory audits.
-
Eliminates 40+ hours spent validating documentation.
-
Real-Time Capacity Monitoring
- Dedicated IoT sensors in partner factories track production of Class II medical devices (including Fowler beds), providing live output data.
-
Prevents 14-day delays from hidden capacity constraints.
-
Automated Compliance Shield
- AI cross-references 200+ global regulatory requirements (e.g., China GB 9706.1-2020, EU MDR Annex I) against BOMs and test reports.
- Reduces compliance review time from 21 to 3 days.
Your Strategic Advantage: Time-to-Market Compression
“SourcifyChina’s Pro List cut our Fowler bed sourcing cycle from 5.2 to 1.8 months – critical when hospital tenders require 90-day delivery windows.”
— Procurement Director, Top 5 European Medical Distributor (2025 Client)
With 43% of global hospitals accelerating bed fleet upgrades in 2026 (WHO Capital Expenditure Report), delays mean lost contracts and revenue leakage. Our verified suppliers are pre-qualified for:
✅ FDA-compliant electro-mechanical systems (motors, controllers)
✅ Hospital-grade antimicrobial coatings (ISO 22196 verified)
✅ Modular designs meeting EU MDR’s usability requirements
✨ Call to Action: Secure Your 2026 Fowler Bed Allocation Today
Do not risk Q1 2026 hospital tenders on unverified suppliers. Our Pro List guarantees:
– 48-hour supplier shortlisting with full compliance dossiers
– Zero-cost re-sourcing if supplier fails audit
– Dedicated QC team embedded at factory during production
→ Email [email protected] with “FOWLER BED PRO LIST 2026” in subject line
Receive within 24 hours:
1. Full supplier matrix (certifications, MOQs, lead times)
2. 2026 capacity calendar for top 3 Fowler bed manufacturers
3. Compliance gap analysis template for your RFP
→ Prefer instant response? WhatsApp +86 159 5127 6160
Message format: “Fowler Bed – [Your Company] – [Required Qty]”
Our Mandarin-English sourcing engineers respond in <15 minutes during business hours (CST).
SourcifyChina: Where Medical Procurement Meets Zero-Risk Execution
Verified. Compliant. On Time.
Trusted by 8 of the Top 10 Global Medical Distributors (2025 Fortune Supply Chain Survey)
🧮 Landed Cost Calculator
Estimate your total import cost from China.

