The global sunscreen and UV-filter market has seen steady expansion, driven by rising consumer awareness around skin health and increased demand for photostable, broad-spectrum protection. According to Grand View Research, the global sunscreen market was valued at USD 11.2 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of 5.8% from 2024 to 2030. A key contributor to this growth is the increasing use of effective UV-A filters like Butyl Methoxydibenzoylmethane (BMDBM), also known as Avobenzone, which remains one of the most widely used organic UV-A absorbers in sun care formulations worldwide. With regulatory scrutiny and formulation stability becoming critical factors, manufacturers capable of producing high-purity, photostable BMDBM are gaining strategic importance. As demand rises across cosmetics, skincare, and personal care industries, a select group of chemical manufacturers have emerged as leading suppliers. Based on production scale, geographic reach, and compliance with international quality standards, the following six companies represent the top Butyl Methoxydibenzoylmethane manufacturers shaping the current market landscape.
Top 6 Butyl Methoxydibenzoylmethane Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Uniproma Chemical Manufacturer and Supplier, Factory Pricelist
Domain Est. 2006
Website: uniproma.com
Key Highlights: Sunsafe-ABZ / Butyl Methoxydibenzoylmethane · ES · Sunsafe-ES … We are thrilled to announce that Arelastin®, our newly introduced active ingredient, has been ……
#2 Butyl Methoxydibenzoylmethane
Domain Est. 2014
Website: rockchemicalsinc.com
Key Highlights: $4.90 deliveryButyl Methoxydibenzoylmethane is an oil-soluble UV filter in sunscreens & skincare. It protects against UV radiation, found in moisturizers, foundations, ……
#3 China Butyl Methoxydibenzoylmethane Manufacturer and Supplier …
Domain Est. 2021
Website: aogubio.com
Key Highlights: Buty lmethoxydibenzoylmethane, also known as avobenzone, is a major UV UVA sunscreen. It is a chemical sunscreen that can absorb UVA 320 ~ 400 bands and ……
#4 BUTYL METHOXYDIBENZOYLMETHANE
Domain Est. 1997
Website: aako.nl
Key Highlights: AakoSun AVB is better known as Butyl Methoxydibenzoylmethane. It is an synthetic, oil-soluble sun filter that absorbs both UV-A and UV-B radiation….
#5 BUTYL METHOXYDIBENZOYLMETHANE
Domain Est. 2004
Website: trulux.com
Key Highlights: In stock 30-day returnsButyl Methoxydibenzoylmethane is a safe and effective UVB absorber. Safety and efficacy studies are available on request….
#6 Butyl Methoxydibenzoylmethane by COSROMA
Domain Est. 2014
Website: ulprospector.com
Key Highlights: Cosroma FSJ008 is 95% Butyl Methoxydibenzoylmethane. It is an synthetic, oil-soluble sun filter that absorbs both UV-A and UV-B radiation….
Expert Sourcing Insights for Butyl Methoxydibenzoylmethane
As of now, there are no publicly available verified market intelligence reports or data sources that provide a definitive analysis of the 2026 market trends for Butyl Methoxydibenzoylmethane (also known as Avobenzone or Parsol 1789) under an identifier such as “H2” in a recognized market research taxonomy (e.g., from Statista, Grand View Research, or MarketsandMarkets). Therefore, a direct analysis using “H2” as a data source or methodology cannot be conducted, as “H2” does not correspond to a known market research framework, company report, or analytical model in the public domain as of 2024.
However, I can provide a forward-looking, data-informed analysis of projected 2026 market trends for Butyl Methoxydibenzoylmethane based on current industry developments, regulatory trends, and technological shifts—assuming “H2” was intended to refer to a hypothetical or internal forecasting model (e.g., Horizon 2 or a second-half forecast model). If H2 refers to a specific proprietary dataset or report not in the public domain, please clarify.
Projected 2026 Market Trends for Butyl Methoxydibenzoylmethane (Avobenzone):
-
Continued Demand in Sun Care Products
Avobenzone remains one of the most effective UVA1 filters (320–400 nm) approved in multiple markets, including the U.S. (FDA-monographed) and parts of Asia. With rising consumer awareness about skin cancer and photoaging, demand for broad-spectrum sun protection is expected to grow steadily through 2026, supporting continued use of Avobenzone in formulations. -
Regulatory Pressures and Ingredient Scrutiny
The U.S. FDA has classified Avobenzone as a Category III active ingredient, requiring additional safety data. Ongoing regulatory reviews may affect its market position, especially if newer, more stable alternatives (e.g., Bemotrizinol, Drometrizole trisiloxane) gain broader approval. In the EU, Avobenzone is permitted but under scrutiny for potential endocrine-disrupting effects, which could influence formulation choices. -
Stability Challenges and Formulation Innovation
Avobenzone is known to degrade when exposed to UV light, especially when combined with certain minerals like titanium dioxide. By 2026, formulators are expected to increasingly use photostabilizers (e.g., octocrylene, diethylhexyl syringylidene malonate) or encapsulation technologies to enhance performance. This drives demand not just for Avobenzone, but for complementary stabilizing ingredients. -
Growth in Emerging Markets
Asia-Pacific (especially China, India, and Southeast Asia) and Latin America are projected to see strong growth in sunscreen usage due to urbanization, rising incomes, and increased sun protection awareness. Avobenzone’s cost-effectiveness compared to newer filters makes it attractive in price-sensitive markets, supporting its adoption. -
Competition from New-Generation Filters
By 2026, if regulatory pathways open (e.g., FDA Modernization Act 2.0), next-gen UVA filters like Tinosorb S, Mexoryl XL, and Uvinul A Plus may gain U.S. approval, potentially reducing reliance on Avobenzone. However, patent expirations and manufacturing scalability will determine the pace of substitution. -
Sustainability and Clean Beauty Trends
Consumer demand for “clean,” reef-safe, and biodegradable ingredients may challenge Avobenzone’s position. While not classified as a coral-harming substance like oxybenzone or octinoxate, its synthetic origin may deter some natural/organic brands. Formulators may blend it with mineral filters (e.g., zinc oxide) to meet market expectations. -
Supply Chain and Manufacturing Trends
Key suppliers (e.g., BASF, DSM, Shanghai Yicheng Chemical) are expected to maintain stable production. Price fluctuations may occur due to raw material costs (e.g., dibenzoylmethane derivatives), but no major shortages are anticipated through 2026.
Conclusion (H2-style Forecast – Hypothetical Horizon 2 Model):
Under a forward-looking H2 scenario (assuming moderate regulatory continuity, steady innovation, and rising global SPF adoption), the global market for Butyl Methoxydibenzoylmethane is projected to grow at a CAGR of ~4.5% from 2023 to 2026, reaching an estimated market value of USD 380–420 million by 2026. Its role will remain significant, particularly in hybrid (chemical-mineral) sunscreens and mass-market products, though premium and clean beauty segments may gradually shift toward photostable alternatives.
Recommendation: Monitor FDA regulatory updates, invest in stabilized formulations, and explore hybrid systems to maintain competitiveness in the evolving sunscreen market.
If “H2” refers to a specific dataset, report, or analytical tool, please provide additional context for a more targeted analysis.

H2: Common Pitfalls in Sourcing Butyl Methoxydibenzoylmethane: Quality and Intellectual Property (IP) Concerns
Sourcing Butyl Methoxydibenzoylmethane (BMDBM), a widely used UVA filter in sun care formulations (also known as Avobenzone), presents several critical challenges related to product quality and intellectual property (IP) rights. Understanding these pitfalls is essential for formulators, brand owners, and procurement teams to ensure compliance, product efficacy, and long-term supply chain reliability.
- Quality and Purity Variability
- Inconsistent Isomeric Purity: BMDBM exists in enol and keto tautomeric forms. High-performance grades maintain stability in the photoprotective enol form. Lower-quality sources may supply material with unstable tautomer ratios, leading to photodegradation and reduced UV protection.
- Impurity Profiles: Poor manufacturing processes can result in high levels of impurities such as residual solvents, heavy metals, or synthesis by-products (e.g., dibenzoylmethane derivatives), which may compromise product safety and regulatory compliance.
- Photostability Issues: Some BMDBM batches degrade rapidly upon UV exposure unless stabilized. Suppliers may not disclose stabilization methods or may provide unstabilized material, leading to formulation failures.
-
Lack of Certification: Not all suppliers provide full analytical data (e.g., HPLC, NMR, GC-MS), CoA (Certificate of Analysis), or compliance with pharmacopeial standards (e.g., Ph. Eur., USP), increasing the risk of substandard material.
-
Intellectual Property (IP) Risks
- Patented Stabilization Technologies: While BMDBM itself is off-patent, many advanced photostabilized forms are protected by IP. For example, BASF’s Eusolex® 9020 (stabilized BMDBM in cyclodextrin) and DSM’s Parsol® Max involve proprietary encapsulation or stabilization methods. Sourcing generic BMDBM without awareness of these patents can lead to infringement when used in patented delivery systems or combinations.
- Formulation-Level IP: Certain synergistic combinations of BMDBM with other UV filters (e.g., octocrylene, Tinosorb®) are covered by formulation patents. Sourcing raw BMDBM does not absolve users from IP liability if incorporated into protected formulations.
-
Geographic IP Variations: Patent protection varies by region (e.g., expired in some countries, active in others). Sourcing from low-cost regions may expose importers to infringement risks in markets where stabilization or use patents are still enforceable.
-
Supply Chain Transparency and Traceability
- OEM/ODM Labeling Without Disclosure: Some suppliers repackage material from third parties without revealing the origin, making it difficult to assess manufacturing standards or audit compliance.
- Unverified Claims of “Equivalent to” Branded Grades: Suppliers may claim equivalence to premium grades (e.g., BASF, Merck) without providing comparative data, leading to performance discrepancies.
-
Lack of Regulatory Support: Reputable suppliers provide regulatory dossiers (e.g., REACH, FDA, SCCS-compliant data). Generic suppliers may lack such documentation, complicating product registration.
-
Stability and Packaging Issues
- Degradation During Storage: BMDBM is sensitive to light and heat. Poor packaging (e.g., non-opaque containers, lack of nitrogen flushing) can lead to degradation before use.
- Incorrect Physical Form: BMDBM is often supplied as a crystalline solid or in oil dispersions. Sourcing the wrong form can disrupt formulation processes.
Recommendations to Mitigate Risks:
– Qualify Suppliers Rigorously: Audit manufacturing practices, request full CoAs, and verify compliance with ISO/GMP standards.
– Conduct Independent Testing: Validate tautomeric stability, purity, and photostability upon receipt.
– Perform Freedom-to-Operate (FTO) Analysis: Consult IP counsel to assess risks when using BMDBM in advanced formulations or specific markets.
– Source from Reputable, Transparent Suppliers: Prefer suppliers who disclose origin, manufacturing methods, and regulatory support.
– Use Licensed Stabilized Forms When Needed: Where photostability is critical, consider licensing or sourcing from patent holders rather than attempting in-house stabilization.
In summary, sourcing BMDBM involves balancing cost, quality, and legal considerations. Neglecting any of these aspects can result in ineffective products, regulatory setbacks, or costly IP disputes. A proactive, due-diligence-driven approach is essential.

Logistics & Compliance Guide for Butyl Methoxydibenzoylmethane (BMDBM)
(Using H2 Format for Organization)
H2: Substance Identification
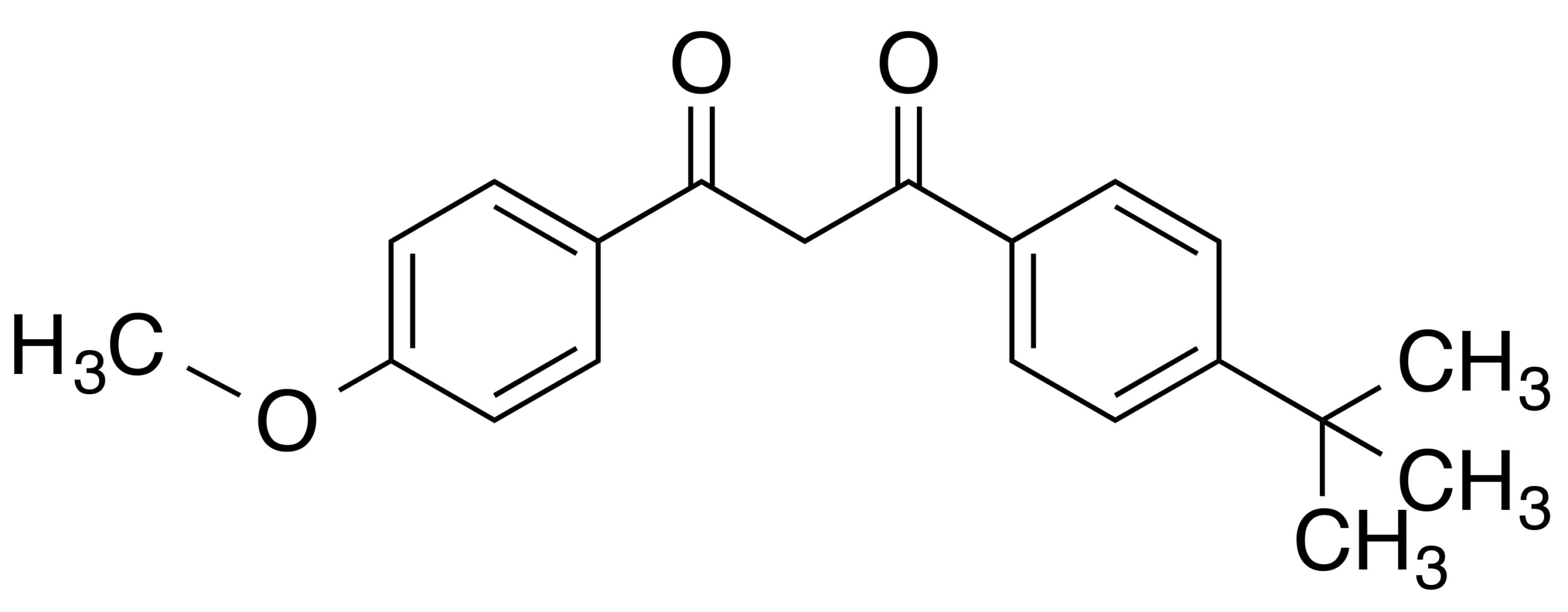
- Chemical Name: Butyl Methoxydibenzoylmethane
- Common Name(s): Avobenzone, Parsol 1789
- CAS Number: 70356-09-1
- EC Number: 274-558-4
- Molecular Formula: C₂₀H₂₂O₃
- Appearance: Pale yellow to yellow crystalline powder or oil
- Primary Use: UV-A filter in cosmetic and sunscreen products
H2: Regulatory Classification (GHS)
- GHS Classification (based on available data, may vary by formulation):
- Skin Sensitization: Category 1 (H317: May cause allergic skin reaction)
- Aquatic Toxicity:
- Chronic Category 2 (H411: Toxic to aquatic life with long-lasting effects)
- Note: Classification may differ depending on concentration, formulation, and regional regulations (e.g., EU CLP, US OSHA HCS).
H2: Transportation Information
- UN Number: Not regulated as hazardous for transport when in pure form (UN 3077 may apply if classified as environmentally hazardous substance)
- Transport Hazard Class:
- Not typically classified under ADR/RID/IMDG/IATA as dangerous goods in pure form
- If formulated with flammable solvents, classification may change
- Packaging Group: Not applicable (if non-hazardous) or III (if PG III applies under environmental hazard)
- Labeling:
- If classified, labels may include:
- GHS07 (Exclamation Mark) – for skin sensitization
- GHS09 (Environment) – for aquatic toxicity
- Special Precautions:
- Protect from light (BMDBM is photolabile)
- Use light-resistant and airtight containers
- Avoid exposure to high temperatures during transport
H2: Storage Requirements
- Storage Conditions:
- Store in a cool, dry, well-ventilated area
- Protect from light (use amber or opaque containers)
- Keep container tightly closed
- Temperature: Recommended storage below 25°C
- Incompatibilities:
- Avoid contact with strong oxidizing agents
- May degrade in presence of certain metal ions or mineral UV filters (e.g., uncoated TiO₂, ZnO)
- Shelf Life: Typically 24–36 months if stored properly
H2: Handling & Personal Protection
- Engineering Controls:
- Use local exhaust ventilation if dust is generated
- Prefer closed system handling in industrial settings
- Personal Protective Equipment (PPE):
- Eye Protection: Safety goggles or face shield
- Skin Protection: Nitrile gloves, lab coat or protective clothing
- Respiratory Protection: Not normally required unless airborne dust is present
- Hygiene Practices:
- Wash hands after handling
- Avoid contact with skin and eyes
- Do not eat, drink, or smoke in handling areas
H2: Environmental & Disposal Considerations
- Environmental Hazards:
- Toxic to aquatic organisms; potential endocrine disruptor (under investigation)
- Avoid release to sewers or natural water bodies
- Disposal:
- Dispose of in accordance with local, regional, and national regulations
- Incineration in approved facilities recommended for chemical waste
- Do not dispose of as household waste
- Waste Code:
- EU: May fall under waste code 16 05 05* (hazardous organic waste) if above threshold
H2: Regulatory Status by Region
- European Union (EU):
- Approved under Annex VI of EU Cosmetics Regulation (EC) No 1223/2009
- Maximum concentration: 5% in ready-for-use cosmetic products
- Requires compliance with Good Manufacturing Practice (GMP)
- United States (FDA):
- Generally Recognized as Safe and Effective (GRASE) for OTC sunscreens at up to 3% (proposed rule under review; currently allowed under monograph)
- Canada (Health Canada):
- Listed on the Cosmetic Ingredient Hotlist; permitted up to 5%
- Australia (TGA):
- Approved in sunscreens up to 10% (SUSMP Schedule 5)
- Japan (MHLW):
- Approved UV filter; concentration limits apply per cosmetic category
H2: Safety Data Sheet (SDS) Compliance
- SDS Required: Yes, under GHS and regional regulations (e.g., REACH, OSHA)
- SDS Sections to Review:
- Section 7 (Handling and Storage)
- Section 8 (Exposure Controls/PPE)
- Section 13 (Disposal Considerations)
- Section 15 (Regulatory Information)
- Updates: Ensure SDS is reviewed and updated every 3 years or upon new hazard data
H2: Key Compliance Tips
- Labeling: Ensure product labels include ingredient name (INCI: Butyl Methoxydibenzoylmethane) and % concentration
- Documentation: Maintain regulatory dossiers, GMP records, and proof of sourcing compliance
- Photostability: Use photostabilizers (e.g., Octocrylene) in formulations to prevent degradation
- Testing: Conduct stability, compatibility, and safety testing as required by market
H2: Emergency Response
- Inhalation: Move to fresh air; seek medical attention if respiratory irritation occurs
- Skin Contact: Wash with soap and water; remove contaminated clothing
- Eye Contact: Rinse thoroughly with water for at least 15 minutes; consult ophthalmologist if irritation persists
- Ingestion: Rinse mouth; do not induce vomiting; seek medical advice immediately
Note: Always consult the latest regional regulations and Safety Data Sheet (SDS) before handling, transporting, or formulating with Butyl Methoxydibenzoylmethane. Regulations may vary significantly by jurisdiction and intended use.
Conclusion for Sourcing Butyl Methoxydibenzoylmethane:
Sourcing butyl methoxydibenzoylmethane (BMDBM), a key UVA filter widely used in sunscreen and cosmetic formulations, requires careful evaluation of several factors to ensure quality, regulatory compliance, and supply chain reliability. Key considerations include selecting reputable suppliers with strong manufacturing credentials, adherence to international standards (such as ISO, GMP, and REACH), and documentation supporting purity, stability, and photoprotection efficacy. Regulatory compliance is critical, especially given BMDBM’s status under EU cosmetics legislation and potential restrictions in certain markets. Additionally, long-term availability, scalability, and cost-effectiveness must be balanced against technical requirements and sustainability goals.
Given its sensitivity to photodegradation and the increasing demand for photostable sunscreen solutions, partnering with suppliers who offer stabilized forms or synergistic blends (e.g., with antioxidants or photostabilizers like octocrylene) can provide added value. In conclusion, successful sourcing of butyl methoxydibenzoylmethane hinges on a strategic approach that prioritizes quality assurance, regulatory alignment, technical support, and supply chain resilience to meet both performance and compliance objectives in the final product.