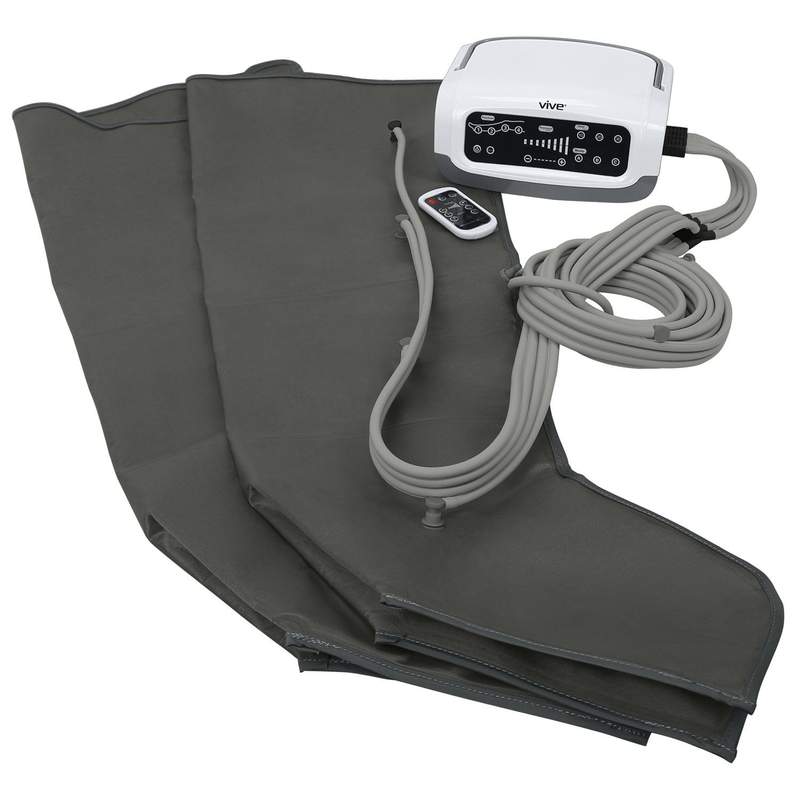
The global market for medical devices supporting circulatory health, including blood circulation pumps for legs, is experiencing robust growth, driven by rising prevalence of vascular disorders, increasing sedentary lifestyles, and an aging population. According to Grand View Research, the global intermittent pneumatic compression devices market—central to leg circulation therapy—was valued at USD 1.2 billion in 2023 and is expected to expand at a compound annual growth rate (CAGR) of 7.3% from 2024 to 2030. This growth trajectory is further fueled by expanding healthcare infrastructure, rising awareness of deep vein thrombosis (DVT) prevention, and increased adoption of home-based care solutions. As demand surges, manufacturers are innovating with smart features, enhanced portability, and clinical-grade performance. In this competitive landscape, nine manufacturers have emerged as leaders, combining regulatory compliance, technological advancement, and clinical efficacy to dominate the market for blood circulation pumps designed to improve lower limb perfusion and patient outcomes.
Top 9 Blood Circulation Pump For Legs Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
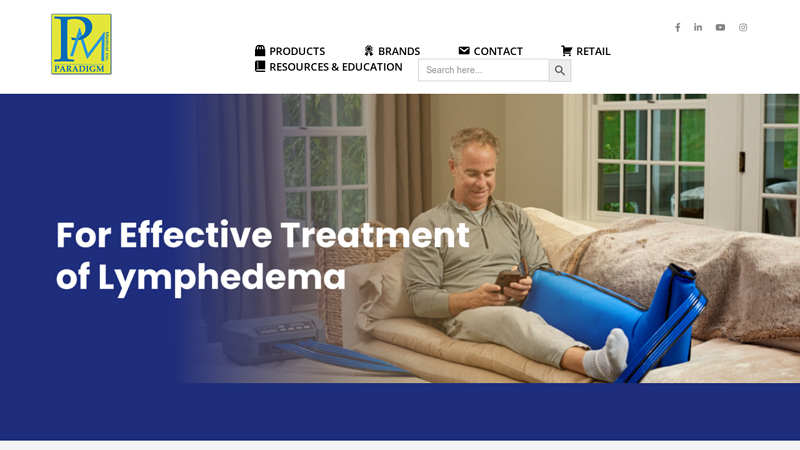
#1 Mastectomy
Domain Est. 1999
Website: paradigmmed.com
Key Highlights: Bio Compression Systems, Inc., is the leading manufacturers of pneumatic compression pump for the treatment of lymphedema, lipedema, venous disease and wound ……
#2 Normatec 3 Legs: Complete Leg Recovery Suite
Domain Est. 2007
Website: hyperice.com
Key Highlights: In stock Rating 5.0 (334) Normatec’s dynamic air compression system is a breakthrough in recovery technology. It enhances blood circulation, reduces muscle soreness, and speeds u…
#3 AV Impulse™ Foot Compression System
Domain Est. 1996
Website: cardinalhealth.com
Key Highlights: The A-V Impulse™ foot compression system helps reduce the incidence of deep vein thrombosis and pulmonary embolism….
#4 SC Pumps
Domain Est. 1996
Website: biocompression.com
Key Highlights: Bio Compression pneumatic compression devices (PCD) treat a variety of conditions. Our innovative design mimics the bodies’ own lymphatic system….
#5 ArtAssist®
Domain Est. 1997
Website: acimedical.com
Key Highlights: ArtAssist is a home-use medical treatment designed to increase blood flow without surgery so that wounds can heal and patients can keep their limbs….
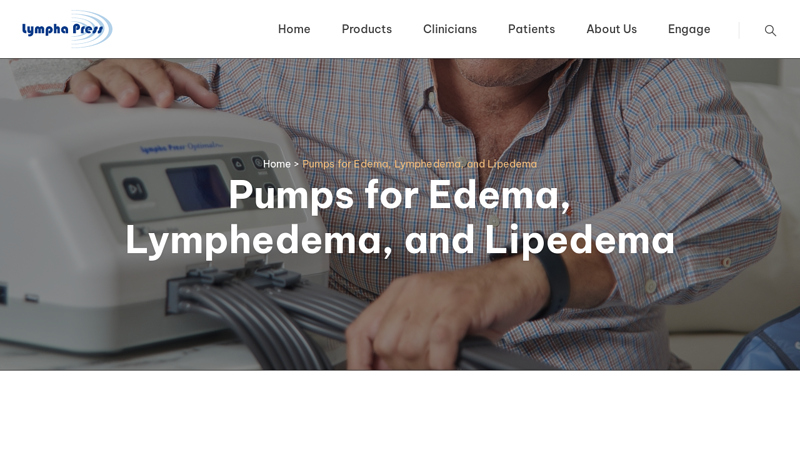
#6 Pumps for Edema, Lymphedema, and Lipedema
Domain Est. 2003
Website: lymphapress.com
Key Highlights: Leg pumps bring big benefits for people whose treatment includes moving fluids in the legs, stimulating lymph drainage, and relieving pain and discomfort….
#7 Vascular Compression Therapy
Domain Est. 2014
Website: zimmerbiomet.com
Key Highlights: Our Vascular Compression Therapy products are designed to reduce the risk of blood clots associated with deep vein thrombosis (DVT)….
#8 Compression Therapy Products
Domain Est. 2016
Website: airosmedical.com
Key Highlights: Our sequential compression therapy products, including lymphedema pumps and accessory garments reduce swelling and pain and increase mobility….
#9 Compression Pumps
Domain Est. 2021
Website: motivmedical.com
Key Highlights: Advanced Compression Therapy Systems for Enhanced Recovery. We provide advanced compression pumps designed to enhance blood flow and support recovery….
Expert Sourcing Insights for Blood Circulation Pump For Legs

H2: 2026 Market Trends for Blood Circulation Pumps for Legs
The global market for blood circulation pumps for legs is projected to experience significant growth and transformation by 2026, driven by demographic shifts, technological advancements, and increased awareness of circulatory health. These devices—commonly known as compression therapy devices or intermittent pneumatic compression (IPC) pumps—are designed to improve venous return, reduce swelling, and prevent deep vein thrombosis (DVT), particularly in individuals with limited mobility, chronic venous insufficiency, or post-surgical recovery needs.
1. Rising Prevalence of Chronic Diseases
A key driver shaping the 2026 market landscape is the growing incidence of diabetes, obesity, and cardiovascular diseases. These conditions often lead to poor peripheral circulation and increase the risk of venous ulcers and DVT. As global populations age—particularly in developed regions like North America and Europe—the demand for non-invasive circulation enhancement devices is expected to surge. The World Health Organization (WHO) estimates that by 2026, over 700 million people will be aged 65 or older, amplifying the need for home-based circulatory care solutions.
2. Technological Innovation and Smart Integration
By 2026, market trends indicate a shift toward smarter, more user-friendly blood circulation pumps. Devices are increasingly incorporating Bluetooth connectivity, mobile app integration, AI-driven pressure modulation, and real-time monitoring features. These advancements enable personalized therapy regimens, remote patient monitoring, and improved compliance. Wearable and portable designs are also gaining traction, allowing users to maintain mobility while receiving treatment—appealing to younger patients and active seniors.
3. Expansion of Home Healthcare
The post-pandemic emphasis on decentralized healthcare delivery continues to accelerate the adoption of home-use medical devices. Blood circulation pumps for legs are transitioning from hospital-centric models to consumer-focused, over-the-counter (OTC) products. By 2026, e-commerce platforms and direct-to-consumer marketing are expected to dominate distribution channels, supported by telehealth consultations and digital prescriptions.
4. Regulatory and Reimbursement Developments
Regulatory bodies such as the FDA and EMA are streamlining approval pathways for Class II medical devices like leg circulation pumps, encouraging innovation and market entry. Additionally, expanded insurance coverage and reimbursement policies in countries like the U.S. and Germany are reducing patient out-of-pocket costs, making these devices more accessible and driving market penetration.
5. Competitive Landscape and Market Consolidation
The market is witnessing increased competition from both established medical device companies (e.g., 3M, BSN Medical, and Tactile Medical) and emerging startups focused on digital health. Strategic partnerships, mergers, and acquisitions are expected to rise by 2026 as companies seek to integrate hardware with data analytics and expand into emerging markets such as India, China, and Latin America.
6. Focus on Preventive Care and Wellness
Beyond clinical use, blood circulation pumps are increasingly marketed for preventive wellness, targeting athletes, frequent travelers, and individuals with sedentary lifestyles. This trend reflects a broader shift toward proactive health management, further broadening the customer base.
In conclusion, the 2026 market for blood circulation pumps for legs will be defined by innovation, accessibility, and a growing emphasis on personalized and preventive care. With favorable demographic, technological, and regulatory tailwinds, the market is poised for robust expansion, offering significant opportunities for manufacturers, healthcare providers, and investors alike.

Common Pitfalls When Sourcing Blood Circulation Pumps for Legs (Quality and Intellectual Property)
Sourcing blood circulation pumps for legs—also known as intermittent pneumatic compression (IPC) devices—requires careful consideration of both product quality and intellectual property (IP) risks. While these devices offer significant therapeutic benefits for patients with circulatory issues, poor sourcing decisions can lead to substandard products, legal complications, and reputational damage. Below are key pitfalls to avoid in both quality assurance and IP protection.
Compromised Product Quality
One of the most significant risks when sourcing medical devices like leg circulation pumps is receiving products that do not meet safety or performance standards. Common quality-related pitfalls include:
- Lack of Regulatory Compliance: Many suppliers, especially in unregulated markets, may not adhere to FDA, CE, or ISO 13485 standards. Devices lacking proper certification can pose health risks and legal liabilities.
- Inferior Materials and Components: Low-cost manufacturers may use subpar materials in air bladders, tubing, or control units, leading to device failure, skin irritation, or inconsistent compression cycles.
- Inaccurate Pressure Delivery: Poorly calibrated pumps may deliver incorrect pressure levels, reducing therapeutic effectiveness or causing tissue damage.
- Absence of Clinical Validation: Some suppliers offer devices without clinical testing or published efficacy data, making it difficult to verify claims about circulation improvement or DVT prevention.
- Inadequate After-Sales Support: Limited access to technical support, repair services, or spare parts can disrupt patient care and increase long-term costs.
Intellectual Property Infringement Risks
Sourcing from overseas manufacturers increases the risk of inadvertently purchasing or distributing products that violate existing patents, trademarks, or design rights. Key IP pitfalls include:
- Selling Copycat Devices: Some suppliers offer products that closely mimic branded IPC devices (e.g., similar外观, functionality, or user interfaces), potentially infringing on patents held by established medical companies.
- Unverified IP Ownership: Suppliers may claim their product is “patent-free” without providing proof. This can expose buyers to litigation if the device incorporates patented technology.
- Lack of Freedom-to-Operate (FTO) Analysis: Failure to conduct an FTO search before sourcing can result in importing or selling devices that violate active patents in your target market.
- Trademark and Branding Confusion: Some devices may use logos or names confusingly similar to well-known brands, risking trademark infringement and consumer deception.
- No Licensing Agreements: If the device uses proprietary algorithms or software, using it without proper licensing may breach copyright or software patents.
Mitigation Strategies
To avoid these pitfalls:
– Work only with suppliers who provide regulatory certifications and quality management system documentation.
– Conduct third-party product testing and request clinical trial data.
– Perform thorough IP due diligence, including patent searches and legal consultations.
– Include IP indemnification clauses in sourcing contracts.
– Consider partnering with original equipment manufacturers (OEMs) that hold legitimate IP rights.
By addressing both quality and IP concerns proactively, businesses can ensure safe, compliant, and legally sound sourcing of blood circulation pumps for legs.

Logistics & Compliance Guide for Blood Circulation Pump For Legs
Product Classification and Regulatory Status
Blood circulation pumps for legs, commonly known as intermittent pneumatic compression (IPC) devices, are typically classified as medical devices. In the United States, these devices are regulated by the FDA and generally fall under Class II medical devices, requiring 510(k) premarket notification. In the European Union, they must comply with the Medical Device Regulation (MDR) 2017/745 and carry the CE mark. Accurate classification is essential to determine applicable regulatory pathways, technical documentation requirements, and conformity assessment procedures.
Regulatory Documentation and Approvals
Prior to distribution, manufacturers must obtain necessary regulatory approvals in target markets. This includes an FDA 510(k) clearance in the U.S., CE certification under MDR in the EU, and country-specific approvals such as Health Canada licenses or TGA registration in Australia. Required documentation typically includes technical files, risk management reports (ISO 14971), clinical evaluation reports (CER), labeling, and instructions for use (IFU). Maintaining updated regulatory files is critical for audit readiness and market access.
Labeling and Packaging Requirements
Labeling must comply with regional medical device directives and include essential information such as device name, model number, manufacturer details, UDI (Unique Device Identifier), CE or FDA marks, contraindications, warnings, and usage instructions. Packaging should ensure device protection during transit and meet environmental and shipping standards. Multilingual labeling may be required for international markets. All labels and IFUs must be validated for clarity and compliance with ISO 15223-1.
Import and Export Compliance
Exporting blood circulation pumps requires adherence to international trade regulations. Exporters must classify the product under the correct Harmonized System (HS) code—typically 9018.90 for therapeutic apparatus—and comply with destination country import controls. U.S. exporters must register with the FDA and submit export certifications if required. Importers must verify local registration, obtain import licenses, and ensure compliance with customs, tariffs, and product standards in the receiving country.
Shipping and Transportation Logistics
These devices should be shipped in secure, tamper-evident packaging that protects against shock, moisture, and temperature extremes. Temperature-controlled logistics may be necessary if accessories (e.g., gel pads or batteries) have specific storage requirements. Use of tracked and insured freight services is recommended. Documentation accompanying shipments must include commercial invoices, packing lists, certificates of origin, and regulatory compliance certificates to avoid customs delays.
Quality Management System (QMS) Compliance
Manufacturers and distributors must implement a Quality Management System compliant with ISO 13485. This ensures consistent design, production, and distribution processes that meet regulatory requirements. The QMS must cover supplier controls, corrective and preventive actions (CAPA), document control, and internal audits. Regular surveillance audits are necessary for maintaining certification and demonstrating regulatory compliance.
Post-Market Surveillance and Vigilance Reporting
After launch, manufacturers are responsible for monitoring device performance and reporting adverse events. In the EU, incidents must be reported through EUDAMED under MDR’s vigilance system. In the U.S., manufacturers must comply with FDA’s Medical Device Reporting (MDR) regulations (21 CFR Part 803). A robust post-market surveillance system, including customer feedback analysis and field safety corrective actions (FSCA), is essential for risk mitigation and regulatory compliance.
Environmental and Disposal Compliance
End-of-life disposal of electronic medical devices must follow environmental regulations such as the EU’s WEEE (Waste Electrical and Electronic Equipment) Directive and RoHS (Restriction of Hazardous Substances). Devices should be labeled for proper recycling, and manufacturers may be required to participate in take-back programs. Batteries, if included, must be handled according to local hazardous waste regulations.
Training and User Support Logistics
Effective logistics include providing user training materials and technical support. Distributors and healthcare providers should receive training on proper device use, maintenance, and troubleshooting. Support infrastructure—such as call centers, online portals, and service networks—must be in place to address user inquiries and facilitate repairs or replacements, ensuring continued compliance with customer support standards.
Summary and Best Practices
To ensure smooth logistics and compliance, stakeholders should: maintain up-to-date regulatory certifications, use certified shipping partners, implement ISO 13485-compliant processes, and establish strong post-market surveillance. Regular compliance reviews and staff training on international regulations will help prevent disruptions and ensure patient safety and market continuity.
Conclusion:
After evaluating various options for sourcing a blood circulation pump for the legs, it is evident that choosing the right device requires careful consideration of medical needs, product efficacy, safety certifications, user comfort, and cost-effectiveness. These devices, such as intermittent pneumatic compression (IPC) pumps and sequential compression devices, play a crucial role in enhancing peripheral circulation, reducing swelling, and preventing complications like deep vein thrombosis (DVT), especially in individuals with limited mobility or circulatory disorders.
Key factors in the sourcing decision include FDA or CE approval, ease of use, adjustability of pressure settings, portability, and compatibility with the user’s lifestyle. Reputable manufacturers and healthcare-recommended brands offer reliable performance and warranty support, which are essential for long-term use. Additionally, consulting with a healthcare professional ensures that the selected device aligns with the user’s specific medical condition and treatment plan.
In conclusion, investing in a high-quality, medically approved blood circulation pump for the legs can significantly improve vascular health and quality of life. By prioritizing safety, clinical effectiveness, and user needs during the sourcing process, individuals and caregivers can make an informed decision that supports optimal circulatory health.