The global bioelectrical impedance analysis (BIA) machine market is experiencing robust growth, driven by rising awareness of body composition monitoring in clinical, fitness, and home healthcare settings. According to a report by Grand View Research, the global BIA devices market size was valued at USD 2.1 billion in 2022 and is expected to expand at a compound annual growth rate (CAGR) of 9.3% from 2023 to 2030. This growth is fueled by increasing prevalence of obesity, metabolic disorders, and chronic diseases requiring precise nutritional and physiological assessments. Additionally, advancements in multi-frequency and segmental BIA technologies have enhanced measurement accuracy, broadening adoption across hospitals, wellness centers, and fitness facilities. As demand surges, a select group of manufacturers are leading innovation, scalability, and regulatory compliance. Based on market presence, technological capabilities, and product range, this report identifies the top 10 BIA machine manufacturers shaping the future of body composition analysis.
Top 10 Bioelectrical Impedance Analysis Bia Machine Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 seca Body Composition Analyzers
Domain Est. 1998
Website: seca.com
Key Highlights: Measure bioimpedance in seconds with our medical Body Composition Analyzers (seca mBCA) – fast, non-invasive, and cost-effective. Evaluation results with cloud- ……
#2 Bodystat Website
Domain Est. 1996
Website: bodystat.com
Key Highlights: Bioelectrical Impedance Analysis (BIA) is used in research as a quick, affordable, accurate and reproducible method to measure hydration, fat-free mass and ……
#3 Bioelectrical impedance analysis instruments
Domain Est. 1997
Website: pmc.ncbi.nlm.nih.gov
Key Highlights: Bioelectrical impedance analysis (BIA) is a widely used, noninvasive method for assessing body composition. Recent technological advances ……
#4 ImpediMed
Domain Est. 1999
Website: impedimed.com
Key Highlights: ImpediMed’s bioimpedance spectroscopy (BIS) devices accurately and noninvasively aid for early detection, assessment, and intervention in chronic diseases….
#5 Dedication of Bioelectric Impedance Analysis (BIA)
Domain Est. 2000
Website: charder.com
Key Highlights: Charder’s devices use Bioelectrical Impedance Analysis (BIA), for fast, simple, non-invasive, and easily repeatable analysis….
#6 Bioelectrical Impedance Analysis (BIA) and Body Composition …
Domain Est. 2002
Website: vitalscan.com
Key Highlights: A tool exists that, in less than 20 seconds, can determine fat mass, extracellular and intracellular water, and skeletal muscle mass….
#7 Bioelectrical Impedance Analysis
Domain Est. 2008
Website: withings.com
Key Highlights: Bioelectrical impedance analysis (BIA) measures body composition using low-voltage currents. Learn how BIA works, its accuracy, and its role in health….
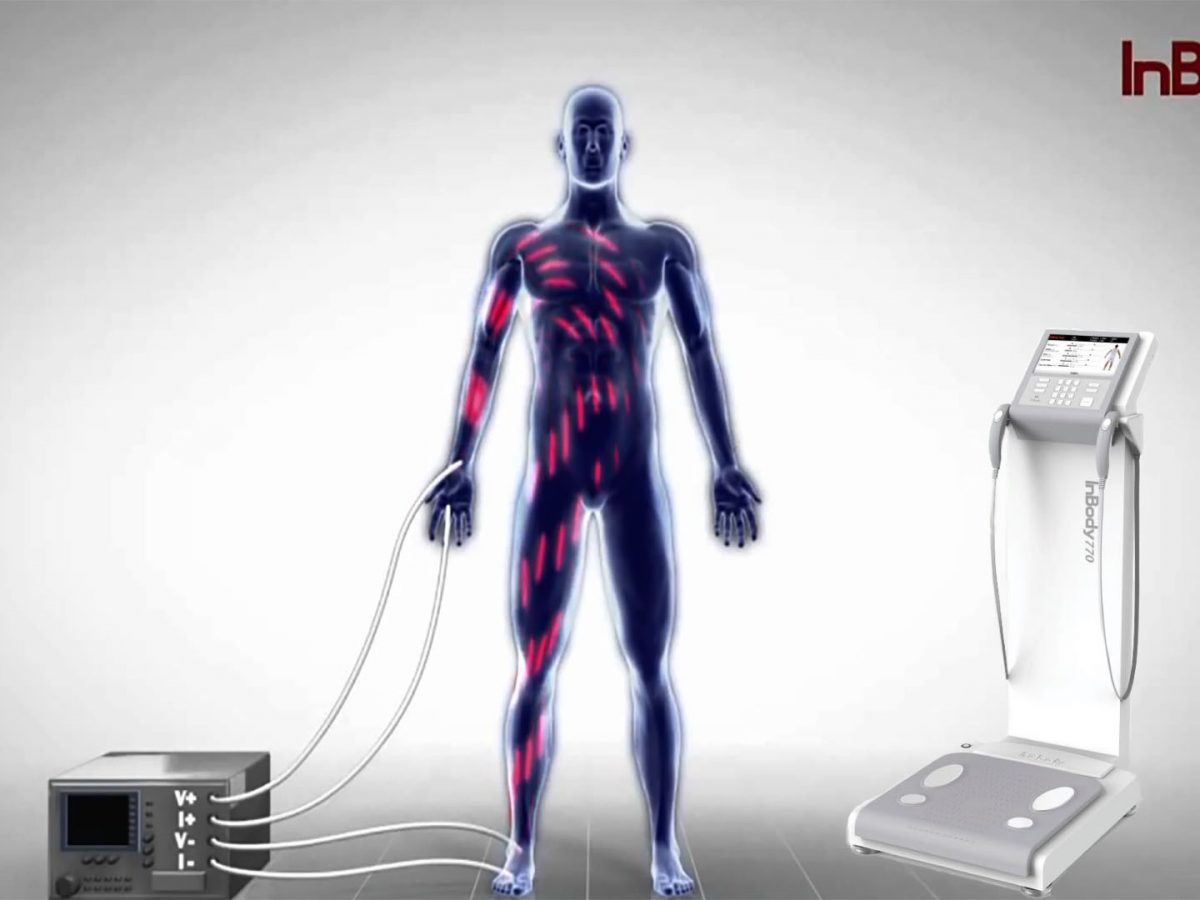
#8 InBody Canada
Domain Est. 2011
Website: inbodycanada.ca
Key Highlights: As the exclusive Canadian distributor of InBody, we sell body composition analyzers for health, fitness, and wellness providers and consumers….
#9 Bioelectrical Impedance Analysis
Website: fitness.marines.mil
Key Highlights: A year-long study to evaluate current body composition standards and ensure they optimize health, performance, and fitness….
#10 Bioelectrical Impedance Analysis
Website: tanita.eu
Key Highlights: Bioelectrical Impedance Analysis is a technique used for estimating body composition. All TANITA body composition monitors use advanced Bioelectrical Impedance ……
Expert Sourcing Insights for Bioelectrical Impedance Analysis Bia Machine

H2: Projected 2026 Market Trends for Bioelectrical Impedance Analysis (BIA) Machines
The global market for Bioelectrical Impedance Analysis (BIA) machines is poised for significant transformation by 2026, driven by technological advancements, rising health consciousness, and expanding applications across clinical and consumer sectors. Key trends shaping the BIA machine market in 2026 include:
-
Integration with Digital Health Platforms
By 2026, BIA devices are increasingly integrated with mobile health (mHealth) applications, electronic health records (EHRs), and wearable ecosystems. This connectivity enables real-time body composition monitoring, longitudinal health tracking, and personalized wellness insights. Cloud-based analytics and AI-driven interpretation of BIA data are becoming standard, allowing healthcare providers and consumers to make data-informed decisions. -
Growth in Home and Consumer Use
The demand for consumer-grade BIA machines—such as smart scales and handheld devices—is accelerating. With the global focus on preventive healthcare and fitness optimization, individuals are adopting BIA-enabled products for home use. Leading consumer electronics brands are incorporating BIA into fitness ecosystems, enhancing market penetration. -
Expansion in Clinical and Medical Applications
Clinically, BIA is gaining recognition for managing chronic diseases such as obesity, sarcopenia, renal failure, and heart failure. By 2026, medical-grade BIA machines are being adopted more widely in hospitals, dialysis centers, and geriatric care for nutritional assessment and fluid status monitoring. Regulatory approvals and clinical validation studies are supporting this expansion. -
Technological Advancements: Multi-Frequency and Segmental BIA
Multi-frequency BIA (MF-BIA) and segmental BIA technologies are becoming mainstream, offering greater accuracy in assessing intracellular and extracellular water, muscle mass distribution, and visceral fat. These advanced systems are preferred in both clinical and athletic performance settings, fueling demand for high-precision devices. -
Rising Prevalence of Obesity and Metabolic Disorders
The global rise in obesity, diabetes, and metabolic syndrome is a major driver for BIA adoption. BIA provides a non-invasive, cost-effective method for monitoring body fat, lean mass, and hydration—critical metrics in managing these conditions. Public health initiatives and corporate wellness programs are increasingly incorporating BIA assessments. -
Emerging Markets as Growth Engines
Asia-Pacific, Latin America, and parts of Africa are emerging as high-growth regions due to improving healthcare infrastructure, rising disposable incomes, and increasing awareness of body composition health. Local manufacturing and partnerships with global BIA providers are expanding market access in these regions. -
Focus on Standardization and Accuracy
As BIA use grows, there is increasing emphasis on standardizing measurement protocols and improving device accuracy. Regulatory bodies and industry consortia are working to establish guidelines, ensuring reliable and reproducible results across devices and populations. -
AI and Predictive Analytics Integration
By 2026, AI algorithms are being used to interpret BIA data in conjunction with other biometrics (e.g., blood pressure, activity levels) to predict health risks such as cardiovascular disease or metabolic decline. This enables proactive interventions and enhances the value proposition of BIA in both clinical and wellness settings.
In summary, the 2026 BIA machine market reflects a convergence of medical necessity, technological innovation, and consumer empowerment. With ongoing advancements in accuracy, connectivity, and application scope, BIA is transitioning from a niche diagnostic tool to a cornerstone of personalized health management.

H2: Common Pitfalls When Sourcing Bioelectrical Impedance Analysis (BIA) Machines – Quality and Intellectual Property Risks
Sourcing Bioelectrical Impedance Analysis (BIA) machines, especially from international suppliers or lesser-known manufacturers, can present several challenges. Two critical areas of concern are product quality and intellectual property (IP) risks. Failing to address these pitfalls can lead to regulatory non-compliance, reputational damage, and legal liabilities.
1. Compromised Quality Standards
One of the most prevalent issues when sourcing BIA machines is receiving devices that do not meet international medical or safety standards. Many low-cost suppliers, particularly in regions with lax regulatory oversight, may produce equipment that appears functional but lacks accuracy, reliability, or durability.
- Inaccurate Measurements: Poorly calibrated BIA machines can deliver inconsistent body composition readings, undermining their clinical or commercial usefulness.
- Substandard Materials: Use of low-grade electrodes, wiring, or casing materials can affect device performance and user safety.
- Lack of Certification: Some suppliers may falsely claim compliance with standards such as ISO 13485, CE marking, or FDA 510(k) clearance. Buyers must independently verify these certifications.
Without rigorous due diligence, businesses risk importing devices that fail performance benchmarks or are recalled after distribution.
2. Intellectual Property Infringement
Another significant risk involves sourcing BIA machines that infringe on existing patents, trademarks, or proprietary technology.
- Counterfeit or Clone Devices: Some manufacturers replicate branded BIA technology without authorization, copying circuit designs, software algorithms, or user interfaces. Distributing such devices can expose buyers to legal action from original equipment manufacturers (OEMs).
- Proprietary Algorithms: Many advanced BIA devices rely on patented algorithms to interpret impedance data and estimate body fat, muscle mass, etc. Unauthorized use of these algorithms—often embedded in firmware—can constitute IP theft.
- OEM Liability: If a sourced BIA machine uses protected technology, both the manufacturer and the importer may be held liable, especially in markets with strong IP enforcement like the U.S. or EU.
To mitigate these risks, buyers should request IP documentation, conduct patent landscape analyses, and include IP indemnification clauses in supply agreements.
Conclusion
When sourcing BIA machines, organizations must prioritize both quality assurance and IP compliance. Skipping supplier audits, third-party testing, or legal reviews may result in financial loss, product recalls, or litigation. Engaging reputable manufacturers, verifying certifications, and ensuring freedom to operate from an IP standpoint are essential steps in responsible procurement.
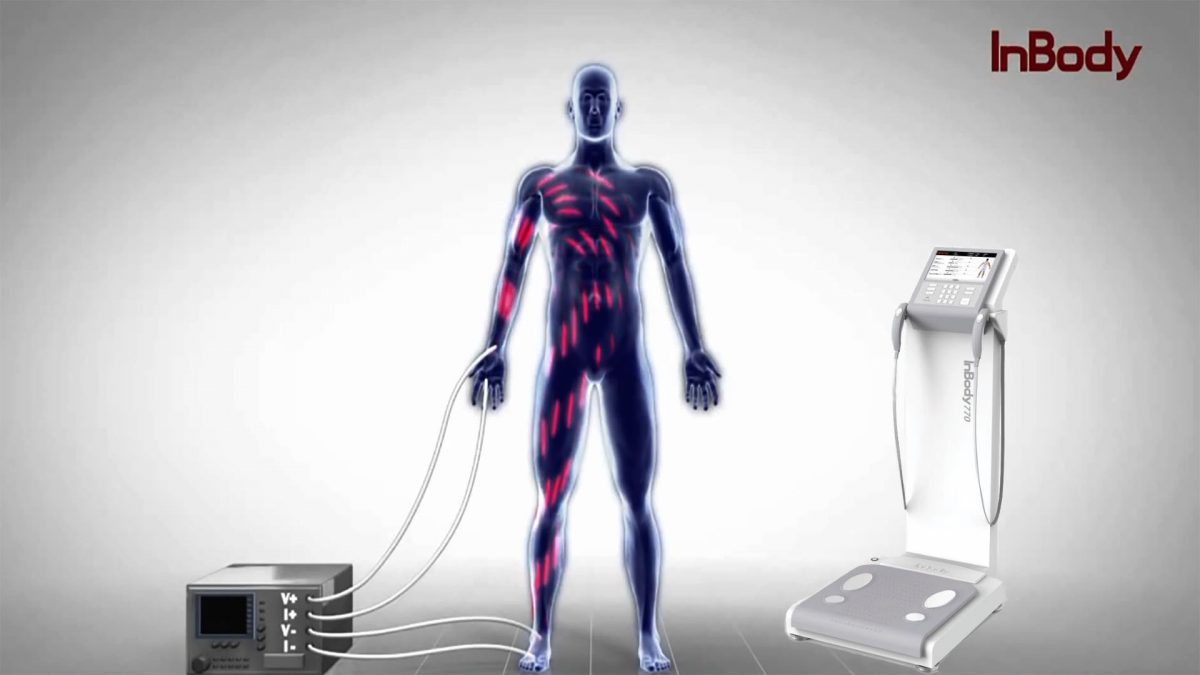
Logistics & Compliance Guide for Bioelectrical Impedance Analysis (BIA) Machines
Regulatory Classification and Approvals
Bioelectrical Impedance Analysis (BIA) machines are typically classified as medical devices and are subject to regulatory oversight in most jurisdictions. In the United States, the Food and Drug Administration (FDA) classifies most BIA devices as Class II medical devices under 21 CFR 880.2925 (Body Fat Measuring Devices). These devices require 510(k) premarket notification to demonstrate substantial equivalence to a legally marketed predicate device. In the European Union, BIA machines must comply with the Medical Devices Regulation (EU) 2017/745 (MDR) and carry the CE mark, usually falling under Class I or Class IIa depending on their intended use and functionality. Manufacturers must establish a Quality Management System (QMS) compliant with ISO 13485 and perform clinical evaluations as part of the conformity assessment.
Import and Export Requirements
Importing and exporting BIA machines requires compliance with international trade regulations and destination-specific medical device requirements. Exporters must ensure that devices meet the regulatory standards of the importing country, which may include local certification (e.g., Health Canada license, China NMPA registration, or ANVISA approval in Brazil). Proper documentation such as a Certificate of Free Sale, Certificate to Foreign Government (CFG), and a Declaration of Conformity (DoC) is often required. Additionally, Harmonized System (HS) codes must be correctly assigned—typically under 9018.90 for electrodiagnostic apparatus—to facilitate customs clearance and determine applicable tariffs. Shippers must also comply with International Air Transport Association (IATA) regulations if transporting devices containing batteries.
Labeling and Packaging Standards
BIA machines must be labeled in accordance with both regulatory and logistical requirements. Labels should include the device name, model number, manufacturer details, serial number, CE or FDA marks, and any applicable warning symbols per ISO 15223-1. Language requirements vary by market; for example, French labeling is mandatory in Canada and parts of Belgium. Packaging must protect the device during transit and comply with environmental regulations such as the EU Packaging and Packaging Waste Directive. Anti-static and shock-absorbent materials are recommended, especially for sensitive electronic components. Tamper-evident seals enhance security and regulatory compliance during distribution.
Shipping and Handling Protocols
Due to their electronic nature, BIA machines require careful handling during shipping. Devices should be powered off and securely packaged in original or equivalent protective materials to prevent damage from vibration, moisture, or temperature extremes. If the device contains rechargeable batteries, shipping must comply with UN 38.3 testing requirements for lithium batteries, especially when transported by air. Temperature-sensitive components should be monitored using data loggers if shipped in extreme climates. Carriers must be informed of the medical device nature of the shipment for appropriate handling and insurance coverage. Use of tracked and insured logistics services is strongly recommended.
Installation, Training, and Post-Market Compliance
Upon delivery, BIA machines should be installed by qualified personnel following manufacturer guidelines to ensure accuracy and safety. End users—such as clinics, fitness centers, or research institutions—require training on proper operation, hygiene protocols, and data interpretation. Manufacturers must maintain post-market surveillance systems to monitor device performance, report adverse events to regulatory bodies (e.g., FDA MAUDE database, EUDAMED), and issue field safety notices if necessary. Regular software updates and calibration verification contribute to ongoing compliance and data reliability. Records of distribution, servicing, and user training should be retained per regulatory requirements (typically 5–10 years).
Conclusion:
In conclusion, sourcing a Bioelectrical Impedance Analysis (BIA) machine requires a careful evaluation of accuracy, reliability, ease of use, and alignment with the specific needs of the intended application—whether clinical, fitness, research, or commercial. With a wide range of devices available, from portable handheld units to advanced medical-grade systems, it is crucial to select a BIA machine that offers validated measurement capabilities, consistent performance, and appropriate features such as segmental analysis, visceral fat assessment, and connectivity options. Additionally, considering factors like brand reputation, regulatory compliance (e.g., FDA or CE marking), after-sales support, and cost-effectiveness ensures long-term value and user satisfaction. Ultimately, investing in a high-quality BIA machine enhances body composition assessment precision, supports better health monitoring, and contributes to more informed decision-making in patient care, fitness programming, or research initiatives.