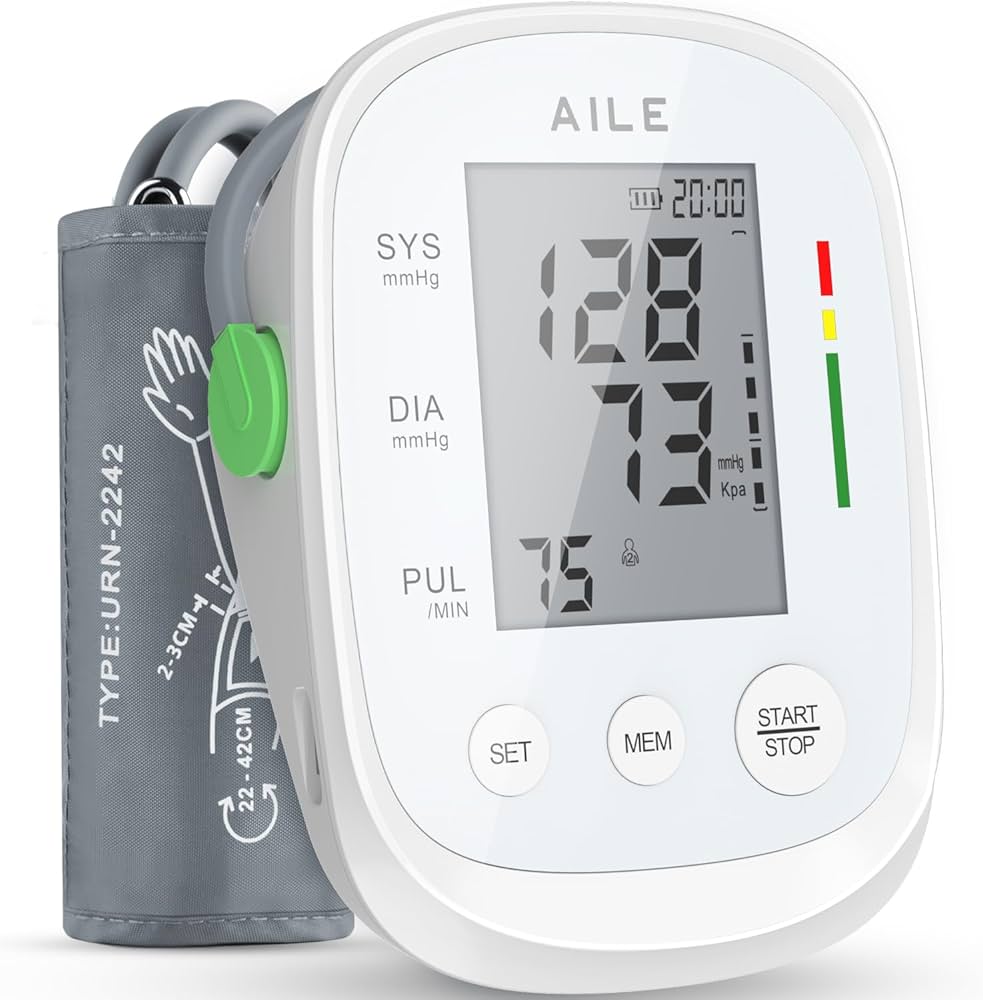
The global blood pressure monitoring devices market is experiencing robust growth, driven by rising hypertension prevalence, increasing awareness of cardiovascular health, and the growing trend toward home-based healthcare. According to Grand View Research, the global blood pressure monitoring devices market size was valued at USD 2.2 billion in 2023 and is projected to expand at a compound annual growth rate (CAGR) of 7.8% from 2024 to 2030. Similarly, Mordor Intelligence forecasts a CAGR of approximately 7.5% over the same period, citing technological advancements and expanding elderly populations as key drivers. Within this expanding landscape, big cuff blood pressure machines have become essential for accurate readings in patients with larger arm circumferences, particularly in clinical and homecare settings. As demand for reliable, standardized measurement tools rises, a select group of manufacturers has emerged at the forefront, combining precision engineering, clinical validation, and user-centric design. Here’s a data-informed look at the top 10 manufacturers leading innovation and market share in the big cuff blood pressure machine segment.
Top 10 Big Cuff Blood Pressure Machine Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Blood Pressure Monitoring
Domain Est. 1998
Website: microlifeusa.com
Key Highlights: Automatic Wrist Blood Pressure Monitor. $29.99 Add to cart. Blood Pressure … Extra Large Blood Pressure Arm Cuff. Quick View. Extra-Large Blood Pressure …Missing: big manufact…
#2 Blood Pressure Monitors & Sets
Domain Est. 1994
Website: mms.mckesson.com
Key Highlights: McKesson Medical-Surgical offers a vast selection of blood pressure products including blood pressure monitors, blood pressure cuffs, blood pressure units ……
#3 Blood Pressure Monitors
Domain Est. 1995
Website: edgepark.com
Key Highlights: A&D Medical Upper Arm Blood Pressure Monitor with Large Cuff. Each of 1 EA. Item # AEUA705VL….
#4 OMRON Easy
Domain Est. 1997
Website: omronhealthcare.com
Key Highlights: 30-day returnsOMRON Easy-Wrap ComFit blood pressure cuff inflates around the upper arm to fit both standard & large-sized arms from 9 to 17″ in circumference….
#5 Upper Arm Blood Pressure Monitors
Domain Est. 1998
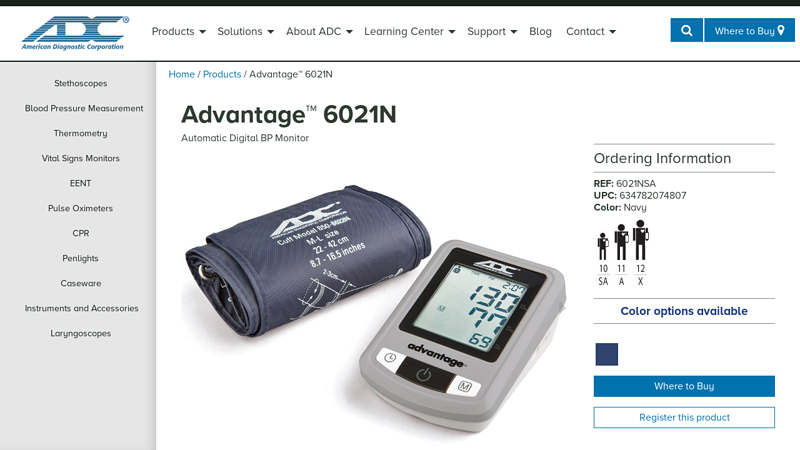
#6 Advantage™ 6021N
Domain Est. 1998
Website: adctoday.com
Key Highlights: The Advantage 6021N measures systolic, diastolic pressure, and pulse, and features a British Hypertension Society AA rating, ensuring unrivaled accuracy….
#7 Non
Domain Est. 1999
Website: gehealthcare.com
Key Highlights: GE HealthCare offers the most comprehensive blood pressure cuff portfolio for precise care, enabling care teams to choose the right cuff for the right ……
#8 Clinically
Domain Est. 2000
Website: pharma-smart.com
Key Highlights: Independently validated, peer reviewed blood pressure management · Weight measurement with BMI calculation · Blood Pressure cuff accommodating 97% adults · Left ……
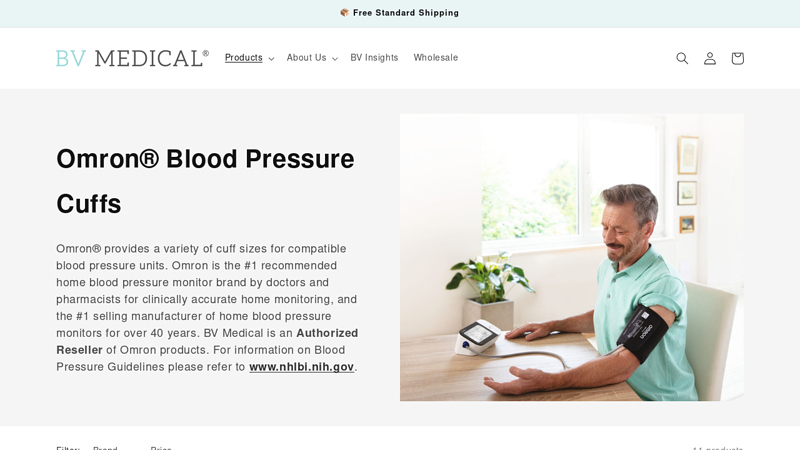
#9 Omron® Blood Pressure Cuffs
Domain Est. 2001
Website: bvmedical.com
Key Highlights: Free delivery 30-day returnsOmron provides a variety of cuff sizes for compatible blood pressure units. Omron is the #1 recommended home blood pressure monitor brand by doctors and…
#10 Shop Blood Pressure Cuffs
Domain Est. 2003
Website: mdfinstruments.com
Key Highlights: Free delivery over $125 Free 90-day returnsHigh quality Blood Pressure Cuffs from MDF Instruments®. Shop different blood pressure monitor cuff types and sizes here!…
Expert Sourcing Insights for Big Cuff Blood Pressure Machine

H2: 2026 Market Trends for Big Cuff Blood Pressure Machines
The global market for big cuff blood pressure machines is poised for significant evolution by 2026, shaped by demographic shifts, technological advancements, and growing awareness of cardiovascular health. These specialized devices, designed for patients with larger arm circumferences (typically over 32–42 cm), are increasingly recognized as essential tools in accurate blood pressure measurement, particularly among aging populations and those with obesity.
1. Rising Prevalence of Obesity and Hypertension
A key driver of market growth is the escalating global prevalence of obesity and hypertension. According to the World Health Organization (WHO), over 1.9 billion adults are overweight, with more than 650 million classified as obese. This demographic often requires larger cuffs for accurate readings, as standard cuffs can lead to significant measurement errors—potentially resulting in misdiagnosis or improper treatment. By 2026, increased screening initiatives and public health campaigns are expected to boost demand for big cuff devices in both clinical and home-use settings.
2. Expansion of Home Healthcare and Remote Monitoring
The shift toward decentralized healthcare—accelerated by the pandemic—will continue to fuel demand for user-friendly, accurate home blood pressure monitors with big cuffs. Integrated features such as Bluetooth connectivity, smartphone app synchronization, and cloud-based data tracking enable remote patient monitoring, supporting telehealth services. By 2026, manufacturers are expected to focus on developing smart big cuff devices that seamlessly integrate into digital health ecosystems.
3. Technological Innovation and Regulatory Emphasis on Accuracy
Regulatory bodies such as the U.S. FDA and the European Medicines Agency are placing greater emphasis on device accuracy and validation. The Association for the Advancement of Medical Instrumentation (AAMI) and the British Hypertension Society (BHS) have strict protocols for cuff size calibration. By 2026, manufacturers will likely invest in automated arm-size detection, adaptive inflation algorithms, and AI-powered analytics to ensure consistent accuracy across diverse patient populations.
4. Growth in Emerging Markets
Emerging economies in Asia-Pacific, Latin America, and parts of Africa are witnessing rising rates of lifestyle-related diseases and improved healthcare access. As public and private sectors expand cardiovascular screening programs, demand for properly sized blood pressure cuffs—including big cuffs—is expected to grow. Localized manufacturing and cost-effective models will be critical for market penetration.
5. Strategic Partnerships and Product Differentiation
By 2026, key players such as Omron, A&D Medical, Beurer, and Welch Allyn are likely to expand their product portfolios with dedicated big cuff lines. Strategic collaborations with healthcare providers, insurers, and digital health platforms will enhance distribution and reimbursement pathways. Additionally, ergonomic design, multilingual interfaces, and inclusivity in product testing will become competitive differentiators.
Conclusion
The 2026 market for big cuff blood pressure machines will be characterized by increased demand driven by public health needs, technological integration, and regulatory standards. As awareness grows about the risks of using improperly sized cuffs, healthcare providers and consumers alike will prioritize accuracy and inclusivity—positioning big cuff devices as a vital component of effective hypertension management.

Common Pitfalls When Sourcing Big Cuff Blood Pressure Machines (Quality & IP)
Sourcing big cuff blood pressure machines—especially for patients with larger arm circumferences—requires careful attention to quality and intellectual property (IP) concerns. Overlooking these aspects can lead to ineffective devices, regulatory non-compliance, or legal risks. Below are key pitfalls to avoid:
Inadequate Cuff Sizing and Calibration
One of the most frequent issues is sourcing devices with cuffs that aren’t truly “big” or properly calibrated for larger arms. Some suppliers may label standard cuffs as “large” without meeting medical definitions (e.g., arms >32 cm or 42 cm requiring thigh cuffs). Poor calibration on oversized cuffs leads to inaccurate readings, risking misdiagnosis.
Substandard Manufacturing and Materials
Low-cost manufacturers may use inferior materials in cuffs (e.g., non-durable bladders) or inaccurate pressure sensors. These machines often fail durability tests or deliver inconsistent results. Without proper certifications (like ISO 13485 or CE marking), there’s no assurance of quality control during production.
Lack of Clinical Validation
Many sourced devices—especially unbranded or white-label models—lack independent clinical validation against international standards (e.g., AAMI/ESH/ISO 81060-2). Using unvalidated machines can compromise patient safety and expose healthcare providers to liability.
Intellectual Property Infringement
OEMs or private labels may unknowingly source devices that copy patented technology from established brands (e.g., oscillometric algorithms, cuff inflation systems). This exposes buyers to IP litigation, product seizures, or forced recalls—especially in regulated markets like the EU or US.
Regulatory Non-Compliance
Big cuff devices must meet regional medical device regulations (e.g., FDA 510(k), EU MDR). Sourcing from manufacturers without proper regulatory documentation increases the risk of import bans or market withdrawal. Some suppliers falsely claim compliance without verifiable evidence.
Poor After-Sales Support and Calibration Services
Low-cost suppliers may offer limited technical support, spare parts, or recalibration options. Blood pressure machines require periodic calibration; without access to service networks, device accuracy degrades over time, affecting clinical reliability.
Counterfeit or Rebranded Devices
In some markets, counterfeit versions of reputable brands are repackaged with big cuffs. These devices may look authentic but lack quality assurance and pose significant safety risks. Always verify manufacturer authenticity and supply chain traceability.
Conclusion
To avoid these pitfalls, conduct thorough due diligence: verify cuff sizing standards, demand clinical validation reports, confirm regulatory approvals, audit IP rights, and choose suppliers with transparent manufacturing practices. Prioritizing quality and legal compliance ensures reliable patient care and long-term sourcing success.
Logistics & Compliance Guide for Big Cuff Blood Pressure Machine
This guide outlines essential logistics and compliance considerations for the distribution, handling, and use of the Big Cuff Blood Pressure Machine. Proper adherence ensures device safety, regulatory compliance, and optimal performance.
Regulatory Classification and Approvals
The Big Cuff Blood Pressure Machine is classified as a Class II medical device under the U.S. Food and Drug Administration (FDA) regulations. It must bear FDA 510(k) clearance, with a unique K-number listed in the device labeling. Equivalent regulatory approvals (e.g., CE marking under the EU MDR, Health Canada license, TGA inclusion) are required for international markets. Only distribute and use the device in regions where it holds valid regulatory certification.
Labeling and Packaging Requirements
All packaging must include:
– Device name and model number
– Manufacturer and authorized representative details
– FDA 510(k) number or equivalent
– Intended use statement
– Contraindications and warnings
– Single-use or reusable designation (if applicable)
– Storage conditions (temperature, humidity)
– Expiration date (if applicable)
– UDI (Unique Device Identifier) barcode compliant with FDA and international standards
Ensure labels are legible, accurate, and available in the local language(s) of the destination market.
Storage and Environmental Conditions
Store the Big Cuff Blood Pressure Machine in a dry, temperature-controlled environment between 15°C and 30°C (59°F to 86°F) and relative humidity of 30% to 70%. Avoid exposure to direct sunlight, extreme temperatures, moisture, dust, and corrosive substances. Devices must remain in sealed, original packaging until ready for use to prevent contamination or damage.
Shipping and Transportation
Use validated shipping methods that maintain environmental conditions throughout transit. Employ temperature-monitored packaging for long-distance or climate-variable routes. Packages should be labeled with “Fragile” and “Protect from Moisture” indicators. International shipments must comply with IATA/ICAO regulations for medical devices, including proper documentation (commercial invoice, packing list, certificates of conformity).
Import/Export Documentation
For cross-border logistics, ensure the following documents are prepared:
– Commercial invoice
– Packing list
– Certificate of Conformity (CE, FDA, etc.)
– Bill of lading or air waybill
– Import permits (as required by destination country)
– U.S. FDA prior notice (for U.S. imports)
– Export control classification number (ECCN), if applicable
Verify all documentation accuracy to prevent customs delays or shipment rejection.
Installation and User Training
The Big Cuff Blood Pressure Machine must be installed and operated only by trained healthcare professionals or authorized personnel. Provide comprehensive user manuals in local languages and conduct training on:
– Proper cuff sizing and placement
– Calibration verification
– Routine maintenance
– Troubleshooting common errors
– Infection control procedures
Maintain records of training completion for compliance audits.
Maintenance, Calibration, and Servicing
Adhere to the manufacturer’s recommended maintenance schedule. Perform calibration checks at least annually or as specified in the technical manual. Use only authorized service centers for repairs. Keep a log of all maintenance activities, including dates, technician names, and actions taken. Devices out of calibration must be removed from service immediately.
Post-Market Surveillance and Adverse Event Reporting
Implement a system to monitor device performance in the field. Report any malfunction, adverse event, or suspected serious injury to the relevant regulatory authority (e.g., FDA MedWatch, EUDAMED) within required timeframes (typically 10–30 days). Maintain records of complaints, investigations, and corrective actions for a minimum of 10 years.
Data Privacy and Cybersecurity (if applicable)
If the device collects or transmits patient data, ensure compliance with data protection regulations such as HIPAA (U.S.), GDPR (EU), or PIPEDA (Canada). Implement secure data transfer protocols, password protection, and audit trails. Conduct regular cybersecurity assessments if the device connects to networks or electronic health records.
Disposal and End-of-Life Management
Dispose of the Big Cuff Blood Pressure Machine in accordance with local medical waste regulations. Electronic components must be recycled through certified e-waste handlers. Follow manufacturer guidelines for decommissioning, including data wiping (if applicable) and physical deactivation. Document all disposal activities for compliance tracking.
Conclusion:
After thorough evaluation of available options, sourcing a big cuff blood pressure machine is a necessary and prudent decision to ensure accurate blood pressure measurements for individuals with larger arm circumferences. Standard cuffs often lead to falsely elevated readings when used on oversized arms, which can result in misdiagnosis or inappropriate treatment. Machines equipped with big cuffs (typically labeled as “large adult” or “thigh” cuffs, accommodating arm circumferences of 32–42 cm or more) provide reliable, clinically valid results, enhancing patient safety and care quality.
When sourcing such devices, it is important to consider factors including device accuracy, compatibility with big cuffs, ease of use, reliability, regulatory approvals (such as FDA or CE marking), and availability of validation studies. Leading brands like Omron, GE Healthcare, and Welch Allyn offer models with appropriate cuff sizes and proven performance in clinical settings.
In conclusion, investing in blood pressure machines with big cuff options supports inclusive and accurate healthcare delivery, particularly in diverse populations. Healthcare providers and procurement officers should prioritize this capability to ensure diagnostic accuracy and improve overall patient outcomes.