The global automatic chest compressor market is experiencing robust growth, driven by rising cardiovascular disease prevalence, increasing out-of-hospital cardiac arrest cases, and advancements in emergency medical care technologies. According to Mordor Intelligence, the global automated external defibrillator (AED) and mechanical CPR devices market—which includes automatic chest compressors—is projected to grow at a CAGR of over 7.5% from 2024 to 2029. Similarly, Grand View Research valued the global mechanical CPR devices market at USD 1.2 billion in 2023 and forecasts a CAGR of 8.1% from 2024 to 2030, citing expanding pre-hospital care infrastructure and growing emphasis on high-quality CPR delivery. As demand for reliable, consistent, and hands-free chest compression systems rises across ambulances, hospitals, and emergency response units, several manufacturers have emerged as key innovators in this critical segment of resuscitation technology. Below are the top seven automatic chest compressor manufacturers shaping the future of cardiac care.
Top 7 Automatic Chest Compressor Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
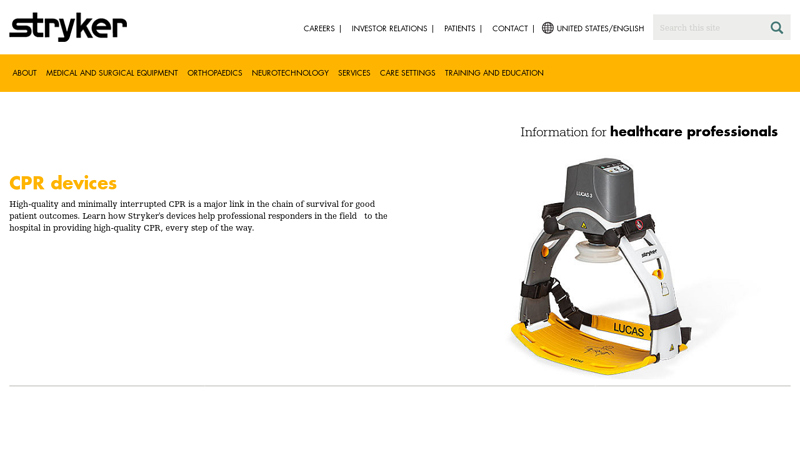
#1 CPR devices
Domain Est. 1995
Website: stryker.com
Key Highlights: Learn how Stryker’s devices help professional responders in the field to the hospital in providing high-quality CPR, every step of the way….
#2 Automated CPR
Domain Est. 1995
Website: zoll.com
Key Highlights: Our revolutionary AutoPulse® Resuscitation System uses complete thoracic compression to squeeze the entire chest. This type of CPR leads to superior cardiac ……
#3 Automatic CPR Machines To Help Save Lives
Domain Est. 1997
Website: michiganinstruments.com
Key Highlights: Our Life Stat and Thumper automatic CPR machines provide consistent external chest compression, giving your patient their best chance for survival….
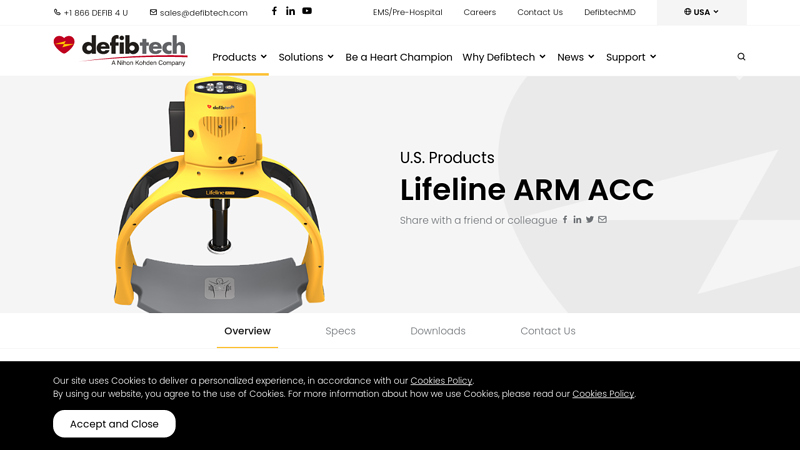
#4 Lifeline Automatic Chest Compressor
Domain Est. 2000
Website: defibtech.com
Key Highlights: The Lifeline ARM is an automated solution for providing victims of sudden cardiac arrest high-quality and continuous CPR….
#5 Automated Chest Compression
Domain Est. 2000
Website: aed.com
Key Highlights: Free delivery over $99 · 30-day returnsAutomated chest compression devices assist emergency responders by performing chest compressions for extended periods of time. These life-sa…
#6 LUCAS 3
Domain Est. 2002
Website: lucas-cpr.com
Key Highlights: You will learn: The basic orientation, use and handling of the LUCAS 3 device (referred to as LUCAS). How to apply the LUCAS device correctly on the patient ……
#7 corpuls cpr
Website: corpuls.world
Key Highlights: The corpuls cpr establishes a new generation of chest compressors for emergency services and clinics for the fastest possible patient care….
Expert Sourcing Insights for Automatic Chest Compressor

H2: Market Trends for Automatic Chest Compressors in 2026
The global market for automatic chest compressors (ACCs) is projected to experience significant growth and transformation by 2026, driven by advancements in emergency medical care, rising incidence of cardiac arrest, and increased adoption of pre-hospital life support technologies. Below are the key market trends shaping the automatic chest compressor landscape in 2026:
-
Increased Adoption in Pre-Hospital and In-Hospital Settings
Emergency medical services (EMS) and hospitals are increasingly integrating automatic chest compressors into standard resuscitation protocols. By 2026, the demand for consistent, high-quality cardiopulmonary resuscitation (CPR) during patient transport and in critical care units is expected to drive widespread adoption, especially in high-income countries with well-developed emergency response systems. -
Technological Advancements and Smart Integration
ACC devices are evolving with features such as real-time feedback, integration with defibrillators, wireless data transmission, and AI-driven performance optimization. In 2026, next-generation devices will likely include IoT connectivity for seamless data sharing with electronic health records (EHRs) and ambulance telemetry systems, improving post-event analysis and protocol refinement. -
Focus on Portability and Ease of Use
Manufacturers are emphasizing lightweight, compact, and battery-efficient designs to enhance usability in diverse environments—including ambulances, aircraft, and rural clinics. This trend supports deployment in resource-limited and mobile settings, expanding market reach beyond traditional hospital use. -
Regulatory Support and Clinical Guidelines
Updated international resuscitation guidelines (e.g., from the American Heart Association and European Resuscitation Council) continue to support high-quality CPR, indirectly promoting the use of automatic devices. Regulatory approvals and reimbursement policies in key markets (U.S., EU, Japan) are becoming more favorable, encouraging healthcare providers to invest in ACC systems. -
Growth in Emerging Markets
While North America and Europe remain dominant, Asia-Pacific and Latin America are emerging as high-growth regions. Increased healthcare spending, rising awareness of cardiac emergencies, and government initiatives to improve emergency care infrastructure are driving ACC adoption in countries like China, India, and Brazil. -
Competitive Landscape and Innovation
The market is witnessing intensified competition among key players such as ZOLL Medical, Stryker (LUCAS device), and Physio-Control. By 2026, innovation in device ergonomics, deployment speed, and integration with broader emergency response ecosystems will be critical differentiators. -
Focus on Training and Protocol Integration
There is growing emphasis on training EMS personnel and hospital staff in the effective use of ACCs. Simulation-based education and protocol standardization are expected to improve outcomes and justify capital investment in these devices.
In conclusion, the 2026 automatic chest compressor market is characterized by technological innovation, expanding applications, and stronger clinical and regulatory support. These trends collectively point toward sustained market growth and deeper integration of ACCs into global emergency medical systems.
Common Pitfalls Sourcing Automatic Chest Compressors (Quality, IP)
When sourcing automatic chest compressors—critical medical devices used during cardiopulmonary resuscitation (CPR)—organizations must carefully evaluate both quality and intellectual property (IP) considerations. Failing to do so can lead to operational failures, legal risks, and compromised patient outcomes. Below are key pitfalls to avoid:
Poor Quality Control and Device Reliability
One of the most significant risks is procuring devices with inadequate quality control. Low-quality automatic chest compressors may fail during emergency situations due to mechanical defects, inconsistent compression depth or rate, or battery issues. Devices that do not meet recognized medical standards (e.g., ISO 13485, IEC 60601) may lack reliability, durability, or safety features essential in high-stress environments. Always verify third-party certifications and conduct thorough performance testing before large-scale procurement.
Non-Compliance with Regulatory Standards
Automatic chest compressors must comply with regional medical device regulations such as FDA 510(k) clearance (U.S.), CE marking (EU), or other local approvals. Sourcing from manufacturers without proper regulatory documentation increases the risk of legal penalties, import restrictions, and liability in case of device failure. Ensure suppliers provide up-to-date regulatory compliance evidence and confirm alignment with your region’s medical device classification requirements.
Lack of Intellectual Property Verification
Failing to assess the intellectual property status of a device can expose buyers to legal disputes. Some suppliers, especially in competitive markets, may use designs or technologies that infringe on existing patents. Procuring such devices—even unknowingly—can result in cease-and-desist orders, product recalls, or litigation. Conduct due diligence by requesting IP documentation, such as proof of patent ownership or licensing agreements, and consider consulting legal experts to perform IP clearance searches.
Insufficient After-Sales Support and Spare Parts Availability
High-quality sourcing extends beyond the initial purchase. Many organizations overlook the importance of long-term support, including software updates, maintenance, training, and availability of consumables or spare parts. Devices from suppliers with weak service networks may become obsolete or unusable within a few years, increasing total cost of ownership and reducing operational readiness.
Inadequate Data Management and Integration Features
Modern automatic chest compressors often include data recording and integration capabilities with electronic health records or hospital systems. Devices with poor data security, lack of interoperability, or non-compliant software may violate privacy regulations (e.g., HIPAA, GDPR). Ensure the device supports secure data export, integrates with existing IT infrastructure, and adheres to cybersecurity best practices for medical devices.
Counterfeit or Substandard Components
In global supply chains, especially with direct sourcing from manufacturers in regions with lax enforcement, there is a risk of receiving devices built with counterfeit or substandard components. These can compromise performance and patient safety. Mitigate this by auditing suppliers, visiting manufacturing facilities when possible, and requiring transparency in the component supply chain.
By proactively addressing these pitfalls—prioritizing verified quality, regulatory compliance, and clear intellectual property rights—healthcare providers and procurement teams can ensure they source automatic chest compressors that are safe, reliable, and legally sound.

Logistics & Compliance Guide for Automatic Chest Compressor
This guide outlines the key logistics and regulatory compliance considerations for the distribution, import, and use of Automatic Chest Compressors (ACCs) in healthcare settings. These devices, also known as mechanical CPR devices, must meet stringent requirements to ensure patient safety and regulatory adherence.
Regulatory Classification and Approval
Automatic Chest Compressors are classified as Class II or Class III medical devices, depending on the jurisdiction. In the United States, the FDA typically classifies ACCs under Class II (e.g., 21 CFR 870.5550), requiring 510(k) premarket notification to demonstrate substantial equivalence. In the European Union, ACCs fall under Class IIb or III of the Medical Device Regulation (MDR) (EU) 2017/745, necessitating conformity assessment by a Notified Body. Manufacturers must obtain CE marking before placing the device on the EU market. Other regions such as Canada (Health Canada), Australia (TGA), and Japan (PMDA) have similar regulatory pathways requiring technical documentation, clinical evaluation, and quality management system certification (e.g., ISO 13485).
Quality Management System Requirements
Manufacturers and distributors must maintain a certified Quality Management System (QMS) compliant with ISO 13485:2016. This includes documented procedures for design control, risk management (ISO 14971), corrective and preventive actions (CAPA), and supplier controls. Regular internal audits and management reviews are essential. Distributors must ensure that ACCs are stored and handled in a manner that maintains device integrity and sterility, where applicable, and maintain traceability records for distribution and post-market surveillance.
Labeling and Documentation
All ACCs must be labeled according to regulatory standards, including the Unique Device Identifier (UDI) system required by the FDA and EU MDR. Labels must display the device name, model, serial number, manufacturer details, CE mark (if applicable), and expiration date (if relevant). The Instructions for Use (IFU) must be provided in the official language(s) of the destination country and include contraindications, warnings, precautions, and step-by-step operational guidance. Packaging must protect the device during transport and indicate any environmental sensitivities (e.g., temperature, humidity).
Import and Customs Clearance
Importers must comply with national customs regulations and provide documentation such as commercial invoices, packing lists, certificates of origin, and regulatory approvals (e.g., FDA 510(k) clearance letter, CE Certificate of Conformity). ACCs may be subject to import permits or special declarations due to their classification as medical devices. Harmonized System (HS) codes, such as 9018.90 for other medical devices, should be used accurately to avoid delays. Importers should also be aware of any import taxes, duties, or restrictions specific to medical equipment.
Shipping and Storage Conditions
ACCs and their accessories must be shipped and stored within specified temperature and humidity ranges, typically 10°C to 40°C and 30% to 70% relative humidity, unless otherwise stated by the manufacturer. Packaging should include shock and moisture protection indicators. Lithium-ion batteries, often used in ACCs, are subject to IATA Dangerous Goods Regulations (DGR) for air transport (UN3480, Class 9). Proper labeling, packaging, and documentation (e.g., Shipper’s Declaration for Dangerous Goods) are required when shipping batteries separately or installed in equipment.
Post-Market Surveillance and Vigilance Reporting
Manufacturers and authorized representatives are responsible for ongoing post-market surveillance (PMS) and reporting of adverse events. In the EU, serious incidents and field safety corrective actions (FSCAs) must be reported to the EUDAMED database under MDR requirements. In the U.S., manufacturers must submit Medical Device Reports (MDRs) to the FDA via the MAUDE database. Distributors should have procedures to report device malfunctions or patient injuries promptly and cooperate in product recalls or safety alerts.
Training and Clinical Implementation
Healthcare providers must receive comprehensive training on ACC operation, maintenance, and integration into resuscitation protocols. Training materials should be validated and available in local languages. Devices should be routinely inspected and serviced according to manufacturer guidelines. Hospitals and emergency services must document training completion and device usage to support clinical governance and compliance audits.
Environmental and End-of-Life Disposal
ACCs may contain electronic components and batteries that require proper disposal under WEEE (Waste Electrical and Electronic Equipment) directives in the EU or equivalent programs in other regions. Users should follow manufacturer instructions for decommissioning and recycling. Reusable components must be cleaned and disinfected according to validated protocols to prevent cross-contamination.
Adherence to this logistics and compliance guide ensures safe, legal, and effective deployment of Automatic Chest Compressors in emergency and critical care environments.
Conclusion for Sourcing an Automatic Chest Compressor:
Sourcing an automatic chest compressor is a critical decision that significantly impacts the quality and efficacy of cardiopulmonary resuscitation (CPR) in emergency and clinical settings. After evaluating key factors such as device reliability, consistency of compressions, ease of use, portability, compatibility with other life-support systems, regulatory approvals, and cost-effectiveness, it is evident that investing in a high-quality automatic chest compression device enhances patient outcomes during cardiac arrest events.
These devices ensure uninterrupted, guideline-compliant chest compressions, reduce rescuer fatigue, and allow medical personnel to focus on other essential interventions. When sourcing, it is vital to choose devices with proven clinical performance, strong after-sales support, and training availability. Ultimately, integrating automatic chest compressors into emergency response protocols represents a strategic advancement in resuscitation care, improving survival rates and aligning with best practices in modern emergency medicine.