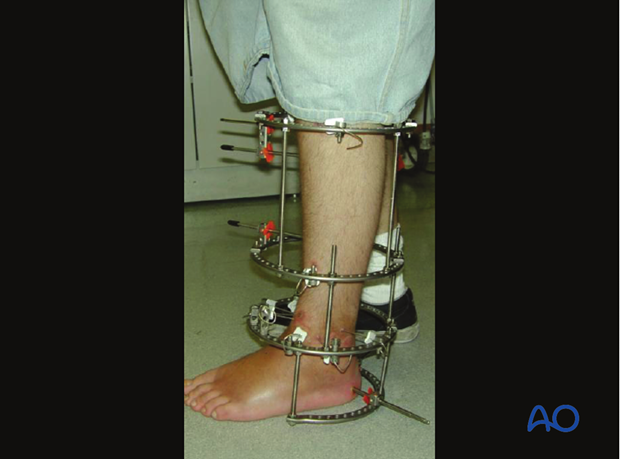
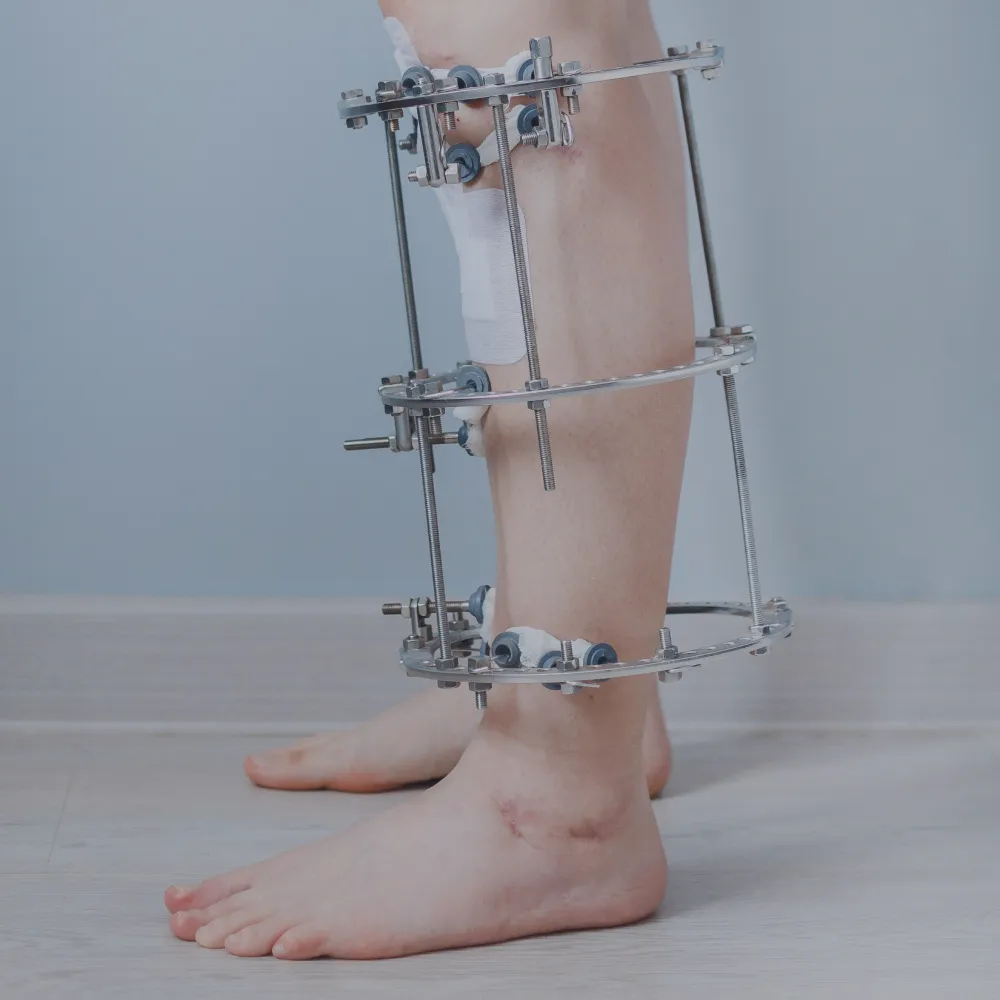
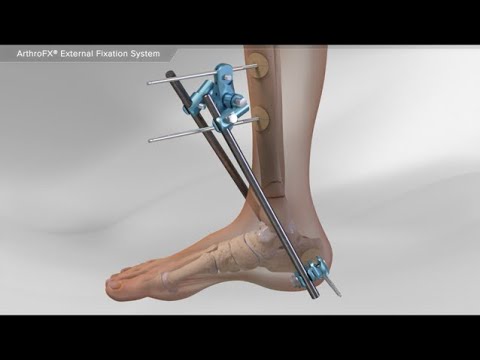
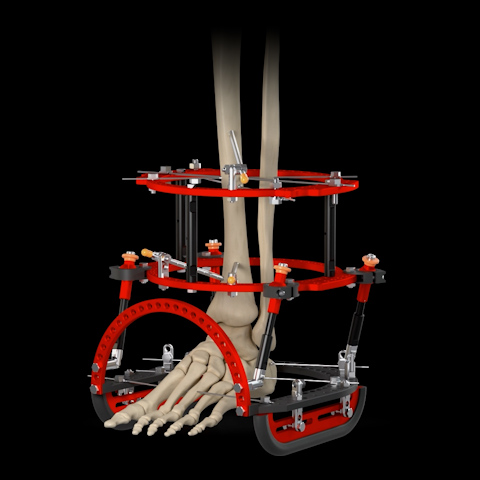
The global external ankle fixator market is experiencing steady expansion, driven by rising incidences of musculoskeletal injuries, increased trauma cases, and growing demand for minimally invasive orthopedic solutions. According to Grand View Research, the global external fixation market was valued at USD 1.8 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 6.3% from 2023 to 2030. This growth is further fueled by advancements in fixator design, rising geriatric populations prone to fractures, and expanding healthcare infrastructure in emerging economies. As demand for precision and stability in ankle and lower limb orthopedic procedures increases, manufacturers specializing in external ankle fixators are playing a critical role in shaping modern fracture management. Based on market presence, product innovation, regulatory approvals, and clinical adoption, the following nine manufacturers have emerged as key players in the ankle fixator landscape.
Top 9 Ankle Fixator External Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Salvation External Fixation System
Domain Est. 1995
Website: stryker.com
Key Highlights: Designed to address fractures, nonunion, and complex foot and ankle deformities including Charcot neuroarthropathy….
#2 ILIZAROV External Fixator
Domain Est. 1995
Website: smith-nephew.com
Key Highlights: The Ilizarov method delivers stability, flexibility and precision with near-immediate weight-bearing in complex fractures and deformities….
#3 External Fixation
Domain Est. 1997
Website: arthrex.com
Key Highlights: The ArthroFX® system was designed to give surgeons a simple, efficient, and versatile solution for temporary or definitive fixation. The system consists of:….
#4 External Fixation System
Domain Est. 1998
Website: acumed.net
Key Highlights: The Acumed External Fixation System provides innovative fracture management solutions for the upper extremities through a dedicated line of external fixators….
#5 Enovis™ Foot & Ankle
Domain Est. 1999
Website: enovis.com
Key Highlights: Our products cover a wide range of foot and ankle solutions, from total ankle replacement and static fixation to sustained dynamic compression and fusion, all ……
#6 Vilex
Domain Est. 1999
Website: vilex.com
Key Highlights: At Vilex, we’re breaking the mold of a traditional medical device company. We’re reimagining the future of foot and ankle care….
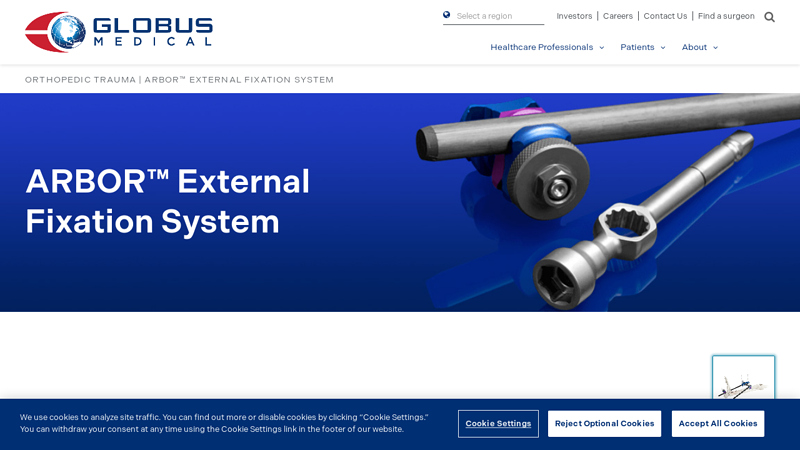
#7 ARBOR™ External Fixation System
Domain Est. 2003
Website: globusmedical.com
Key Highlights: The ARBOR™ External Fixation System provides streamlined instruments and implants for quick placement and simple handling during complex fracture care….
#8 External Fixation Systems
Domain Est. 2010
Website: paragon28.com
Key Highlights: Paragon 28 has designed systems for TC Fusions to help you treat your challenging patient populations! Learn more about our reconstruction portfolio today!…
#9 XtraFix® External Fixation System
Domain Est. 2014
Website: zimmerbiomet.com
Key Highlights: The XtraFix External Fixation System allows surgeons to build rigid constructs for long bone and pelvic fractures with few components and steps….
Expert Sourcing Insights for Ankle Fixator External
H2: 2026 Market Trends for Ankle Fixator External
The global external ankle fixator market in 2026 is poised for steady growth, driven by converging demographic, technological, and clinical factors. Key trends shaping the landscape include:
-
Aging Population & Rise in Complex Trauma: The increasing global geriatric population directly correlates with higher incidences of osteoporosis, fragility fractures (like Pilon fractures), and comorbidities requiring complex ankle stabilization. Simultaneously, high-energy trauma (e.g., motor vehicle accidents, falls) remains a significant driver, particularly in developing economies experiencing rapid urbanization. This dual pressure expands the primary patient pool for external fixation.
-
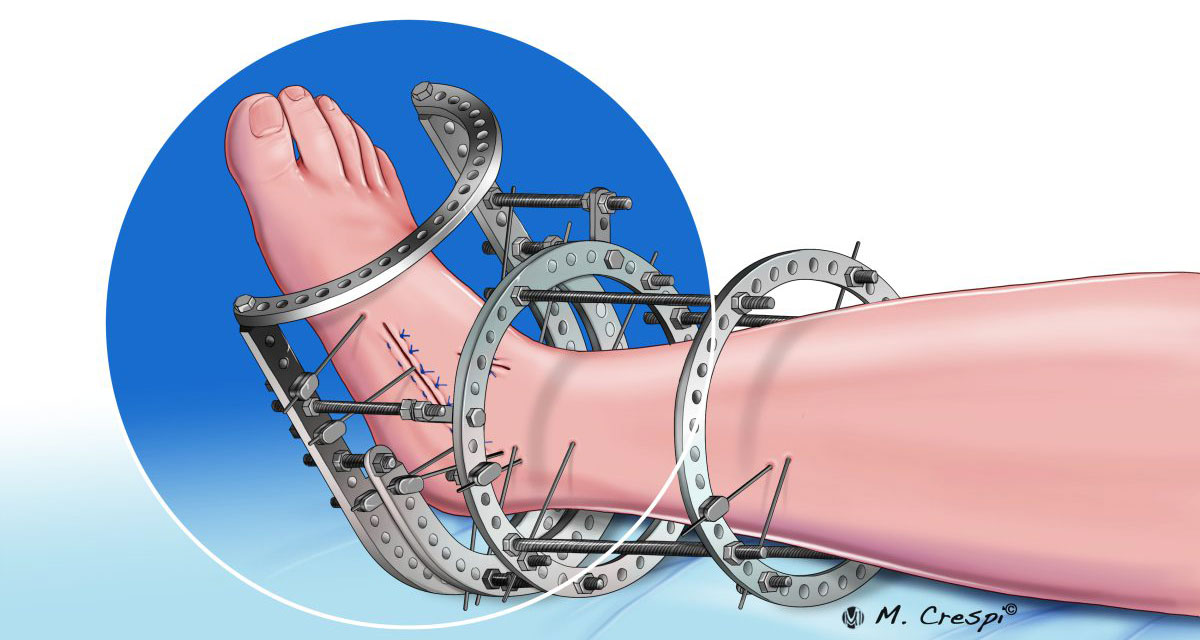
Shift Towards Circular and Hybrid Frames (Ilizarov, Taylor Spatial Frame): While simple uniplanar frames persist, there’s a pronounced trend favoring circular (e.g., Ilizarov) and hexapod (e.g., Taylor Spatial Frame – TSF) fixators. These offer superior stability for complex reconstructions, precise multiplanar correction of deformities (malunions, non-unions, Charcot foot/ankle), limb lengthening, and management of infected non-unions or severe soft tissue compromise. Their biomechanical advantages are increasingly recognized, driving adoption despite higher cost and complexity.
-
Integration of Advanced Imaging and Navigation: The use of pre-operative 3D CT planning and intra-operative fluoroscopy (including 3D C-arm) is becoming standard for frame application, especially with hexapod systems. This enhances precision in pin placement, frame construction, and alignment correction, reducing complications and improving outcomes. Computer-assisted planning software for TSF and similar systems is becoming more sophisticated and user-friendly.
-
Focus on Minimally Invasive Techniques and Pin Site Care: Surgeons are increasingly adopting percutaneous techniques for pin/wire insertion to minimize soft tissue damage and infection risk. Concurrently, significant R&D and clinical focus is on improving pin site care protocols and developing antimicrobial-coated pins (e.g., silver, chlorhexidine) to combat the persistent challenge of pin site infections, a major cause of morbidity and treatment failure.
-
Patient-Centric Design and Comfort: Recognizing the burden of external fixation (bulk, discomfort, hygiene challenges), manufacturers are focusing on lighter materials (titanium, advanced composites), low-profile clamps, and improved frame configurations to enhance patient comfort, mobility, and psychological well-being during the often prolonged treatment period.
-
Consolidation and Competition: The market features established players (e.g., Smith & Nephew, Orthofix [now part of LivaNova], Stryker, Zimmer Biomet) competing with specialized innovators (e.g., Ortho-SUV Ltd. – TSF). Consolidation within the medtech sector may impact distribution and R&D focus. Competition drives innovation in frame design, software, and ancillary products (pins, clamps).
-
Cost-Effectiveness and Reimbursement Pressures: Healthcare systems globally are scrutinizing costs. While advanced frames (TSF) offer superior correction capabilities, their higher initial cost requires justification through demonstrable clinical benefits (reduced revision rates, faster healing, fewer complications). Reimbursement policies significantly influence adoption rates, particularly in price-sensitive markets.
-
Expanding Applications in Diabetic Foot and Charcot Neuroarthropathy: The rising prevalence of diabetes fuels demand for external fixation in managing complex Charcot foot and ankle deformities, infected diabetic foot ulcers requiring stabilization, and limb salvage procedures where internal fixation is contraindicated due to poor bone quality or infection.
Conclusion for 2026:
The external ankle fixator market in 2026 will be characterized by growth driven by demographic and trauma trends, a clear technological shift towards advanced circular/hexapod frames for complex cases, and intense focus on improving precision (imaging/navigation), reducing complications (pin site management), and enhancing patient experience. Success for manufacturers and providers will depend on offering integrated solutions (frame + software + pins), demonstrating cost-effectiveness, and supporting surgeons in mastering these sophisticated techniques. The market will remain niche compared to internal fixation but is indispensable for complex reconstructions and specific patient populations.

Common Pitfalls When Sourcing Ankle Fixator External Devices: Quality and Intellectual Property Risks
Sourcing external ankle fixators—particularly from international or low-cost manufacturers—can present significant challenges related to product quality and intellectual property (IP) protection. Avoiding these pitfalls is crucial for ensuring patient safety, regulatory compliance, and commercial success.
Quality-Related Pitfalls
Insufficient Material and Manufacturing Standards
External fixators require high-grade biocompatible materials (e.g., medical-grade titanium or stainless steel) and precision engineering. Sourcing from suppliers that cut corners on material specifications or lack ISO 13485 certification increases the risk of device failure, corrosion, or adverse tissue reactions. Always verify material certifications and conduct independent mechanical testing.
Inconsistent Sterility and Packaging Controls
As sterile medical devices, external fixators must be manufactured and packaged under controlled cleanroom conditions. Suppliers may claim compliance but fail to maintain consistent sterility assurance levels (SAL). Audit sterilization processes and review batch release documentation before scaling procurement.
Lack of Clinical Validation and Biocompatibility Testing
Some suppliers offer “custom” or “OEM” fixators without providing full biocompatibility data (e.g., ISO 10993 testing) or clinical performance history. Relying on untested designs risks patient harm and regulatory rejection during submissions to the FDA, CE, or other authorities.
Poor Design for Usability and Patient Safety
Fixators with poor ergonomics, sharp edges, or suboptimal clamping mechanisms can lead to complications like pin loosening or soft tissue damage. Evaluate prototypes for surgeon usability and patient comfort before mass production.
Intellectual Property (IP) Risks
Inadvertent Infringement of Patented Designs
Many external fixator systems (e.g., Ilizarov, Taylor Spatial Frame) are protected by active patents. Sourcing generic versions that closely mimic patented mechanisms—even if modified slightly—can expose your company to infringement lawsuits. Conduct thorough freedom-to-operate (FTO) analyses before engaging suppliers.
Supplier Claims of “IP-Free” or “Open-Design” Products
Some manufacturers falsely claim their designs are IP-free to attract business. These claims are often unverified and can mislead buyers into adopting infringing technology. Require legal indemnification clauses in contracts and perform independent patent landscaping.
Lack of IP Ownership in Custom Development
When co-developing a new fixator design with a supplier, failure to clearly define IP ownership in the contract may result in shared or supplier-held rights. Ensure agreements explicitly transfer all design rights, tooling IP, and regulatory documentation to your organization.
Counterfeit or Grey Market Devices
Unverified suppliers may offer branded fixators at suspiciously low prices, indicating counterfeit or diverted products. These devices lack traceability, proper labeling, and regulatory approvals. Only source through authorized distributors or directly from OEMs with verifiable chain-of-custody records.
Mitigating these pitfalls requires due diligence, third-party audits, robust contractual safeguards, and proactive IP management throughout the sourcing process.

Logistics & Compliance Guide for Ankle Fixator External
This guide outlines the key logistics and regulatory compliance considerations for the distribution, handling, and sale of the Ankle Fixator External medical device. Adherence to these guidelines ensures patient safety, legal compliance, and efficient supply chain operations.
Regulatory Classification & Registration
The Ankle Fixator External is classified as a medical device and must comply with regional regulatory frameworks. Typically, it falls under Class IIa or IIb (EU MDR) or Class II (U.S. FDA), depending on its intended use and risk profile. Ensure the device is:
- Registered with the appropriate regulatory bodies (e.g., FDA in the United States, notified bodies under EU MDR in Europe, Health Canada, TGA in Australia).
- Accompanied by a valid CE Mark (for EU), FDA 510(k) clearance or PMA approval (for U.S.), or equivalent certification for other markets.
- Labeled with required regulatory symbols, UDI (Unique Device Identifier), and traceability information.
Labeling and Packaging Requirements
All packaging must meet international and local regulatory standards:
- Include legible labeling with product name, model number, UDI, batch/lot number, expiration date (if applicable), sterile status, and manufacturer details.
- Display appropriate symbols per ISO 15223-1 (e.g., sterile, single-use, do not reuse).
- Provide multilingual labeling for distribution across different regions (e.g., EU requires all 24 official EU languages).
- Ensure tamper-evident and sterile barrier packaging to maintain product integrity.
Storage and Handling Conditions
Proper storage is critical to maintain device safety and performance:
- Store in a clean, dry, temperature-controlled environment (typically 15°C to 30°C).
- Avoid exposure to direct sunlight, moisture, and extreme temperatures.
- Keep devices in original packaging until point of use to prevent contamination or damage.
- Implement a first-expiry, first-out (FEFO) inventory rotation system if shelf life applies.
Transportation and Distribution
Ensure safe and compliant transport across the supply chain:
- Use validated packaging to protect devices during transit, especially for sterile or fragile components.
- Maintain temperature and humidity controls where required.
- Partner with logistics providers experienced in medical device shipping and compliant with GDP (Good Distribution Practice).
- Monitor shipments with tracking systems and ensure chain of custody documentation.
Import/Export Documentation
Cross-border movement requires accurate documentation:
- Provide commercial invoices, packing lists, certificates of origin, and regulatory certificates (e.g., Certificate of Free Sale, CE Certificate).
- Comply with import regulations in destination countries (e.g., import licenses, local representative requirements).
- Ensure Harmonized System (HS) code classification is accurate (e.g., 9021.29 for orthopedic appliances).
Post-Market Surveillance & Vigilance Reporting
Compliance continues after product release:
- Establish a post-market surveillance (PMS) system to collect and analyze feedback, complaints, and adverse events.
- Report serious incidents and field safety corrective actions (FSCAs) to relevant authorities within mandated timeframes (e.g., 15 days under EU MDR).
- Maintain comprehensive technical documentation and update it regularly.
Training and Documentation
Ensure all personnel involved in logistics and distribution are trained:
- Train staff on handling sterile medical devices, regulatory requirements, and recall procedures.
- Maintain records of training, device distribution, and quality audits.
- Keep device master records (DMR) and quality management system (QMS) documentation up to date (per ISO 13485).
Adhering to this logistics and compliance guide ensures safe, legal, and efficient distribution of the Ankle Fixator External, minimizing risk and supporting regulatory compliance across global markets.
Conclusion:
Sourcing an external ankle fixator requires careful evaluation of clinical requirements, patient needs, regulatory standards, and supplier reliability. It is essential to prioritize devices that offer stability, adjustability, biocompatibility, and ease of use to ensure optimal patient outcomes. Conducting thorough due diligence on suppliers—including certification, quality control, delivery timelines, and post-sale support—will help secure a safe and cost-effective solution. Ultimately, collaboration between medical professionals, procurement teams, and trusted manufacturers is key to successfully sourcing external ankle fixators that meet both clinical and operational needs.